Monogenic Obesity as” De Novo Mutation” in ADCY Detection on Pediatrician Age. Characterization of a Case
Abstract
Obesity
constitutes one of the most frequent reasons for consultation in clinical
practice, both in adults and in the paediatric field.
The
susceptibility of each patient to the development of obesity, expressed as an
excessive accumulation of adipose tissue as a phenotypic manifestation common
to different conditions, environments and under the determinants of a marked
genotype.
In
recent years, the pathophysiological basis of appetite control, satiety and
energy expenditure has been clarified. The underlying genetic, non-genetic and
endocrinological causes and conditions have also been identified.
Among
the advances in the study of aetiologies, a growing number of so-called
monogenic aetiologies have been characterized in recent years, thanks to
increased knowledge of the pathophysiological basis of obesity.
The
causes of monogenic obesities preferentially affect genes involved in the
appetite-satiety regulation of the leptin-melanocortin pathway: Leptin (LEP), Leptin
Receptor (LEPR), Proopiomelanocortin (POMC), Proprotein Convertase Subtilisin-Kexin-like
Proprotein Convertase 1 (PCSK1), Melanocortin Receptor 4 (MC4R), Src-Homology-2B
Adaptor Protein 1 (SH2B1), Steroid Receptor Coactivator 1 (SRC1), Carboxypeptidase
E (CPE), and Steroid Receptor Coactivator 1 (SRC1) and G Protein Alpha Activity-Stimulating
Polypeptide 1 (GNAS).
Keywords: Monogenic,
Pediatric, Obesity, Protein
Abbreviations: LEP: Leptin; LEPR: Leptin Receptor; POMC: Proopiomelanocortin; PCSK1: Proprotein Convertase Subtilisin-Kexin-like Proprotein Convertase 1; MC4R; Melanocortin Receptor 4; SH2B1: Src-Homology-2B Adaptor Protein 1; SRC1: Steroid Receptor Coactivator 1; CPE: Carboxypeptidase E; SRC1: Steroid Receptor Coactivator 1; GNAS: G Protein Alpha Activity-Stimulating Polypeptide 1
1. Introduction
Obesity
constitutes one of the most frequent reasons for consultation in clinical
practice, both in adults and in the paediatric field.
The
susceptibility of each patient to the development of obesity, expressed as an
excessive accumulation of adipose tissue as a phenotypic manifestation common
to different conditions, environments and under the determinants of a marked
genotype.
In
recent years, the pathophysiological basis of appetite control, satiety and
energy expenditure has been clarified. The underlying genetic, non-genetic and
endocrinological causes and conditions have also been identified.
Among
the advances in the study of aetiologies, a growing number of so-called
monogenic aetiologies have been characterized in recent years, thanks to
increased knowledge of the pathophysiological basis of obesity.
The
causes of monogenic obesities preferentially affect genes involved in the
appetite-satiety regulation of the leptin-melanocortin pathway: Leptin (LEP),
Leptin Receptor (LEPR), Proopiomelanocortin (POMC), Proprotein Convertase
Subtilisin-Kexin-like Proprotein Convertase 1 (PCSK1), Melanocortin Receptor 4
(MC4R), Src-Homology-2B Adaptor Protein 1 (SH2B1), Steroid Receptor Coactivator
1 (SRC1), Carboxypeptidase E (CPE), and Steroid Receptor Coactivator 1 (SRC1)
and G Protein Alpha Activity-Stimulating Polypeptide 1 (GNAS). Of particular
interest are the reviews carried out by Martos-Moreno and collaborators in this regard, as well
as highlighting some widely studied syndromic entities in pediatrics, such as
Bardet-Biedl and Alstrom syndromes1,2.
It is therefore necessary to thoroughly investigate the cause of a patient's obesity in order to be able, if necessary, to individualize the therapeutic approach: from the well-known hygiene and dietary measures to pharmacological advances, such as recombinant leptin, the melanocortin analogue setmelanotide and GLP-1 analogues.
2. Case Report
We
present the case of a boy currently 11 years old with monogenic obesity
presenting a pathogenic variant in heterozygous variation in the ADCY3 gene
(600291) on chromosome 2p23. There are only 10 families described in the
literature.
This
is a child being followed up at the paediatric endocrinology clinic since the
age of 8 months of age for presenting a high height and weight gain well above
height, which became more evident after the introduction of the child's family
originated in Morocco.
The
family origin is Moroccan. There is no consanguinity. The parents are healthy
and the siblings are not affected by significant obesity. The pregnancy was normal
and the somatometry at birth for 39 weeks of gestation was adequate: weight of
3300 grams (+0.85 SDS) and height of 50 cm (+0.3 SDS).
The
physical examination at the first consultation already drew attention with a
somatometry at 8 months of life of Weight: 17.9kg (>2 SDS) Height: 81.9cm
(>2 SDS) and BMI: 26.6 Kg/m2 (+ 5.5 SDS) Body surface area: 0.6 m2.
The
phenotype is peculiar, cushingoid in appearance. Good general condition. No
rashes or petechiae, no skin patches, no palpable goitre. Pubarche I Penis
buried in fat Gonads prepubertal.
Exclusive
breastfeeding until 5 months of age, after which conventional complementary
feeding is introduced, with progressive increase in weight/height. He was
referred to the clinic for primary care studies.
We completed basic studies without finding any altered parameters. Intensive follow-up with hygienic-dietary measures was initiated. No success over the years (see Graph 1 and Figure 1) in containing the BMI.
Graph 1: Intensive follow-up with hygienic-dietary measures was initiated. No success over the years.
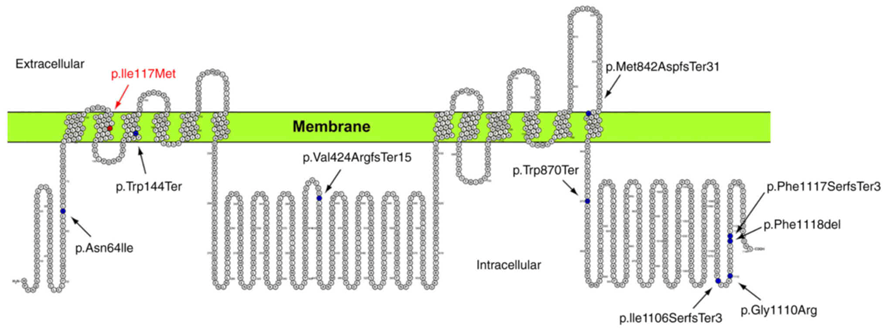
Figure 1: Representation of genotype and ADCY3 gene.
Pituitary
function and thyroid function are preserved. The cushingoid phenotype is
maintained. In addition, she is under multidisciplinary follow-up for mild
intellectual disability, language disorder. Given the suspicion of a syndromic
condition, a karyotype and X-fragment study was performed and was negative.
However,
given the high clinical suspicion, in 2023 an extension of the genetic study
was requested at the external reference laboratory qGenomics. A massive
sequencing of the exome was performed, detecting the heterozygous nonsense
variant in the ADCY3 gene compatible with monogenic obesity type OMMIN 617885.
Moreover, the segregation study in parental samples has shown that the variant
originated de novo.
The ADCY3 gene encodes adenylyl cyclase 3, an enzyme that catalyzes the production of Cyclic Adenosine Monophosphate (cAMP). This enzyme plays a role in various physiological and metabolic processes. Mutations in ADCY3 have been linked to obesity and other metabolic disorders.
2.1. Key Features of ADCY3
Enzyme: ADCY3
is a membrane-associated enzyme that converts ATP to cAMP, a crucial second
messenger in cellular signaling pathways.
Expression:
It
is widely expressed in various human tissues, including the brain, heart, and
skeletal muscle.
Function:
ADCY3
is involved in a number of physiological processes, including brown fat
function, olfaction, and glucose homeostasis.
Obesity: Genetic
studies have demonstrated that ADCY3 polymorphisms are associated with obesity
in both European and Chinese populations. Loss-of-function variants in ADCY3
have been linked to increased risk of obesity and type 2 diabetes.
Epigenetics: Increased DNA methylation in the ADCY3 gene has been implicated in the pathogenesis of obesity.
2.2. Therapeutic Target
ADCY3 dysfunction has been suggested as a potential therapeutic target for anti-obesity drug development. In summary, ADCY3 is a key enzyme involved in cellular signaling, and its function is crucial for various metabolic and physiological processes. Genetic variants and dysfunction of ADCY3 have been linked to obesity and other metabolic disorders, making it a potential target for drug development3. (Saeed et al., 2018) (4).
Stergiakouli et al.4 performed
genomewide association studies involving BMI and height-adjusted BMI ('BMI[x]')
in a British and a Dutch cohort of children (ALSPAC and Generation R,
respectively). A missense SNP in the ADCY3 gene (rs11676272) showed evidence of
genomewide significance in the ASPAC cohort (p = 4 x 10(-9)), but only when
height was adjusted for, and this association was replicated in the Generation
R cohort (p = 0.0001). Analysis of ADCY3 expression in lymphoblastoid cell
lines showed a strong association between the rare (adiposity-increasing) G
allele at rs11676272 and reduced levels of ADCY3 expression (p = 1 x 10(-53)).
The authors concluded that rs11676272 represents a functional polymorphism in
ADCY3 that is associated with fat mass in childhood, and noted that the effect
size is comparable to common variation at the FTO locus (610966), but only when
height is correctly taken into account.
Grarup et al.5 identified a splice site
variant in the ADCY3 gene (600291.0001) with a minor allele frequency of 2.3%
in a Greenlandic study population of 4,038 individuals. The 7 homozygous
carriers had BMIs, body fat percentages, and waist circumferences that were all
significantly greater than those of the remaining study population, and an
association with type 2 diabetes (T2D) was found that remained significant
after adjustment for BMI. Homozygous carriers showed severely decreased RNA
expression, whereas heterozygotes had an intermediate expression level. In
addition, analysis of exome-sequencing data from 18,176 samples from the
Accelerating Medicines Partnership Type 2 Diabetes Knowledge Portal (AMP-T2D)
database identified heterozygosity for 7 predicted loss-of-function variants in
ADCY3 in 8 individuals, and there was enrichment of carriers of ADCY3 variants
among T2D cases compared to nondiabetic controls (odds ratio, 8.6; p = 0.044).
A number sign (#) is used with this entry
because of evidence that susceptibility to obesity is conferred by homozygous
or compound heterozygous variation in the ADCY3 gene (600291) on chromosome
2p23.
3. Discussion
Patients with biallelic mutations in the ADCY3 gene show hyperphagia within the first 2 years of life and develop severe obesity. Other features include hyposmia or anosmia, and some patients exhibit mild to moderate intellectual disability3.
3.1.
Clinical Features
Saeed et al.3 reported 4 children with severe early-onset obesity from 3 consanguineous Pakistani families, as well as an obese boy from a European American family. Hyperphagia in the probands was first reported between 6 months and 2 years of age. Other features included anosmia in 3 patients and hyposmia in 2, and 2 probands exhibited mild to moderate intellectual disability. In addition, the oldest patient, a 15-year-old Pakistani girl, had undergone menarche at age 14 years but had no subsequent menstrual cycles; she also exhibited hyperlipidemia and insulin resistance. No dysmorphic features were observed in the patients. Family members were all normal weight, except for the parents of 1 of the Pakistani patients (family 2), who were upper-middle class and accustomed to fat- and carbohydrate-enriched food.
3.2. Molecular Genetics
Stergiakouli et al.4 performed
genomewide association studies involving BMI and height-adjusted BMI ('BMI[x]')
in a British and a Dutch cohort of children (ALSPAC and Generation R,
respectively). A missense SNP in the ADCY3 gene (rs11676272) showed evidence of
genomewide significance in the ASPAC cohort (p = 4 x 10(-9)), but only when
height was adjusted for, and this association was replicated in the Generation
R cohort (p = 0.0001). Analysis of ADCY3 expression in lymphoblastoid cell
lines showed a strong association between the rare (adiposity-increasing) G
allele at rs11676272 and reduced levels of ADCY3 expression (p = 1 x 10(-53)).
The authors concluded that rs11676272 represents a functional polymorphism in
ADCY3 that is associated with fat mass in childhood, and noted that the effect
size is comparable to common variation at the FTO locus (610966), but only when
height is correctly taken into account.
Saeed et al.3 characterized the
rs11676272 (S107P) variant in BHK cells, and observed that the mutant showed
catalytic activity comparable to that of wildtype ADCY3.
Grarup et al.5 identified a splice site
variant in the ADCY3 gene (600291.0001) with a minor allele frequency of 2.3%
in a Greenlandic study population of 4,038 individuals. The 7 homozygous
carriers had BMIs, body fat percentages, and waist circumferences that were all
significantly greater than those of the remaining study population, and an
association with type 2 diabetes (T2D) was found that remained significant
after adjustment for BMI. Homozygous carriers showed severely decreased RNA
expression, whereas heterozygotes had an intermediate expression level. In
addition, analysis of exome-sequencing data from 18,176 samples from the
Accelerating Medicines Partnership Type 2 Diabetes Knowledge Portal (AMP-T2D)
database identified heterozygosity for 7 predicted loss-of-function variants in
ADCY3 in 8 individuals, and there was enrichment of carriers of ADCY3 variants
among T2D cases compared to nondiabetic controls (odds ratio, 8.6; p = 0.044).
In 138 probands with severe early-onset obesity from consanguineous Pakistani families, Saeed et al.3 performed whole-exome sequencing, and in 4 severely obese children from 3 families, they identified homozygous mutations in the ADCY3 gene (see, e.g., 600291.0002 and 600291.0003). Independent whole-exome sequencing in an obese boy from a nonconsanguineous European American family revealed compound heterozygosity for mutations in ADCY3 (600291.0004 and 600291.0005). All mutations occurred at highly conserved sites, and all segregated with disease in the respective families.
4. References
- Martos-Moreno GA, Argente J. Obesidades monogénicas
en la infancia: hacia una medicina de precisión Rev Esp Endocrinol Pediatr.
2022;13: 23-32.
- Martos-Moreno GA, Argente J. Obesidades pediátricas:
de la lactancia a la adolescencia. An Pediatr (Barc). 2011;75: 63.
- Saeed S, Bonnefond A,
Tamanini F, et al. Loss-of-function
mutations in ADCY3 cause monogenic severe obesity. Nature Genet.
2018;50: 175-179.
- Stergiakouli E, Gaillard
R, Tavare JM, et al. Genome-wide
association study of height-adjusted BMI in childhood identifies functional
variant in ADCY3. Obesity, 2014;22: 2252-2259.
- Grarup N, Moltke I
andersen MK, et al. Loss-of-function
variants in ADCY3 increase risk of obesity and type 2 diabetes. Nature
Genet. 2018;50: 172-174.