Unveiling the Hidden Horn: A Rare Case of Rudimentary Uterine Horn with Endometriotic Diseases
Abstract
Background: A rudimentary uterine horn is a rare
congenital anomaly that results from the abnormal development of one Müllerian
duct, while the other duct remains underdeveloped. This condition is often
associated with a unicornuate uterus and may lead to severe complications, such
as endometriosis, due to retrograde menstruation. The diagnosis of this anomaly
is challenging due to non-specific symptoms and high misdiagnosis rates.
Case summary: We present the case of a 45-year-old woman
who experienced progressive dysmenorrhea for 10 years and prolonged menstrual
periods for 3 months. Imaging studies, including ultrasound and MRI, suggested
a double uterus with multiple fibroids and potential hydrosalpinx. During
laparoscopic surgery, the patient was found to have a rudimentary uterine horn
with a small connection to the unicornuate uterus, right ovarian cysts, severe
pelvic adhesions, and endometriosis. The surgical procedures included the
removal of the rudimentary horn, right ovarian cystectomy, and excision of
fibroids from both the left and right uteri. Postoperative pathology confirmed
leiomyoma and adenomyosis. The patient is currently under follow-up with normal
ultrasound and laboratory findings.
Conclusion: This case highlights the importance of early
recognition and accurate diagnosis of rudimentary uterine horn, which is often
misdiagnosed due to atypical presentations. A combination of imaging
techniques, clinical evaluation, and surgical exploration is critical for
proper management. Early intervention can help alleviate symptoms, prevent
complications like endometriosis, and improve patients' quality of life.
Keywords: Rudimentary uterine horn; Unicornuate uterus;
Endometriosis; Congenital uterine anomaly
1. Introduction
The
rudimentary uterine horn is a rare form of congenital uterine anomaly,
resulting from the development of one Müllerian duct and the developmental
deficiency of the contralateral Müllerian duct. It is characterized by the
presence of only the uterine corpus and fallopian tube, with an absence of the
cervix and vaginal structure on the affected side1.The
unicornuate uterus accounts for approximately 5% of Müllerian duct anomalies, and
is often associated with a rudimentary uterine horn. Clinically, it can be
divided into three types1. Manifestations
of a rudimentary uterine horn is diverse and subtle2,with
some individuals remaining asymptomatic and the condition going unnoticed
indefinitely. Additionally, the diagnosis of this condition is challenging and
prone to misdiagnosis, which can lead to a range of complications. Non-rudimentary
uterine horn pregnancy (RHP) is often misdiagnosed as endometriosis, while
rudimentary uterine horn pregnancy is most likely to be mistaken for tubal
pregnancies. A pregnancy in a rudimentary uterine horn can lead to
complications such as uterine rupture3,posing
a risk to the patient's life. We report a case of a rudimentary uterine horn
combined with a unicorous uterus and associated with endometriosis (EMT) to
enhance awareness and understanding of this condition and to reduce the
incidence of misdiagnosis and missed diagnosis.
2.
Case presentation
2.1. Clinical and pathological
data’s
Patient, female, 45 years old, presented to
our hospital on June 23, 2024, due to "progressive dysmenorrhea for 10
years and prolonged menstrual period for 3 months." The outpatient
ultrasound suggested the possibility of a rudimentary uterine horn, a uterine
fibroid on the left side, and adenomyosis on the right side (Figure 1), leading to admission to the hospital. The
patient has regular menstrual cycles, with a 30-day cycle, moderate flow, and
severe dysmenorrhea. Last menstruation: 2024-07-05. Previous menstruation:
2024-05-24. Past medical history: diabetes and hypertension for 9 years.
Obstetric history: 1-0-0-1, cesarean section in 2003.Preoperative expert
ultrasound (2024-07-15): Didelphic uterus (single cervix, single cervical
canal) with multiple fibroids in the left uterus, the largest measuring
approximately 454437mm, one of which is submucosal, adenomyosis with fibroids
in the right uterus, a small amount of fluid in the right uterine cavity, and a
poorly echoic area on the right measuring 74×57×32mm, suggesting the possibility
of hydrosalpinx (Figure 2). On July 16, 2024, the patient's CA-125
level was measured at 279.40 U/ml, and CA 199 was 102.4 U/m. A pelvic MRI
revealed no significant abnormalities in the liver, gallbladder, spleen,
pancreas, and left kidney (Figure 3). A urinary system ultrasound
indicated a right kidney absence status (Figure 4).
On
July 17, 2024, the patient underwent a combined laparoscopic and hysteroscopic
surgery. Intraoperative findings included: the left uterus was mid-positioned,
enlarged to the size of a 2-month pregnancy, with an irregular shape. There
were two myoma-like tissue protrusions on the anterior wall of the uterus,
approximately 4 cm and 3 cm in diameter, mostly protruding outside the serosa,
and hard in texture. There was one myoma-like tissue protrusion on the
posterior wall of the uterus, about 5 cm in diameter, partially protruding
outside the serosa, and hard in texture. The left ovary and fallopian tube
appeared normal; the right uterus was mid-positioned, enlarged to the size of a
2-month pregnancy, with a full shape, and closely adhered to the left uterus.
The right fallopian tube was thickened and tortuous, with the ampulla visible.
The right ovary had a cystic mass about 6 cm in diameter. The right ovary,
fallopian tube, and parts of the intestines, mesentery, and pelvic wall were
closely adhered and encapsulated. Scattered brown endometrial implantation
lesions were seen on the intestinal wall, about the size of rice grains, and no
significant fluid was observed in the pelvic cavity. The adhesions throughout
the pelvic cavity were released, the mesentery and isthmus of the right
fallopian tube were transected, and the right fallopian tube was removed; the
viscous chocolate-like fluid in the cystic cavity of the right ovarian tumor
was aspirated, the ovarian rupture was opened along the long axis of the ovary,
and the cyst wall was bluntly peeled off and completely removed. The raw
surface was rinsed with physiological saline and hemostasis was achieved with
bipolar electrocoagulation. The endometrial implantation lesions on the
intestinal wall were excised; the adhesions between the left and right uteri
were separated, the right uterine artery was clamped, and the right uterus was
removed. It was observed that the two uteri were connected by a small hole with
a diameter of about 2mm, and the raw surface was sutured; the anterior wall of
the uterus was cut horizontally with a unipolar electro hook to the pseudo
capsule of the myoma, and three uterine fibroids on the left side were peeled
off, the largest being about 4×4×3cm, and the raw surface was continuously
sutured with a 1/0 absorbable suture for the muscle layer and the myometrium.
The right uterine body was rotated and cut with a uterine rotator, and several
myoma-like tissues were observed on the cross-section of the uterus, and three
uterine fibroids were similarly rotated and cut. The right ovarian cyst, right
fallopian tube, right uterus, three uterine fibroids on the left side, and the
intestinal wall endometriosis lesions were all sent for pathological
examination. Hysteroscopic surgery was performed to remove the protruding
tissue in the uterine cavity, with a total size of about 4.0×4.0×2.0cm, and a
curettage was performed, and a small amount of endometrial tissue was scraped
out. The uterine cavity excision and the scraped material were sent for routine
pathological examination. Intraoperative findings are illustrated in (Figure
5). Postoperative hysteroscopic re-examination showed no obvious
space-occupying lesions or active bleeding. Intraoperative rapid pathology
showed: (right ovarian tumor) benign cyst; (left uterine fibroids) leiomyoma.
(Entire right uterus) ① (uterus) Leiomyoma. ② (uterus)
Adenomyosis. The surgery went smoothly, with 2000ml of fluid replenished during
the surgery, 500ml of blood loss, and 400ml of urine output.
Postoperative pathology indicates:
endometriotic lesions on the intestinal wall. Right ovarian tumor: endometrial
cyst of the ovary. Right uterus: ① Leiomyoma ②
Adenomyosis. Right fallopian tube: hydrosalpinx with chronic inflammation. Left
uterine fibroid: leiomyoma. Endometrial scrapings and electro-resection
specimens from the left uterus: submucous leiomyoma.
Postoperative follow-up: On August
19, 2024, the patient came to our hospital's outpatient department for
follow-up: CRP (panel), blood cell [five-category] analysis showed: basophil
percentage 1.1%, eosinophil count 0.66×10^9/L, basophil count 0.10×10^9/L, red
cell distribution width CV 15.6, platelet count 601×10^9/L, plateletcrit 0.42%.
Ultrasound examination: The uterus is anteverted, measuring 50×40×32mm, with
regular shape and clear contour. The myometrium of the uterus is uneven. The
endometrial thickness is 6mm, uneven (Figure
6).
Outpatient treatment with dienogest was prescribed. Currently, regular
follow-up is ongoing, and follow-up ultrasound, CRP, and blood cell tests are
all normal.
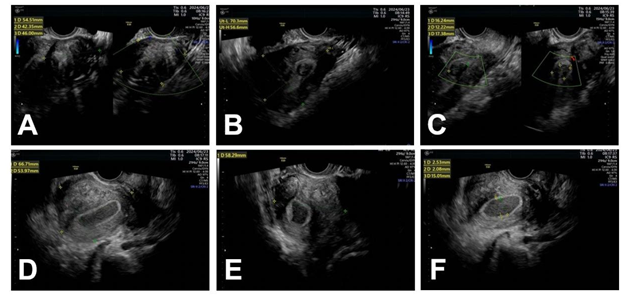
Figure 2: Expert ultrasound. The left image is the left uterus, and the right
image is the right uterus.
Figure 3: Pelvic MRI. No significant abnormalities observed in the liver, gallbladder, spleen, pancreas, and left kidney.
Figure 4: Urinary system ultrasound. The left image shows absence of the right kidney, and the right image shows a solitary left kidney.
Figure 5: Intraoperative Findings.
A: Right
ovarian chocolate cyst;
B: A 2mm
small opening connecting the rudimentary uterus to the unicornuate uterus;
C: Uterine
fibroid on one side of the unicornuate uterus;
D:
Variations in branching and course of blood vessels on the rudimentary uterus
side; E: Comparison of pelvic floor anatomy between the unicornuate uterus and
the rudimentary uterus reveals vascular variations on the rudimentary uterus
side.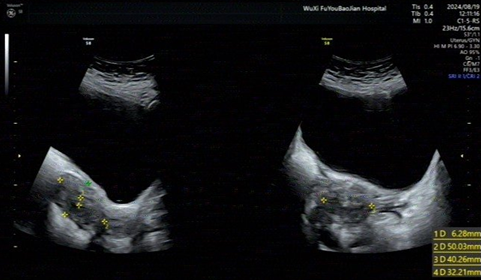
Figure 6: Follow-up ultrasound.
2.2.
Diagnosis, differential diagnosis, and analysis of the causes of misdiagnosis
of a rudimentary uterine horn
The incidence of rudimentary uterine horn is
low, and its clinical manifestations are atypical, making misdiagnosis and
missed diagnosis likely. When patients present with the following clinical
manifestations, the possibility of a rudimentary uterine horn should be
considered: (1) Dysmenorrhea: A major clinical manifestation of type II
rudimentary uterine horn. When secondary dysmenorrhea occurs, one should be
alert to uterine abnormalities. Severe dysmenorrhea may be due to retrograde
menstruation in the unicornuate uterus, which does not communicate with the
normal uterine cavity, leading to endometriosis and blood stasis4. History of recurrent miscarriages and
infertility: A rudimentary uterine horn affects the patient's fertility, as the
uterine muscle wall is poorly developed and cannot withstand the growth and
development of the fetus, making it prone to severe pregnancy complications
such as uterine rupture5. (3) Abdominal
pain: Commonly seen in the mid to late stages of pregnancy with a rudimentary uterine
horn, it may occur due to uterine rupture or torsion6. (4) Pelvic mass: Gynecological
examination may reveal a mass closely related to the uterus, which can be
mistakenly diagnosed as an ovarian tumor. (5) Signs of urinary system
abnormalities: Further confirmation of the presence of urinary system deformities
can be obtained through imaging examinations. In addition to clinical
manifestations, auxiliary tests can also be combined to assist in the diagnosis
of rudimentary uterine horn. These mainly include two-dimensional ultrasound,
three-dimensional ultrasound, MRI, and hysterosalpingography (HSG).
Two-dimensional ultrasound is cost-effective but cannot accurately observe the
coronal section of the uterus, increase the rate of misdiagnosis and missed
diagnosis of the disease. In contrast, three-dimensional ultrasound has higher
sensitivity and is of great value in disease diagnosis. Studies have shown that
combining three-dimensional ultrasound with routine two-dimensional ultrasound
helps to improve the accuracy of disease diagnosis7.Additionally,
MRI is helpful for classifying rudimentary uterine horn, and HSG can be used to
assist in the diagnosis of uterine anomalies.
The misdiagnosis rate of rudimentary uterine
horn is quite high, and it needs to be differentiated from the following
conditions: (1) Ovarian tumors: Both can manifest as masses in the adnexal
region, connected to the uterus by a pedicle. (2) Endometriosis: A rudimentary
uterine horn may lead to endometriosis due to retrograde menstruation, but
endometriosis itself is not a rudimentary uterine horn, and it needs to be
distinguished through clinical manifestations and imaging examinations. (3)
Tubal pregnancy: Both rudimentary uterine horn pregnancy and tubal pregnancy
can present with a history of amenorrhea followed by rupture and abdominal
pain, and the interstitial part of the tubal pregnancy may also protrude,
making it difficult to differentiate in the early stages of pregnancy. In
summary, the main reasons for the misdiagnosis and missed diagnosis of
rudimentary uterine horn are the lack of understanding of the condition by
physicians, insufficient history taking and examination, leading to incomplete
clinical data, and over-reliance on imaging examinations.
3. Discussion
According to the ASRM classification,
unicornuate uterus is categorized as Type II. Clinically, this malformation
group is further divided into four subtypes: Type a refers to a rudimentary
uterine horn that communicates with the unicornuate uterine cavity; Type b,
where the rudimentary horn has a cavity but does not communicate with the
unicornuate uterus; Type c, a rudimentary horn without a cavity that is
connected to the unicornuate uterus only by a fibrous band; and Typed, the
absence of a rudimentary horn8.
This anomaly results from abnormal development of one side of the Müllerian
duct during embryogenesis and is frequently associated with urinary tract
malformations. Statistics indicate that unicornuate uterus is accompanied by
renal hypoplasia or ectopic kidney in 15% of cases, with 40% of this exhibiting
congenital renal agenesis on the side of the rudimentary horn8,9. If the rudimentary uterine horn
contains functioning endometrium, it undergoes cyclic shedding during
menstruation, leading to retrograde menstrual flow and accumulation within the uterine
cavity. This condition increases the likelihood of gynecological issues, such
as dysmenorrhea and chronic pelvic pain, and may gradually develop into
endometriosis and infertility10.
As a rare congenital uterine anomaly, the rudimentary uterine horn is often
difficult to diagnose and treat early in clinical practice due to nonspecific
early symptoms and limited accuracy of imaging studies11.
We present a rare case of a Type IIa
rudimentary uterine horn malformation associated with ipsilateral renal
agenesis and severe endometriotic disease. Preoperative transvaginal ultrasound
suggested the possibility of a rudimentary uterine horn, while MRI indicated a
double uterus. Although research shows a good correlation between MRI and
surgical findings in diagnosing congenital uterine malformations12, in this case, the small communication
between the rudimentary horn and the unicornuate uterus may have led to a
misdiagnosis on MRI. The patient’s right kidney agenesis was a key diagnostic
clue, highlighting the need for clinicians to assess patients based on clinical
symptoms, comprehensive imaging studies, and cutting-edge medical knowledge to
devise optimal treatment plans.
The primary goals in treating a rudimentary
uterine horn are to alleviate symptoms, prevent complications, and preserve
reproductive health. In this case, the patient had experienced progressively
worsening dysmenorrhea over 10 years, severely affecting her quality of life,
making laparoscopic resection of the rudimentary horn an appropriate choice13.
Endometriotic disease encompasses both
endometriosis and adenomyosis, conditions in which active endometrial tissue is
ectopically located. Adenomyosis refers to the invasion of endometrial tissue
into the myometrium, forming endometriotic lesions, while endometriosis refers
to the presence of endometrial tissue outside the uterine cavity, primarily in
pelvic organs and the peritoneum. The pathogenesis of endometriosis remains
controversial and includes theories such as retrograde menstruation, autoimmune
response, metaplasia of coelomic epithelium, and lymphovascular spread. The
retrograde menstruation theory suggests that fragments of endometrial tissue
containing active glands and stroma reflux through the fallopian tubes into the
peritoneal cavity, where they adhere and invade the underlying mesothelium14. This theory aligns with epidemiological
evidence linking EMs to factors such as increased menstrual bleeding, shorter
cycle length, higher frequency of menstruation, and an increased incidence of
reproductive tract obstructions15.
Common complications of congenital Müllerian duct malformations include
hematosalpinx, endometriosis, chronic pelvic pain, and adhesions, all secondary
to retrograde menstruation16. In
cases where the rudimentary uterine horn does not communicate with the
unicornuate uterus, the inability to expel menstrual blood often leads to
endometriotic disease. In this case, the pathogenesis appears to align with the
retrograde menstruation theory, as intraoperatively, a 2 mm communication
between the rudimentary horn and the unicornuate uterus was identified. The
outflow tract had been obstructed for years, causing progressively worsening
dysmenorrhea. Fragments of menstrual blood containing active endometrial cells
likely refluxed through the fallopian tubes into the ovaries and pelvic cavity,
resulting in adenomyosis in the rudimentary horn and endometriotic lesions in
the bowel wall and right ovary. Treatment of endometriotic disease requires a
tailored approach based on the patient's age, symptoms, and reproductive
desires, along with long-term postoperative management.
Once the diagnosis is established, surgical
management becomes critical. In this case, we first addressed the endometriotic
lesions in the bowel wall and right ovary to prevent the spread of chocolate
cyst fluid in the pelvic cavity. Next, we proceeded with the resection of the
rudimentary uterine horn. Given that rudimentary horns are often associated
with urinary tract anomalies, careful attention must be paid to the ipsilateral
ureter and major uterine vessels during surgery. A study indicated that in non-communicating
rudimentary horns, the ipsilateral ureter is located higher than usual because
it lies close to the vascular connection of the rudimentary horn. Therefore,
when transecting the round ligament and entering the broad ligament and
retroperitoneal space, the ureter must be identified. Additionally, a firmly
attached rudimentary horn may receive its blood supply not only from the
ipsilateral uterine artery but also from myometrial arcuate arteries
originating from the contralateral uterine artery, requiring meticulous
hemostasis during dissection17.
Although this case involved a communicating rudimentary horn, the pelvic
anatomical variations remained relevant. Preoperative ultrasound indicated the
patient had a solitary left kidney and right renal agenesis, obviating the need
for identification of the right ureter during resection. However, we carefully
dissected the adhesions and clearly delineated the vascular anatomy, noting
that the internal and external iliac arteries ran parallel. Intraoperatively,
we observed that the vascular branches supplying the rudimentary horn deviated
significantly from normal uterine vasculature. After resecting the rudimentary
uterine horn, we confirmed that both sacrouterine ligaments on the left side
were intact, and the uterine arteries followed a normal course. However, the
pelvic vasculature and nerve structures on the side of the rudimentary horn
exhibited notable variations. Finally, we used laparoscopy and hysteroscopy to
address the uterine fibroids in the unicornuate uterus. A transverse incision
was made in the anterior uterine wall through the seromuscular layer to reach
the pseudocapsule of a 4x4x3 cm fibroid, which was enucleated. Similarly, three
fibroids were removed laparoscopically, followed by hysteroscopic resection of
a 4x4x2 cm submucosal fibroid.
4. Conclusion
This case report presents a rare congenital
uterine anomaly involving a rudimentary uterine horn, unicornuate uterus, and
endometriosis. The patient underwent laparoscopic surgery to remove the
rudimentary horn, ovarian cysts, and multiple uterine fibroids, with
postoperative pathology confirming benign leiomyoma and adenomyosis. The case
underscores the challenges in diagnosing rudimentary uterine horns due to
non-specific symptoms and emphasizes the importance of combining clinical
assessment with imaging for accurate diagnosis and treatment.
5. References
- Chinese experts' consensus on the nomenclature and
definition revision of female genital malformation. Zhonghua fu chan ke za zhi,
2022;57: 575-580.
- Sánchez-Ferrer ML, Prieto-Sanchez MT, Sánchez Del Campo F.
Variations in clinical presentation of unicornuate uterus with
non-communicating rudimentary horn (class IIB of the American Fertility
Society classification). Taiwanese journal of obstetrics & gynecology, 2018;57:
110-114.
- Li X, Peng P, Liu X, et al. The pregnancy outcomes of
patients with rudimentary uterine horn: A 30-year experience. PLoS One, 2019;14:
0210788.
- Cruciat G, Staicu A, Florian A, et al. Spontaneous Fistula
and Abdominal Wall Endometriosis Due to Occult Existence of Unicornuate Right
Uterus with Rudimentary Non-Communicating Functioning Left Horn. Diagnostics,
2024;14.
- Shin SY, Kwon H, Kim HC, et al. Successful pregnancy outcome
via in-vitro fertilization and laparoscopic resection of non-communicating
rudimentary horn pregnancy containing early pregnancy: a case report. BMC
pregnancy and childbirth, 2024;24: 115.
- Abbasi Z, Das S, Thapa U, et al. Ruptured Ectopic Pregnancy
in an Accessory Horn of Uterus: A Case Report. Cureus, 2019;11: 6436.
- McGuinness B, Llarena N, Falcone
T, et al. Uncommon Surgical Emergencies in the Adult Gynecologic Patient: Two
Cases of Missed Diagnosis of Outflow Tract Obstruction from Congenital Uterine
Anomalies. Case reports in obstetrics and gynecology, 2022: 3179656.
- Makroum AAE, Abdelrazik MM, Hassan MSE. Minilaparotomy for
Excision of a Functioning Noncommunicating Rudimentary Horn and Endometrioma in
a Patient with Solitary Kidney: A case report. Gynecology and minimally
invasive therapy, 2020;9: 91-94.
- Abboud K, Giannini A, D'oria O, et al. Laparoscopic
Management of Rudimentary Uterine Horns in Patients with Unicornuate Uterus: A
Systematic Review. Gynecologic and obstetric investigation, 2023;88: 1-10.
- Khati NJ, Frazier AA, Brindle KA. The unicornuate uterus and
its variants: clinical presentation, imaging findings, and associated
complications. Journal of ultrasound in medicine: official journal of the
American Institute of Ultrasound in Medicine, 2012;31: 319-331.
- Yin SF, Chai JG, Feng RL, et al. Case report: Rudimentary
uterine horn with ovarian endometriosis manifested as pelvic ectopic kidney.
Frontiers in medicine, 2023;10: 1182355.
- Minto CL, Hollings N, Hall-Craggs M, et al. Magnetic
resonance imaging in the assessment of complex Müllerian anomalies. BJOG: an International
Journal of obstetrics and gynaecology, 2001;108: 791-797.
- Begum J, Maharana N, Behera SS, et al. Laparoscopic Approach
Towards Non-Communicating Functional Rudimentary Uterine Horn: A Report of Two
Cases. Cureus, 2020;12: 11419.
- Sampson JA. Metastatic or Embolic Endometriosis, due to the
Menstrual Dissemination of Endometrial Tissue into the Venous Circulation. Am J
Pathol, 1927;3: 93-110.
- Missmer SA, Hankinson SE, Spiegelman D, et al. Reproductive
history and endometriosis among premenopausal women. Obstetrics and gynecology,
2004;104: 965-974.
- Liu YH, Jain S,
Lee CL, et al. Incidence of mullerian defects in fertile and infertile women.
The Journal of the American Association of Gynecologic Laparoscopists, 2000;7:
435-436.
- Fedele L, Bianchi S, Zanconato G, et al. Laparoscopic removal of the cavitated noncommunicating rudimentary uterine horn: surgical aspects in 10 cases. Fertil Steril, 2005;83: 432-436.