Father with Alzheimer’s Disease Increases the Risk of Cervical Adenocarcinoma: Two-Sample Mendelian Randomization Study
Abstract
Objective: Based
on the two-sample Mendelian randomization method, this study explores the
causal relationship between Alzheimer 's family history and cervical malignant
tumors.
Methods: We
extracted data from the IEU Open GWAS database and Finnish R9 database from the
Alzheimer family history, cervical malignant tumors (such as cervical squamous
cell carcinoma, cervical adenocarcinoma and other cervical malignant tumors).
By setting the correlation strength and eliminating the linkage disequilibrium,
the instrumental variables are screened out. Inverse variance weighting (IVW)
was used as the main analysis method, supplemented by weighted median, MR-Egger
and weighted pattern as supplementary analysis methods to evaluate causal
effects. In addition, the Cochran Q test of IVW and MR-Egger was used for
heterogeneity analysis and the MR-Egger intercept and MR-PRESSO analysis method
were used for pleiotropic test. At the same time, the stability of the results
was evaluated using the leave-one-out method. Finally, meta-analysis was used
to further clarify the causal relationship between the two.
Results: The
results of MR analysis between father's disease history and cervical
adenocarcinoma suggested that father 's Alzheimer 's disease increased the risk
of cervical adenocarcinoma (OR=1.99; 95 % CI:1.04-3.80, P =0.037) and no association was found in the MR analysis
between the other two samples. Heterogeneity test suggested that there was no
heterogeneity in the study. The results of the leave-one-out method and the
multi-effect test showed stability.
Conclusion: The
study confirmed that the father with Alzheimer 's disease will increase the
risk of cervical adenocarcinoma, but has no significant effect on cervical
squamous cell carcinoma. In addition, the mother with Alzheimer 's disease or
other family history of Alzheimer 's disease has no significant effect on the
occurrence of cervical malignant tumors. In the future clinical work, we should
appropriately strengthen the monitoring and follow-up of women with Alzheimer
's disease in their fathers to prevent or detect cervical malignant lesions
early.
Keywords: Cervical cancer, Alzheimer's
disease, Mendelian randomization
1.
Introduction
Alzheimer's
disease (AD) is a progressive neurodegenerative disorder that is the most
common cause of dementia among older adults. The “World Alzheimer Report 2023”
indicates that in 2019, there were 55 million people globally living with
dementia and this number is projected to increase to 139 million by 20501. Genetically, AD can be classified into
sporadic AD (SAD) and familial AD (FAD) based on its heritability2, The familial form of AD, which accounts
for about 5-10% of all cases, typically has an earlier onset and follows a
Mendelian pattern of inheritance3.
On the other hand, cervical cancer is the fourth most common cancer in women,
with an estimated 570,000 new cases in 2018 globally. Despite the introduction
of human papillomavirus vaccines and improved screening methods, there were
over 311,000 deaths due to cervical cancer in the same year4. Both diseases pose significant challenges
in terms of early detection and intervention, highlighting the need for a
deeper understanding of their etiologies.
Mendelian
Randomization (MR) has emerged as a robust method for investigating the causal
relationships between genetic variants and complex diseases. By using genetic
variants as instrumental variables, MR leverages the random assortment of
alleles at conception to infer causality from observational data while
minimizing confounding and reverse causation5.
For instance, MR studies have provided insights into risk factors for coronary
artery disease and type 2 diabetes that have been validated through randomized
controlled trials6.To be an effective tool for causal inference in
MR studies, genetic variation must satisfy three core assumptions : ( 1 )
genetic variation as an instrumental variable must be truly related to exposure
( Alzheimer 's family history or cervical malignancy ) , ( 2 ) Genetic
variation was not associated with exposure-outcome confounding factors.,( 3 )
Genetic variation only affects the outcome through exposure and has nothing to
do with other pathways. In this study, we aimed to explore the causal
relationships between genetic variants and both familial AD and cervical cancer
using MR analysis. Our findings suggest potential causal relationships between
specific genetic variants and both familial AD and cervical cancer. our study
contributes to a better understanding of the genetic underpinnings of familial
AD and cervical cancer. The application of MR provides a powerful approach to
unraveling complex disease mechanisms that could pave the way for novel
therapeutic targets.
2. Method
2.1. Data source
In
this study, the IEU Open GWAS database and the Finnish R9 database data were
used for two-sample MR analysis to explore the causal relationship between the
family history of Alzheimer 's disease and cervical malignant tumors. The data
are summarized in (Table 1).
|
Variable |
ID |
Sample
size |
Multitude |
Sex |
The
year of publication |
|
Father has Alzheimer's disease |
ebi-a-GCST005920 |
260,279 |
Europe |
Mixed |
2018 |
|
Mother has Alzheimer's disease. |
ebi-a-GCST005923 |
288,676 |
Europe |
Mixed |
2018 |
|
Family
history of Alzheimer's |
ebi-a-GCST005921 |
314,278 |
Europe |
Mixed |
2018 |
|
Cervical adenocarcinoma |
FinlandR9 |
167301 |
Europe |
F |
2023 |
|
cervical squamous cell carcinoma |
FinlandR9 |
167353 |
Europe |
F |
2023 |
|
Cervical malignancy |
FinlandR9 |
167558 |
Europe |
F |
2023 |
2.2. Selection of instrumental variables
In order to obtain SNPs
significantly associated with exposure, we set the P value to 5 × 10-8 as the
genome-wide significance threshold. At the same time, because the existence of
linkage disequilibrium (LD) will lead to the deviation of the final analysis
results, we set that the LD of SNPs significantly related to exposure should
meet r 2 10 000. Our MR analysis excluded palindromic SNPs with a moderate
allele frequency. In addition, we performed F-statistic to quantify the
strength of the genetic tool for all SNPs. The calculation formula is (β2 /
se2) and SNPs with F-statistic values<10 are excluded. The remaining SNPs
are considered to be strong variable tools for further analysis.
2.3.
MR analysis
Statistical analysis was
performed using the R programming language (version 4.3.0). MR analysis was
performed using the ‘TwoSampleMR’ software package (version 0.5.6) and the ‘MRPRESSO’
software package (version 1.0) was used to apply MRPRESSO analysis to identify
outliers and detect pleiotropic effects. For the causal analysis between
exposure and outcome, we used the random effect inverse variance weighting
method (IVW) as the main analysis method and MR-Egger, weighted median and
weighted mode as supplementary analysis methods. Since the outcome indicators
are all dichotomous variables, we obtained the corresponding odds ratio (OR)
and 95% confidence interval (95% CI) by converting the ratio estimates. OR>1
indicates that the exposure factor is a risk factor for the outcome variable or<
1 indicates that the exposure factor is a protective factor for the outcome
variable and P< 0.05
indicates statistical significance. The IVW method assumes that all the SNPs
included in the analysis can be used as effective IV, which can provide great
help for the analysis. The pleiotropic nature of genetic variation may lead to
the failure of the three hypotheses of IV. The weighted median gives an
accurate estimate based on the assumption that the effective number of IV is 50
% and the causal effect can still be accurately calculated. MR-Egger regression
assumes that all IVs are invalid IV and the estimation accuracy of this method
is relatively low. The weighted model was used to evaluate the robustness of MR
results.
2.4. Sensitivity analysis
In
addition, we will conduct a series of sensitivity analyses, including
heterogeneity and pleiotropy. IVW and MR-Egger regression were used to test
heterogeneity and Cochran Q statistics were calculated to quantify
heterogeneity. When P<0.05,
it represents heterogeneity. If there is heterogeneity, we deal with IVW with
random effects for analysis. Horizontal pleiotropic for our study is very
important because the influence of horizontal pleiotropy may lead to the
instability of effect estimation. The test of pleiotropic level mainly includes
MR-Egger intercept and MR-PRESSO analysis. When P < 0.05, it represents
horizontal pleiotropic. The MR-Egger intercept method estimates the possibility
of horizontal pleiotropy by calculating the intercept term obtained after
linear regression analysis. MR-PRESSO analysis can evaluate the overall
pleiotropicity of the study and screen out abnormal SNPs that may have
horizontal pleiotropicity. We used the software package to set the distribution
number in MR-PRESSO analysis to 5 000.Global test was used to observe whether
there was pleiotropic effect and the robustness of MR analysis results was
evaluated by eliminating SNPs one by one and judging the influence of each SNP
on MR analysis results (Figure 1).
Figure 1: Technical route.
The
summary data of family history of Alzheimer's disease were obtained from the
IEU Open GWAS database and the summary data of cervical malignant tumors were
obtained from the Finnish R9 database. The instrumental variables that met the
requirements were selected. The causal relationship between family history of
Alzheimer 's disease and cervical malignant tumors was determined by MR
analysis, sensitivity analysis, leave-one-out method and Meta analysis.
3. Results
3.1. Selection of instrumental variables
First,
we used the R language to extract instrumental variables from the IEU Open GWAS
database that meet the appeal criteria for family history of Alzheimer 's
disease (including father history, mother history and family history).
Instrumental variables were extracted from the Finnish R9 database data for
cervical malignancies (including cervical adenocarcinoma, cervical squamous
cell carcinoma and cervical cancer) that met the appeal criteria. Because of
the large volume of data, the results of follow-up studies suggest that fathers
with Alzheimer 's disease increase the risk of cervical adenocarcinoma in their
offspring. Therefore, the process of MR analysis is shown below with fathers
with Alzheimer 's disease as the exposure factor and offspring with cervical
adenocarcinoma as the outcome variable. For the choice of instrumental
variables, we included 18 independent SNPs (r 2 10,000) that were significantly
associated with father 's Alzheimer 's disease. When these SNPs were associated
with the summary data of the outcome, we harmonized the exposure and outcome
data (removing unmatchable and palindromic data), which resulted in the
deletion of three SNPs (rs72563085, rs883178, rs11595013). Finally, the SNP of
IVs analyzed as a father with Alzheimer 's disease and cervical adenocarcinoma
is shown in (Table 2). For the instrumental variables used for the final
analysis, all F statistics are greater than 10. It is shown that these are
robust IVs and satisfy the strong correlation hypothesis of MR analysis and the
same method is used to select the instrumental variables of other exposure and
outcome factors.
Table 2: Instrumental
variables for MR studies on the relationship between father's AD and cervical
adenocarcinoma.
|
SNP |
Chr |
EA |
0A |
Beta |
Eaf |
F |
P值 |
|
rs10753570 |
1 |
G |
A |
-0.0536797 |
0.565006 |
22.36424831 |
2.26E-06 |
|
rs114373075 |
1 |
A |
C |
1.13221 |
0.00739156 |
22.24894507 |
2.39E-06 |
|
rs13082929 |
3 |
A |
G |
-0.232869 |
0.227641 |
21.76172568 |
3.09E-06 |
|
rs146652660 |
10 |
T |
C |
0.836881 |
0.00782675 |
21.8733233 |
2.91E-06 |
|
rs184336441 |
7 |
T |
C |
-0.0103829 |
0.021446 |
21.38922554 |
3.75E-06 |
|
rs188423362 |
8 |
C |
T |
-0.0479785 |
0.0776668 |
23.4974 |
1.25E-06 |
|
rs2972558 |
19 |
T |
C |
0.189375 |
0.766033 |
55.46667642 |
9.51E-14 |
|
rs35765215 |
18 |
G |
A |
-0.23809 |
0.137722 |
20.91159245 |
4.81E-06 |
|
rs429358 |
19 |
C |
T |
0.380142 |
0.182736 |
1127.185568 |
1.00E-200 |
|
rs56141511 |
11 |
G |
A |
-1.02904 |
0.0236884 |
21.6872662 |
3.21E-06 |
|
rs58124010 |
17 |
T |
C |
0.333966 |
0.169088 |
20.99074949 |
4.62E-06 |
|
rs6733839 |
2 |
T |
C |
0.127465 |
0.373747 |
31.11999985 |
2.43E-08 |
|
rs673751 |
11 |
C |
A |
-0.176672 |
0.645357 |
29.29215115 |
6.22E-08 |
|
rs72940158 |
2 |
G |
A |
0.11539 |
0.052581 |
21.51055077 |
3.52E-06 |
|
rs878190 |
11 |
G |
A |
0.0220269 |
0.499013 |
21.21379929 |
4.11E-06 |
3.2. Two-sample MR analysis
We
used the statistical power calculation website to calculate the statistical
power of two sample MR studies (https://shiny.cnsgenomics.com/mRnd/) and the
statistical power of MR analysis was 99 %. The data were detected by IVW
method, MR-Egger method, WME method, Simple Mode method and Weighted Mode
method. The results showed that the results of IVW method, MR-Egger method, WME
method, Simple Mode method and Weighted Mode method were consistent. IVW as the
main analysis method, suggesting that fathers with Alzheimer 's disease
increased the risk of cervical adenocarcinoma by 99 % (OR=1.99; 95 % CI:
1.04-3.80, P = 0.037).
Secondary analysis methods included MR-Egger (OR = 2.19; 95 % CI: 0.84-5.73, P = 0.132), weighted median (OR = 2.07;
95 % CI: 1. 12-3. 84, P = 0. 021),
weighted model (OR = 2. 09; 95 % CI: 1. 13-3. 88, P = 0. 035), the OR values were greater than 1 (Figures 2 and
3). IVW suggested that fathers with Alzheimer 's disease were not
associated with uterine squamous cell carcinoma and other cervical malignant
tumors and mothers with Alzheimer 's disease or a family history of Alzheimer
's disease did not increase the risk of cervical malignant tumors (Figure 2).
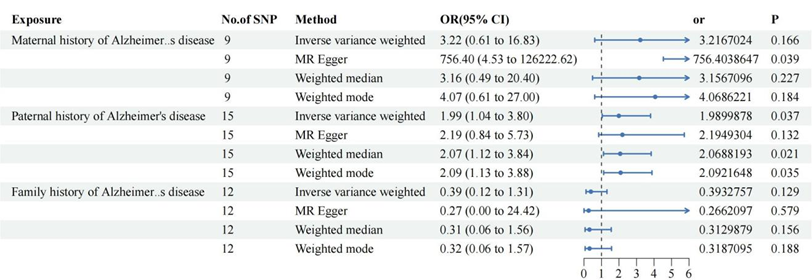
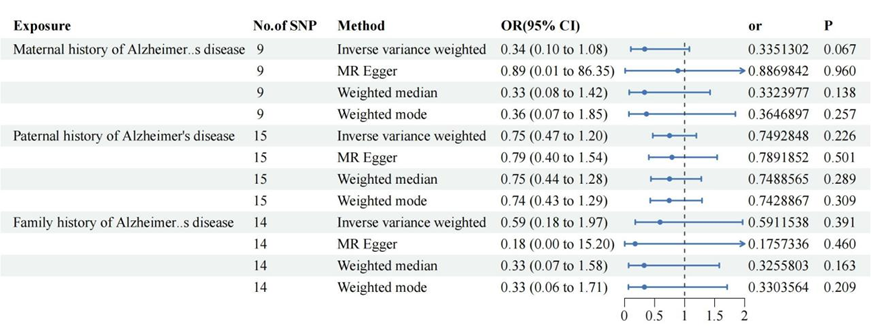
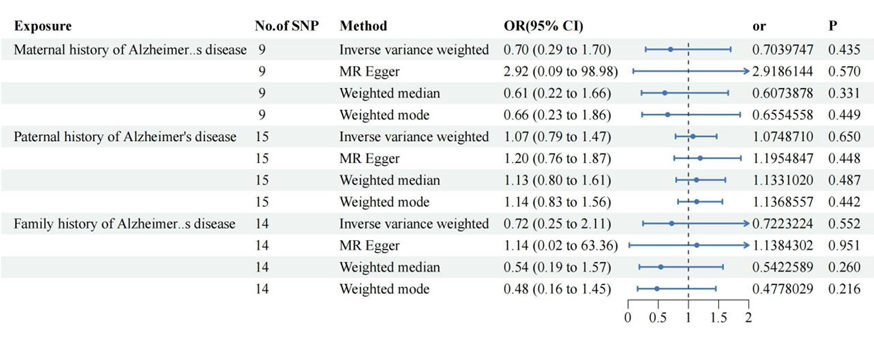
Figure 2: Alzheimer 's family history of cervical cancer risk.
IVW
suggested that fathers with Alzheimer 's disease were not associated with
uterine squamous cell carcinoma and other cervical malignant tumors and mothers
with Alzheimer 's disease or a family history of Alzheimer 's disease did not
increase the risk of cervical malignant tumors.
Figure 3: Scatter plot of Alzheimer 's family history and
risk of cervical cancer.
Scatter
plot to visualize the casual relationship between Alzheimer ‘s family history
and cervical malignant tumors. Figures A to I respectively showed the effect of
ebi-a-GCST005920, ebi-a-GCST005923 and ebi-a-GCST005921 on cervical squamous
cell carcinoma, cervical adenocarcinoma and other cervical malignant tumors.
Figures A,B and C suggested that Alzheimer 's family history was a protective
factor for cervical squamous cell carcinoma. Other figures suggested it was a
risk factor for cervical adenocarcinoma and other cervical malignant tumors.
3.3. Sensitivity analysis and visualization
MR-Egger
regression (Cochran Q = 18.47, P = 0.14) and IVW (Cochran Q = 18.58, P = 0.18)
of fathers with Alzheimer 's disease and cervical adenocarcinoma indicated that
there was no heterogeneity in the study, as shown in Table 3. The funnel plot
was a visualization of heterogeneity (Figure 4). MR-Egger intercept did
not show horizontal pleiotropic (irritability: Egger intercept: − 0.02, P = 0.78).
See (Figure 5). No abnormal values were found by MR-PRESSO test and the
Global test was 0.36, indicating that there was no pleiotropic effect, as shown
in Table 3. We used the leave-one-out method to remove SNPs one by one to
determine whether the causal association was caused by a single IV. The final
results showed that the results of the two-sample MR analysis were robust, as
shown in (Figure 6).
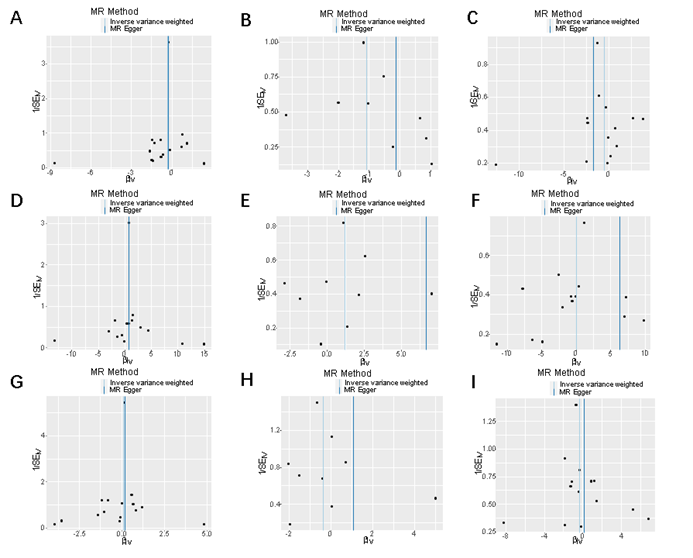
Figure 4: Funnel plot of Alzheimer 's family history and risk
of cervical cancer.
Funnel
plot: Visualization of overall
heterogeneity testing between Alzheimer 's family history and cervical
malignant tumors. If there is symmetry in the funnel plot, it means there is no
heterogeneity.
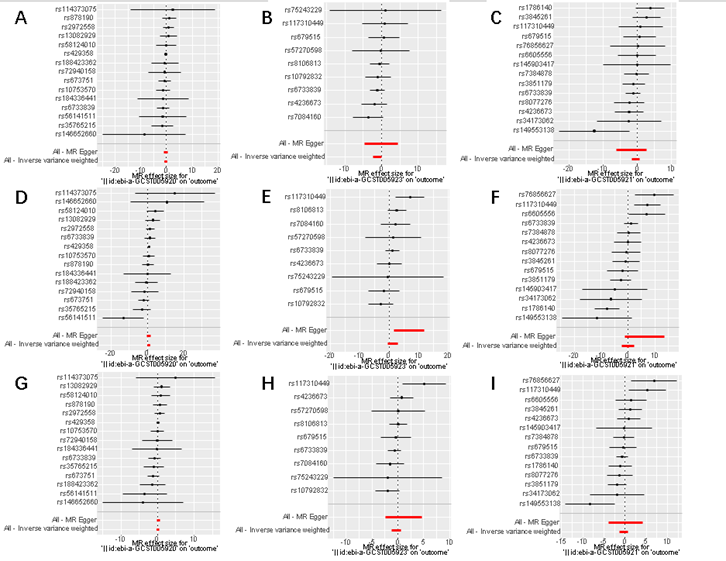
Figure 5: A forest map of the risk of Alzheimer 's family
history and cervical cancer.
Forest
plot representing the casual effects of Alzheimer 's family history and
cervical malignant tumors. Figures A to I respectively showed the effect of
ebi-a-GCST005920, ebi-a-GCST005923 and ebi-a-GCST005921 on cervical squamous
cell carcinoma, cervical adenocarcinoma and other cervical malignant tumors.
Figures D and G reflected that the father with Alzheimer 's disease will
increase the risk of cervical adenocarcinoma and other cervical malignant
tumors. Figures A and B showed that the father with Alzheimer 's disease and
the mother with Alzheimer 's disease will increase the risk of cervical
squamous cell carcinoma.
Figure 6: Leave-one-out analysis the risk of Alzheimer 's
family history and cervical cancer.
Leave-one-out
analysis: Eliminating SNPs one by one and judging the influence of each SNP on
MR analysis results to affirming the robustness of the results. Only Figure D
's overall results of all SNPs do not cross/contain 0, indicating the stability
of the results.
3.4.
Heterogeneity and pleiotropic analysis of other MR studies
Heterogeneity
and pleiotropic analysis results of other MR studies are shown in (Table 3).
The results suggest that there may be heterogeneity and pleiotropicity in the
study of Alzheimer 's family history and cervical malignant tumors and there is
no pleiotropicity and heterogeneity in other studies.
Table 3:
Heterogeneity and multieffectiveness test.
|
Expose |
Outcome |
Heterogeneity test |
Multipleiotropy test | |||
|
Method |
Q |
P值 |
Method |
P | ||
|
Father has Alzheimer's disease |
Cervical adenocarcinoma |
MR-Egger |
18.47 |
0.14 |
MR-Egger |
0.78 |
|
IVW |
18.58 |
0.18 |
PRESSSO |
0.36 | ||
|
Mother has Alzheimer's disease. |
MR-Egger |
6.75 |
0.46 |
MR-Egger |
0.07 | |
|
IVW |
11.47 |
0.18 |
PRESSSO |
0.22 | ||
|
Family history of Alzheimer's |
MR-Egger |
9.73 |
0.46 |
MR-Egger |
0.86 | |
|
IVW |
9.76 |
0.55 |
PRESSSO |
0.62 | ||
|
Father has Alzheimer's disease |
cervical squamous cell carcinoma |
MR-Egger |
5.74 |
0.96 |
MR-Egger |
0.84 |
|
IVW |
5.78 |
0.97 |
PRESSSO |
0.98 | ||
|
Mother has Alzheimer's disease. |
MR-Egger |
2.93 |
0.89 |
MR-Egger |
0.67 | |
|
IVW |
3.12 |
0.93 |
PRESSSO |
0.94 | ||
|
Family history of Alzheimer's |
MR-Egger |
14.09 |
0.30 |
MR-Egger |
0.59 | |
|
IVW |
14.45 |
0.34 |
PRESSSO |
0.39 | ||
|
Father has Alzheimer's disease |
Cervical malignancy |
MR-Egger |
9.30 |
0.75 |
MR-Egger |
0.53 |
|
IVW |
9.72 |
0.78 |
PRESSSO |
0.82 | ||
|
Mother has Alzheimer's disease. |
MR-Egger |
9.34 |
0.23 |
MR-Egger |
0.44 | |
|
IVW |
10.23 |
0.25 |
PRESSSO |
0.33 | ||
|
Family history of Alzheimer's |
MR-Egger |
25.84 |
0.01 |
MR-Egger |
0.82 | |
|
IVW |
25.95 |
0.02 |
PRESSSO |
0.03 | ||
4. Discussion
Our
MR analysis has revealed a potential causal relationship between genetic
variants associated with Alzheimer's disease in fathers and an increased risk
of cervical cancer. The use of various MR methods, including IVW, MR-Egger,
weighted median and mode-based estimates, has provided consistent evidence
supporting this association7.Furthermore,
our sensitivity analyses strengthen the argument for a causal link by
demonstrating robustness against heterogeneity and pleiotropy. The absence of
significant heterogeneity or horizontal pleiotropy-as evidenced by Cochran Q
tests and MR-Egger intercepts-suggests that the observed association is not
confounded by these factors8.
Additionally, MR-PRESSO did not detect outliers, indicating that our results
are unlikely to be driven by individual SNPs with disproportionate effects.
The
identification of genetic variations through MR analysis has provided novel
insights into the pathophysiology of familial Alzheimer's disease and cervical
cancer, underscoring the importance of genetic predisposition in their
etiology. APOE, which has been extensively documented as a major genetic risk
factor for Alzheimer's disease9.
The ε4 allele of APOE is associated with an increased risk of developing
Alzheimer's disease, while the ε2 allele appears to have a protective effect10. Our findings suggest that individuals
carrying certain APOE variants may also have an elevated risk of developing
cervical adenocarcinoma, indicating a possible shared genetic pathway
influencing both conditions. This observation could pave the way for further
investigation into common molecular mechanisms underlying neurodegenerative and
oncogenic processes.
The
genetic predisposition to Alzheimer's disease may share common pathways with
the development of certain cancers, which could be mediated by immune system
dysregulation11.The immune system
plays a critical role in both neurodegeneration and tumor surveillance. Chronic
inflammation, for instance, is a hallmark of Alzheimer's disease and has been
implicated in the pathogenesis of various cancers, including cervical cancer12. The interplay between inflammatory
processes and immune response modulation could provide insights into shared
genetic susceptibilities that influence both conditions. Further research into
the immunological aspects underlying this association could yield valuable
information on the mechanisms at play. For example, exploring how specific
alleles might influence immune cell function or cytokine production could
illuminate pathways amenable to therapeutic intervention13. Ultimately, understanding these
connections could pave the way for novel strategies in preventing or treating
both Alzheimer's disease and cervical cancer through immune modulation.
Reflecting
on this study's limitations, it is crucial to recognize that our conclusions
rely on public database and statistical approaches without wet-lab validation.
The sample sizes for some genetic variants might have been insufficient for
detecting subtle associations or ensuring conclusion robustness. Moreover, we
did not perform clinical validation analysis; thus, cautious interpretation is
warranted before clinical application. Additionally, employing multiple
datasets may introduce batch effects that could confound our findings.
In conclusion, this investigation applied MR
methods to explore putative causal links between particular genetic variations
and both Alzheimer's disease familial history as well as cervical malignancies.
Our principal outcomes indicate a potential causative association with specific
genes. Sensitivity analyses were performed addressing heterogeneity and
pleiotropy concerns which bolstered result credibility despite limitations such
as lack of experimental confirmation and possible dataset-related batch effects.
These insights pave the way for future research endeavors which may ultimately
lead towards enhanced diagnostics or treatments pending further clinical
corroboration.
5. Declarations
5.1. Abbreviations
MR: Mendelian Randomization
IVW: Inverse variance weighting
AD: Alzheimer's disease
SAD: sporadic AD
FAD: familial AD
LD: Linkage disequilibrium
OR: odds ratio
5.2. Ethics
approval and consent to participate
Ethical review was not required for this study
as it involves the use of publicly available, anonymized data from a database.
No new data were collected and no new ethical approval was required.
5.3. Consent
for publication
All authors have agreed to the publication of
this article.
5.4. Availability
of data and materials
The datasets can be freely obtained from the
IEU Open GWAS Database (https://gwas.mrcieu.ac.uk/) and the Finnish R9 Database
(https://r9.finngen.fi/).
5.5. Competing
interests
No, I declare that the authors have no
competing interests as defined by BMC or other interests that might be
perceived to influence the results and/or discussion reported in this paper.
5.6. Funding
statement
This study was funded by Medical Key Strategic
Project of Wuxi Health Commission.
5.7. Authors'
contributions
Li was responsible for data collection and
organization, as well as paper writing and revision. Xiong was in charge of
data analysis and processing and paper writing. Gu took the lead in research
design and planning and paper writing and supplementation. Zhao was responsible
for research supervision and coordination and management of research resources.
Yue was in charge of research technical support and paper writing and
polishing. Jiang was responsible for fund support and paper improvement. Each
author played their part while collaborating with each other in the research
process, jointly completing this research project. Every author made
significant contributions to the final outcome of the paper, ensuring the
scientific nature and rigor of the research.
5.8. Acknowledgements
The authors would like to thank the researchers
and study participants for their contributions.
6.
References
- Long
S, Benoist C, Weidner W. World Alzheimer Report 2023: Reducing dementia risk:
never too early, never too late, 2023.
- Jia
L, Fu Y, Shen L, et al. PSEN1, PSEN2 and APP mutations in 404 Chinese pedigrees
with familial Alzheimer's disease. Alzheimer's & dementia: The journal of
the Alzheimer's Association, 2020;16: 178-191.
- Bird
TD. Genetic aspects of Alzheimer disease. Genetics in Medicine, 2008;10:
231-239.
- Arbyn
M, Weiderpass E, Bruni L, et al. Estimates of incidence and mortality of
cervical cancer in 2018: a worldwide analysis. The Lancet. Global health, 2020;8:
191-203.
- Smith
DG, Hemani G. Mendelian randomization: genetic anchors for causal inference in
epidemiological studies. Human molecular genetics, 2014;23: 89-98.
- Holmes
MV, Asselbergs FW, Palmer TM, et al. Mendelian randomization of blood lipids
for coronary heart disease. European heart journal, 2015;36: 539-550.
- Burgess
S, Thompson SG. Interpreting findings from Mendelian randomization using the
MR-Egger method. European journal of epidemiology, 2017;32: 377-389.
- Bowden
J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments:
effect estimation and bias detection through Egger regression. International
journal of epidemiology, 2015;44: 512-525.
- Corder
EH, Saunders AM, Strittmatter WJ, et al, Gene dose of apolipoprotein E type 4
allele and the risk of Alzheimer's disease in late onset families. Science (New
York, N.Y.), 1993;261: 921-923.
- Liu
CC, Liu CC, Kanekiyo T, et al. Apolipoprotein E and Alzheimer disease: risk,
mechanisms and therapy. Nature reviews. Neurology, 2013;9: 106-118.
- Perry
VH, Cunningham C, Holmes C. Systemic infections and inflammation affect chronic
neurodegeneration. Nature reviews. Immunology, 2007;7: 161-167.
- Coussens LM, Werb Z. Inflammation and cancer. Nature, 2002;420: 860-867.
- Brodin P, Davis MM. Human immune system variation. Nature reviews. Immunology, 2017;17: 21-29.