A Case Study of Intracranial Hypertension due to Neuroschistosomiasis
Abstract
Introduction:
Neuroschistosomiasis is a rare and severe complication of schistosomiasis. It
affects the Central Nervous System. Its prevalence in areas with proximity to
freshwater bodies and poor sanitation is attributable to the aquatic life cycle
and fecal-oral mode of transmission of the parasite. It presents a multitude of
challenges in regards to diagnosis and treatment.
Methods:
The case study reports of a 34 years old female who was misdiagnosed to have Benign
Intracranial Hypertension and presented with severe unrelenting headache and
visual disturbance. Upon examination, a fundoscopy revealing severe
papilledema. Her other blood tests were unremarkable that led her to the
misdiagnosis, until a positive bilharzia IgM antibodies were found. The patient
was treated accordingly using Praziquantel with low dose steroids.
Results:
Initiation of Praziquantel regimen and monitoring for a period of 2 months saw
an insidious resolution in symptoms and of elevated intracranial pressure over
a period of 9 months.
Conclusion:
It if often overlooked that raised intracranial pressure maybe due t
Neuroschistosomiasis,. Any patient presenting with the same and a history of
exposure to schistosoma-endemic areas any time in their past history should be
tested and treated with a positive Bilharzia IgM antibody prior to surgical interventions
being considered.
Keywords:
Neuroschistosomiasis; Benign intracranial hypertension; Cerebral
schistosomiasis; Papilledema.
INTRODUCTION
Schistosomiasis is a widely prevalent and potentially
devastating tropical parasitic disease. It affects more than 200 million people
with an additional 800 million people at risk of infection worldwide, thus
imposing a significant burden on public health. It has the second greatest
socio-economic ramification of any parasitic disease, following malaria.
According to the Global Burden of Disease Study 2016, the estimated
schistosomiasis global burden is 1.9 million disability-adjusted life years
(DALYs)1. Schistosomiasis is endemic
in more than 78 countries, with more than 90% of the infections occurring in
sub- Saharan Africa. Approximately six million individuals in Kenya are
afflicted with schistosomiasis with fifteen million more at risk of infection2. The geographical distribution varies among
the species. S. haemotobium is primarily found along the coast, Kano plains in
Western Kenya and certain areas around Lake Victoria. S. mansoni is prevalent
in Western regions and some parts of Central Kenya3.
Recent studies report a prevalence of 2.1% for S. mansoni, and 14.8% for S.
haematobium among school going children4.
The World Health Organization (WHO) has advocated for
integrated programs involving Mass Drug Administration (MDA) in endemic areas,
with the goal of eradicating schistosomiasis globally using a single dose of
Praziquantel 40 mg/kg5. Efforts to
eradicate schistosomiasis in Kenya have been ongoing since the MDA (Mass Drug
Administration) initiative initiated in 2009 with nationwide expansion in 20124. This has come with partial success as
schistosomiasis remains endemic in areas with inadequate access to clean water
and sanitation facilities.
The parasitic disease is caused by trematode blood
flukes of the genus Schistosoma that reside in the vascular system of humans
and other vertebrate hosts. The most important schistosomes that parasitize
humans are S. haemotobium, S. mansoni and S. japonicum.
Schistosomiasis is characterized by complex
pathogenesis. Upon contact with contaminated water sources, infective larvae
penetrate human skin, initiating the infection. Once inside the body, they
mature into adult worms, residing in mesenteric veins or vesicular venous
plexuses. Infections with S. mansoni and S. japonicum are associated GI
symptoms and chronic liver diseases. S. haemotobium results in urinary
schistosomiasis. Chronic infection may lead to deposition of eggs in host
tissue, inciting granulomatous reactions and fibrosis, which form the basis of
the disease’s diverse clinical presentation encompassing complications
affecting various organs, including the rare occurrence in the central nervous
system (neuroschistosomiasis). S. haemotobium, S. mansoni or S. japonicum
account for most cases of neuroschistosomiasis6.
Neuroschistosomiasis results from embolization of eggs
to the CNS. Once deposited, schistosome eggs release proteolytic enzymes in the
nervous tissue inducing a local eosinophilic inflammation. Ectopic eggs may
produce granulomatous lesions throughout the body. Chronic, severe infections,
due to accumulation of a vast number of eggs in tissues, leads to fibrosis,
calcification and occasionally dysplasia and malignant change. Cerebral
schistosomiasis is caused by S. japonium, resulting in acute encephalitis. The
higher incidence of CNS involvement of S.japonicum may be owed to their small
size eggs, which are released in higher numbers from the worm, and can be
carried easily to the brain. Neuroschistosomiasis may present in two clinical
syndromes, Spinal cord neuroschistosomiasis and localized Cerebral or
Cerebellar neuroschistosomiasis7.
The patients with cerebral neuroschistosomiasis may
present with signs and symptoms of elevated intracranial pressure and focal
neurological deficit, whereas those with spinal cord neuroschistosomiasis
experience progressive myelopathy, inclusive of cases of Cauda-equina root
involvement8. Cerebral
neuroschistosomiasis is clinically classified into acute schistosomal
encephalopathy and pseudotumoral encephalic schistosomiasis (PES). The latter
is a chronic form of cerebral neuroschistosomiasis and is rarely encountered in
clinical practice9.
Neuroschistosomiasis, although rare, carries
significant morbidity and mortality. It affects between 2% and 4% of the
estimated 200 million people with systemic schistosomal infections10. 90% of cases of neuroschistosomiasis are
found in sub-Saharan Africa, with cases of travel induced neuroschistosomiasis
outside of endemic areas11. Due to
the smaller number of cases reported and the increased rate of misdiagnosis,
only about 500 cases have been reported globally since 193012.
This case study involves a patient who was
misdiagnosed to have Benign Intracranial Hypertension instead of
Neuroschistosomiasis. Benign Intracranial Hypertension is an idiopathic
disorder characterized by increased Cerebro-spinal Fluid (CSF) pressure within
the intracranial cavity. It produces signs and symptoms such as visual
disturbances, headache, nausea and neurological deficits. Granuloma formation in
neuroschistosomiasis due to deposition of eggs may obstruct flow of CSF within
the ventricular system or subarachnoid space also causing similar intracranial
hypertension.
There is a gross lack of understanding of the
mechanisms of pathogenesis, diagnosis and treatment evidenced by the common
misdiagnosis of this condition in common clinical practice.
CASE REPORT
A HIV-seronegative 34-year-old female of african
ethnicity presented with a severe 8/10 headache and bilateral visual
disturbance, notable blurring, without concomitant nausea or vomiting.
Consciousness and cognition were preserved. Upon fundoscopic examination,
pronounced bilateral papilledema was confirmed prompting an urgent MRI scan.
The findings revealed features of Intracranial Hypertension possibly
Idiopathic, without any MR evidence of dural sinus thrombosis or space
occupying lesions. Further, bilateral optic nerve vertical tortuosity with
peri-optic nerve sheath effusions were observed along with prominence of
draining cortical veins, however, without identifiable intraluminal filling
defects. The results of the Glaucoma Hemifield Test (GHT) indicated abnormal
values for both eyes with deviations from normal limits.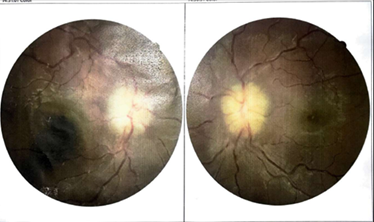
Figure
1. Fundoscopy on initial examination prior
to treatment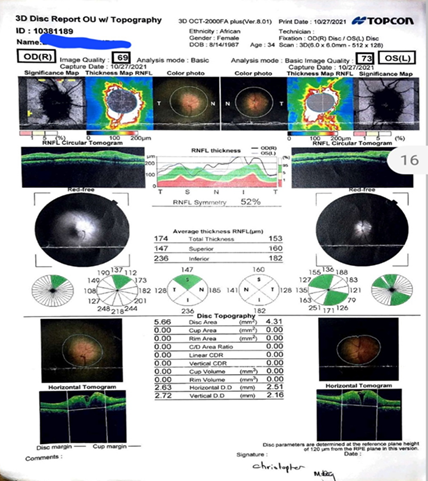
Figure
2. OCT scans prior to treatment
A complete blood count revealed a mildly iron
deficient picture with normal leukocyte and differential levels, inclusive of
eosinophil levels within normal limits. Malaria antigen was tested negative,
urine and stool lab examinations were unremarkable except for a positive
Helicobacter Pylori Antigen and ESR was elevated (54 mm/hr)
Based on the available results, a Ventriculoperitoneal
(VP) shunt was recommended as a measure to alleviate intracranial hypertension.
Upon physician assessment and review for possible prior to surgery, a serum Interferon
Gamma Release Assay (IGRA) was performed to confirm the suspicion of ocular
tuberculosis, yielding an equivocally positive result. Most importantly, serum
ELISA confirmed the presence of Bilharzia IgM antibodies.
After confirmation of the presence of Bilharzia IgM
antibodies, prompt intervention with praziquantel 2400mg twice a day for 3 days
was prescribed and repeated after a month, with 12mg of daflazocort once a day
for 2 weeks upon initiation of treatment, that was tapered to 6mg once a day
thereafter. Diamox (acetazolamide) 500mg twice a day was continued for 6 months
to maintain intracranial pressure and progress was monitored. Follow up
conducted after two months revealed relative improvement and in at 4 months,
the absence of Bilharzia IgM was achieved. A complete resolution of symptoms of
intracranial hypertension without diamox was found at 9 months from initial
dose of praziquantel given. A control MRI displayed normal findings indicating
an overall positive response to treatment. In spite of the resolution of
infection, anemia persisted on follow up.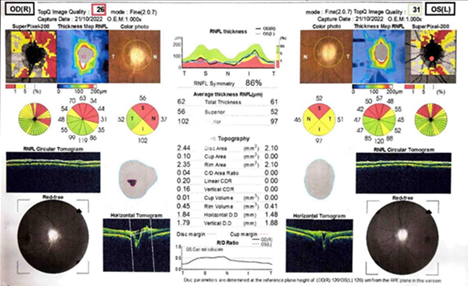
Figure
3. Fundocscopic examination and OCT scan 1
year post-intervention
A review of this relatively unique case done 18 months
post treatment confirmed a non recurrence of symptoms and a successful complete
resolution of the disease.
DISCUSSION
Cerebral neuroschistosomiasis commonly present with
symptoms of elevated ICP (headache, Cushing response, dizziness, vomiting,
papilledema, visual disturbances, speech disturbances, hemiparesis, nystagmus
and ataxia)13, neurological
manifestations (disturbed consciousness levels, dilated poorly reactive pupils,
increased muscle tone, exaggerated deep tendon reflexes and hyperventilation
with deep inspiration and expiration) and complications of increased ICP
(Ischaemia, tonic convulsions and herniation syndromes). Focal neurological deficits
may be present. These clinical manifestations closely mimic those of brain
tumors.
Diagnosis of cerebral schistosomiasis is a challenge
as neurological symptoms and imaging findings can be overlap with other
conditions such as brain tumors. Granuloma formation around eggs is a typical
finding in histopathology14. This
diagnostic approach remains the gold standard to confirm the diagnosis15.
Neuroimaging is an essential modality in diagnosis of
neuroschistosomiasis. Contrast-enhanced magnetic resonance imaging may exhibit
a linear enhancement pattern surrounded by multiple enhancing punctate nodules,
also known as ‘arborized’ appearance. These imaging findings are suggested to
be specific for cerebral neuroschistosomiasis. A review of 33 patients with
cerebral schistosomiasis, MRI scans presented a typical pattern of single or
multiple lesions compromising multiple intensely enhancing nodules, sometimes
with areas of linear enhancement16.
Praziquantel is the most effective drug used in the
treatment of neurochistosomiasis17.
Following diagnosis, Steroids, 8mg daily, may be considered before praziquantel
to decrease inflammation that may result from the cytotoxic effect of
praziquantel. Administration of 40-60 mg/kg of Praziquantel every 8 hours for
three days duration should be given, it acts by causing tetanic contraction and
paralyzing the parasite18. A second
dose is required 4-6 weeks following initial treatment to eliminate any
remaining parasites as praziquantel is ovastatic19
and has little effect on eggs or immature worms, which also makes it
ineffective in the early stages of infection20.
The case being studied is of a 34 year old female
highlights the diagnostic challenges associated with neuroschistosomiasis
attributable to atypical symptoms of raised intracranial pressures alone. Past
medical history and relevant exposure history is a significant guide to the
possibility of such presentation being a form of the an often overlooked cerebral
neuroschistosomiasis. This would necessitate the confirmation of this suspicion
through serology. The presence of Bilharzia IgM antibodies would allow non
surgical treatment for the condition.
It is yet to be established whether the progression to
neuro schistosomiasis, in the absence of immunosuppression, has a genetic
predisposition in certain populations as is the case here.
CONCLUSION
Many patients presenting with idiopathic benign
intracranial hypertension of late onset without and apparent cause may actually
have an infective etiology. Screening for infectious such as schistosomiasis
should be a strong consideration for physicians handling such cases prior to
recommending surgical interventions.
Acknowledgement
and Conflict of Interest: We would like to thank
the patient for allowing us to present their details in this case. The authors
have no conflicts of interest to declare.
REFERENCES
1. GBD
2016 DALYs, Hale Collaborators. Global, regional, and national
disability-adjusted life-years (DALYs) for 333 diseases and injuries and
healthy life expectancy (HALE) for 195 countries and territories, 1990-2016: a
systematic analysis for the Global Burden of Disease Study 2016. Lancet
2017;390:1260-1344.
2. Sang
HC, Muchiri G, Ombok M, Odiere MR, Mwinzi PNM. Schistosoma haematobiumhotspots
in South Nyanza, western Kenya: prevalence, distribution and
co-endemicitywithSchistosoma mansoniand soil-transmitted helminths. Parasit
Vectors 2015;7:125.
3. Hotez
PJ, Fenwick A. Schistosomiasis in Africa: An emerging tragedy in our new global
health decade. PLoS Negl Trop Dis 2009;3(9):e485.
4. Mwandawiro
CS, Nikolay B, Kihara JH, et al. Monitoring and evaluating the impact of
national school-based deworming in Kenya: study design and baseline results.
Parasit Vectors 2013;6:198.
5. Mutapi F, Maizels R,
Fenwick A, Woolhouse M. Human schistosomiasis in the post mass drug
administration era. Lancet Infect Dis 2017;17(2):e42-e48
6. Ross AG, Vickers D, Olds
GR, Shah SM, McManus DP. Katayama syndrome. Lancet Infect Dis 2007;7(3):218-224.
7. Nascimento-Carvalho CM,
Moreno-Carvalho OA. Neuroschistosomiasis due to Schistosoma mansoni: A review
of pathogenesis, clinical syndromes and diagnostic approaches. Rev Inst Med
trop S Paulo 2005;47(4):179-184.
8. Ferrari TC. Involvement
of central nervous system in the schistosomiasis. Mem Inst Oswaldo Cruz 2004;99:59-62.
9. Ibrahim M, Gad K, Khan T,
et al. Pseudotumoral Encephalic Schistosomiasis: A Literature Review. World
Neurosurg 2024;184:5-13.
10. Ferrari TCA, Moreira PRR.
Neuroschistosomiasis: Clinical symptoms and pathogenesis. Lancet Neurol
2011;10(9):853-864.
11. Clerinx
J, Van Gompel A. Schistosomiasis in travellers and migrants. Travel Med Infect
Dis 2011;9(1):6-24.
12. Steinmann P, Keiser J,
Bos R, Tanner M, Utzinger J. Schistosomiasis and water resources development: Systematic
review, meta-analysis, and estimates of people at risk. Lancet Infect Dis
2006;6(7):411-425.
13. Colley
DG, Bustinduy AL, Secor WE, King CH. Human schistosomiasis. Lancet 2014;383(9936):2253-2264.
14. Zaqout A, Abid FB,
Murshed K, et al. Cerebral schistosomiasis: Case series from Qatar. Int J
Infect Dis 2019;86:167-170.
15. Liu H, Lim CC, Feng X, et
al. MRI in cerebral schistosomiasis: characteristic nodular enhancement in 33
patients. AJR Am J Roentgenol. 2008;191(2):582‐588.
16. Bill PL, Shakir RA, Newman PK, et al. Tropical
Neurology. Philadelphia, PA: WB Saunders Co. Schistosomiasis 1996;295-316.
17. Vale
N, Gouveia MJ, Rinaldi G, Brindley PJ, Gärtner F, da Costa JM. Praziquantel for
schistosomiasis: single-drug metabolism revisited, mode of action, and
resistance. Antimicrob Agents Chemother 2017;61(5):2582-2516.
18. Lighter J, Kim M, Krasinski
K. Intramedullary schistosomiasis presenting in an adolescent with prolonged
intermittent back pain. Pediatric Neurol 2008;39(1):44-47.
19. Xiao
SH, Sun J, Chen MG. Pharmacological and immunological effects of praziquantel
against Schistosoma japonicum: a scoping review of experimental studies. Infect
Dis Poverty 2018;7.