A Rare Case of Cervical Vagus Nerve Schwannoma in an Adult Patient: Diagnostic and Therapeutic Strategy
Abstract
Schwannomas originating from the Schwann cells are a
rather uncommon benign tumor. Schwannomas often don't cause any symptoms, but
larger tumors may cause vague symptoms because they squeeze nearby anatomical
tissues. The pathognomonic symptoms of vagus nerve schwannomas are isolated
neck masses, hoarseness of voice and paroxysmal coughing that occurs upon
palpation. Imaging tests may be quite important in making a difficult
preoperative diagnosis. The preferred course of treatment is surgery that removes
the tumor entirely. Nerve damage during surgical resection is associated with
significant morbidity. We report a case of vagus nerve schwannoma in a male
adult patient in this study.
Keywords: Schwannoma;
Vagal schwannoma; Vagus nerve; Cervical mass; Magnetic Resonance imaging
Introduction
Schwann cells give rise to the benign tumor known as
schwannoma, which grows slowly1-6.
Another name for it is a neurilemmom1,7.
In 1908, Prague doctor Josay Verocay was the first to characterize schwannoma
as a pathological disease1. Less
than 5% of soft tissue sarcomas are malignant schwannomas; the remainder are
benign8,9. Schwannomas equally
impact the sexes. Although it can affect people of any age, the majority of
instances occur in those between the ages of three and five. The likelihood of
a malignant change in them is extremely low1,4-7,10,11.
Of the instances, extracranial schwannomas make up between 1/4 and 1/3 of the
total. The primary site of origin in the neck region is the vagus nerve, which
is followed by the cervical sympathetic chain. When combined, they make up
around 25% of all extracranial schwannomas in the head and neck12,13. There are two types of cervical
schwannomas: medial and lateral. The last four cranial nerves or the cervical
sympathetic chain, give rise to the medial groups. The groups that emerge from
the cervical or brachial plexus trunk are known as the lateral groups1,8. A rare tumor called extracranial
cervical vagal nerve schwannoma (VNS) usually manifests itself between the ages
of three and five. As of 2016, there are only 133 cases that have been reported
in the literature3,11.
Furthermore, the literature only has 50 cases of cervical sympathetic
schwannoma8.
A rare tumor called extracranial cervical vagal nerve
schwannoma (VNS) usually manifests itself between the ages of three and five.
As of 2016, there are only 133 cases that have been reported in the literature3,11. Furthermore, the literature only has
50 cases of cervical sympathetic schwannoma.8The main symptom of patients with
VNS in the neck is hoarseness because of vocal cord paralysis.
When the mass is palpated, patients may also have an
involuntary cough, which clearly suggests the origin of the vagal nerve3,4,6,7,10,11,14. Dysphonia and dysphagia
are other possible symptoms, especially if the tumor is large11.
Preoperative consideration of schwannomas is highly
challenging due to their rarity and the absence of a neurologic dysfunction as
a presenting symptom. When it comes to neck tumors, a number of differential
diagnoses might be considered. These include lipomas, teratomas, thyroid cysts
or nodules, inflammatory cervical lymphadenopathy, submandibular salivary gland
tumors, neurofibromas, and metastatic cervical lymphadenopathy1,3,6,7,10,11,13,14. Complete surgical
resection is the cornerstone of treatment, but due of its close proximity to
the carotid artery and the vagus nerve, from which it originates, it may be
technically difficult3,4. The
tumor's location, size, and relationship to surrounding vessels all influence
the strategy. The main side effect after VNS excision is hoarseness of voice4.
Case report
A 26-year-old male patient was referred to the
outpatient clinic of the Oral and Maxillofacial Surgery Department with a
history of a mass in the right lateral neck area. Patients with no specific
pathological antecedents. The history of her ilness goes back to 1 year the
history of his malaria goes back 1 year with the appearance of a right latero
cervical mass progressively increasing in volume with no inflammatory signs
opposite, no associated compressive signs and no fistulization to the skin. However,
the patient had begun to experience some nonspecific symptoms in the previous
year. The main complaints were mild hoarseness of voice, episodes of
bradycardia. His physical examination revealed, a non-tender well-limited soft,
painless right latero-cervical mass, mobile in relation to the 2 planes
involving sectors 2 and 3, with no inflammatory signs opposite the mass.
Notably, the palpation of the neck induced paroxysmal cough. No cervical lymph
nodes were palpated. All the cranial nerve examinations were normal. At the
nasofibroscopy: no backflow or visible mass the 2 vocal cords are mobile.
On the cervical CT scan, a voluminous mass well
limited to the right side of the cervix with carotid and jugular vascular
contact, the site of some calcification without contrast in arterial washout or
typical intercarotid topography, suggesting a glomus origin. (Figure 1). 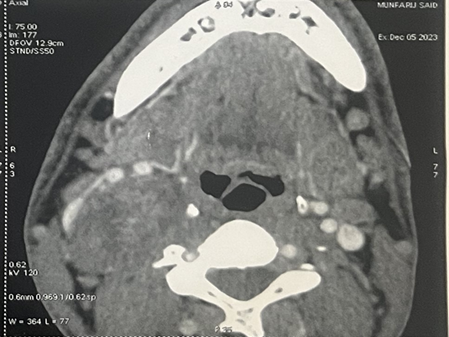
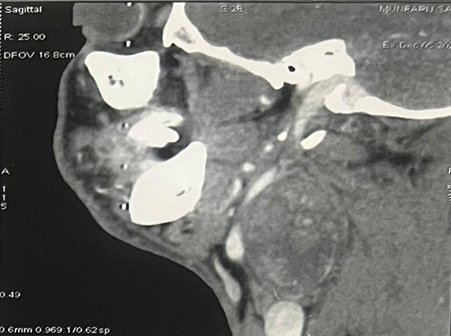
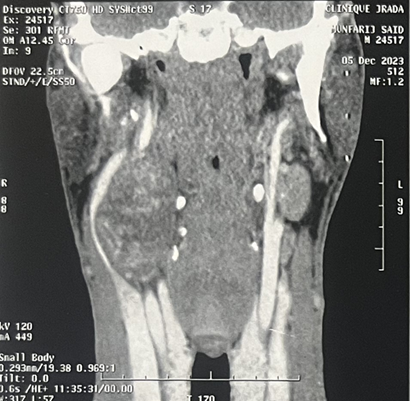
Figure 1: CT imaging of a vagal schwannoma (red arrow) on the right
cervical side: (A) sagittal (B) axial and coronal (C) view.
Magnetic resonance imaging (MRI)
of the neck confirmed a 39x38x60 mm properly limited mass with high signal
intensity on T1-weighted MRI and a heterogeneous, low signal intensity on T2
weighted MRI (Figure 1). After
intravenous administration of gadolinium-based contrast material, the lesion
showed irregular peripheral enhancement. Carotid artery angiography was
performed because of the close relationship between the mass and the carotid
artery. The angiography showed a normal filling in the right common, internal,
and external carotid arteries and their branches. (Figure 2).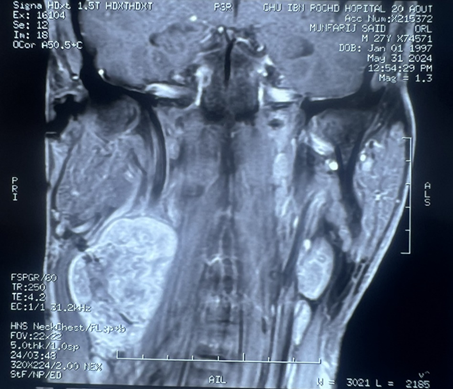
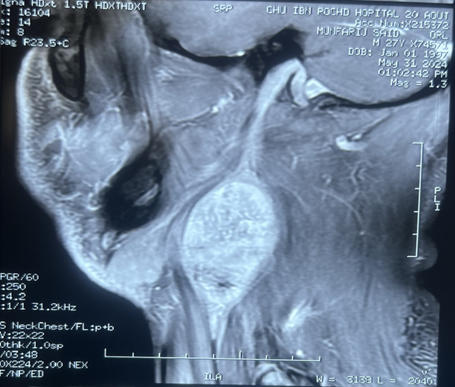
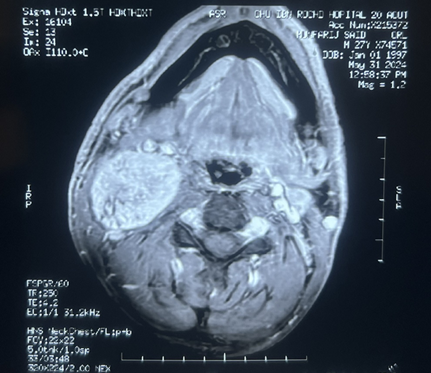
Figure 2: Coronal (A) sagittal (B) and axial(C) view. Gray
arrow: well-circumscribed, h mass with dimensions of 39x38x60 mm in the right
lateral cervical region directly under the right sternocleidomastoid muscle,
separating the internal jugular vein and the carotid artery. Yellow arrow:
compressed internal jugular vein. Black arrow: carotid artery.
Under general anesthesia, an oblique cervical
incision was made starting from the right mastoid apex. A yellowish mass lesion
was observed that was 5x4cm in diameter, originating from the vagus nerve and
medially adjacent to the common carotid artery. It was extending laterally to
the internal jugular vein and sternocleidomastoid (SCM) muscle, and superiorly
to the skull base (Figure 2). The
mass was carefully dissected from the vagus nerve and other adjacent
structures, with care taken to protect the vagal nerve’s integrity. The
specimen was sent for frozen section and a schwannoma originating from the
vagus nerve was confirmed. No extra surgical intervention was considered and
the operation was completed. Postoperative follow up of the patient was uneventful
and he was discharged on the 7th postoperative day.
Discussion
Schwannomas
in the head and neck are slow-growing, benign tumors. But it's crucial to
remember that malignant transformation occurs with an incidence rate of 8-13.9%,
which is something to keep in mind when managing schwannoma2. They most often occur as a single mass,
and they are typically asymptomatic. In the later stages, other nonspecific
symptoms such dysphagia, nasal obstruction, and dyspnea in the supine position
arise because the surrounding anatomical structures are compressed by the
larger mass2. Upon cervical mass
palpation, patients with vagus nerve schwannomas may occasionally show with
hoarseness of voice and a particular pathognomonic symptom of paroxysmal cough1,2. Inflammatory cervical lymphadenitis,
metastatic lymphadenopathy, salivary gland neoplasms, paraganglioma, lymphoma,
branchial cleft cyst, carotid body tumors, neurofibroma, and carotid aneurysm
should all be considered in the differential diagnosis of such neck form3.
On the
other hand, preoperative imaging tests and fine needle aspiration cytology
(FNAC) may be useful in supporting the diagnosis. Furthermore, because imaging
tests provide information about the lesion and the surrounding anatomy, they
are crucial to surgical planning1.
Since MRI offers a more thorough depiction of the soft tissues, it is seen as
being preferable in this situation versus CT scans1,2.
Schwaannomas with high attenuation relative to the surrounding muscle with low
to moderate heterogeneous contrast enhancement are described by
contrast-enhanced computed tomography scans. Internal cystic alterations
resulting from bleeding, necrosis, or mucinous degeneration are occasionally
seen. T2-weighted MR pictures reflect a heterogeneously hyperintense signal,
but T1-weighted MR images display the isointense signal of the schwannoma in
relation to the surrounding muscle.
Imaging can show how the mass relates to
nearby structures and provide some information about the mass composition,
which is helpful in differentiating between the items in the differential list.
The contents of the carotid sheath can be displaced to distinguish between
vagal and cervical sympathetic chain schwannomas. Vagal schwannomas tend to
divide the internal jugular vein and carotid artery, whereas the latter shift
both laterally since they are not inside the carotid sheath. Since cervical
lymph nodes are lateral to the carotid sheath, lymph node involvement might be
taken into consideration when both the carotid and jugular veins are moved
medially. At the carotid bifurcation level, carotid body tumors divide the
carotid arteries into the internal and exterior segments7. Schwaannomas appear round or elliptical
on ultrasound imaging, with distinct borders and internal echo reflection.
Ultrasound can show the link between the nerves if the diameter of the
originating nerve is large enough1.
Schwannomas
typically appear inhomogeneous on CT, with a well-defined mass linked to
peripheral enhancement and interior cysts8.
The most precise and sensitive preoperative method is magnetic resonance
imaging (MRI), which enables a more precise determination of the nerve of
origin. Specific indications (split fat, fascicular, target) and signal
patterns (low signal intensity on T1-weighted images, high signal intensity on
T2-weighted images) on MRI are indicative of schwannomas9. On improved T1 imaging, certain authors
have shown that a schwannoma is a lesion with a particular peripheral
hyperintense rim and center low intensity10.
Furthermore, MRI rather than CT may be able to clarify the connection between
the schwannoma and its originating nerve.
Surgical
indications for the treatment of extracranial head and neck schwannomas should
carefully consider the advantages of the procedure against the possibility of
nerve palsy following excision. Several treatment strategies, including radical
tumor excision, intracapsular enucleation, and waiting and seeing, have been
suggested for the management of schwannomas11.
Generally, the tumor's size, location, and relationship to nearby vessels
determine which technique is best.
Although
the surgical technique is the preferred course of action, the observational
strategy is also warranted due to the lesion's noninvasive nature and sluggish
progression. Intracapsular enucleation, debulking, and radical excision with
nerve grafting are the primary surgical alternatives12. Radical excision is removing the tumor
entirely while sacrificing the perineum's nerves that are attached to it. Nerve
transplantation is therefore scheduled. A more conservative procedure called
intracapsular resection involves removing just the tumor's core while
maintaining the tumor capsule connected to the perineum. Recurrence risk is
elevated with tumor debulking. Nerve integrity may not always be preserved even
with intracapsular dissection, therefore prior to surgery, patients should be
well informed about It may be difficult to maintain nerve integrity even with
intracapsular dissection, thus patients should be fully told about the
possibility of neurologic impairments prior to surgery. Surgery's primary objective
should always be to remove the tumor with the least amount of neurologic
damage. The nerve of origin is probably going to be impacted by the procedure
used. According to reports, 100% and 67% of patients experience nerve palsy
after intracapsular dissection and full excision, respectively13.
Intracapsular enucleation is the preferred
method of tumor removal because the complications are usually temporary and
treatment is not necessary in most cases; in fact, some authors report a
preservation of neural function of more than 30% when compared to radical tumor
resection with a primary anastomosis14.
It's interesting to note that a number of studies have demonstrated that
surgeons can keep good vocal function after surgery by protecting nerves as
much as possible by knowing the anatomical features of the recurrent laryngeal
and nonlaryngeal reflexes and the eccentric growth pattern of the tumor15,16. Additionally, some writers suggest
using an electromyography (EMG) system during tumor removal as a way to assist
prevent motor nerve paralysis in the treatment of extracranial schwannomas of
the head and neck. This could help preserve function. A comparable approach has
been used in clinical practice for parotid gland and thyroid surgery17.
According
to numerous reports in the literature, primary repair or a nerve transplant
should be carried out using a microsurgical method, with or without
medialization of the vocal cord, if the nerve cannot be saved during surgery21. Lastly, a wait-and-see strategy was
proposed by some writers18,
delaying surgery only in the event that the symptoms deteriorate and
neurological weakening turns out to be clinically significant. After a vagal
nerve schwannoma is removed, common postoperative issues arise because the
tumor originated directly from the nerve fibers. These consist of vocal cord
palsy, nerve damage, and voice hoarseness. As a result, post-operative care and
preoperative evaluation of speech and swallowing are crucial to these patients'
voice and swallowing rehabilitation. In particular, the majority of patients
report having hoarseness. These consist of vocal cord palsy, nerve damage, and
voice hoarseness. As a result, post-operative care and preoperative evaluation
of speech and swallowing are crucial to these patients' voice and swallowing
rehabilitation. To be more precise, after schwannoma removal, hoarseness is
noted by the majority of patients, but after tumor resection, vocal cord
paralysis affects 85% of patients. Pharyngolaryngeal anesthesia, aspiration,
and cranial nerve palsies IX, XI, and XII are additional frequent side effects
after schwannoma excision that may be temporary or permanent19. Lastly, Horner's syndrome and changes in
heart rate are two further rare problems that have been reported20.
The vagus
nerve lowers heart rate, a fact that has been extensively reported in the
literature22. Patients who are
not candidates for surgery might benefit from radiation therapy if they are
symptomatic or from an observational strategy (i.e., "wait and see"),
albeit the research does not provide sufficient evidence to support either
course of action. In contrast to the auditory nerve, which is primarily treated
with radiation, there is really less data regarding the effectiveness of
radiation for schwannomas of the head and neck23.
However, there is growing evidence supporting the use of radiation therapy for
non-surgical candidates with schwannomas of other cranial nerves (III, IV, V,
and VI), with a local control rate ranging from 90% to 100%24,25. Regarding the radiobiological
behavior of vagal schwannomas specifically, several writers have highlighted
that this entity's inherent radioresistance serves as one of the primary
barriers to the effectiveness of radiation therapy29.
Because of this, the standard schedule choice described in the literature for
comparable clinical situations is usually hypofractionated and requires the use
of stereotactic radiotherapy26.
However, it is important to consider the
possibility of acute adverse events when contemplating radiotherapy as an
alternate therapeutic option for surgical candidates. These adverse events
typically resolve on their own and can be made better by giving small doses of
steroids. Large-volume and dumbbell-shaped tumors are important predictors of
the incidence of these acute adverse effects27.
Regarding the potential use of a wait-and-see strategy, it is crucial to
suggest it only to patients who agree to routinely visit the doctor and undergo
imaging exams as directed by the doctor; in fact, protocol may be changed to
active treatment in cases of radiologic growth, pain, or new cranial nerve
dysfunction28. 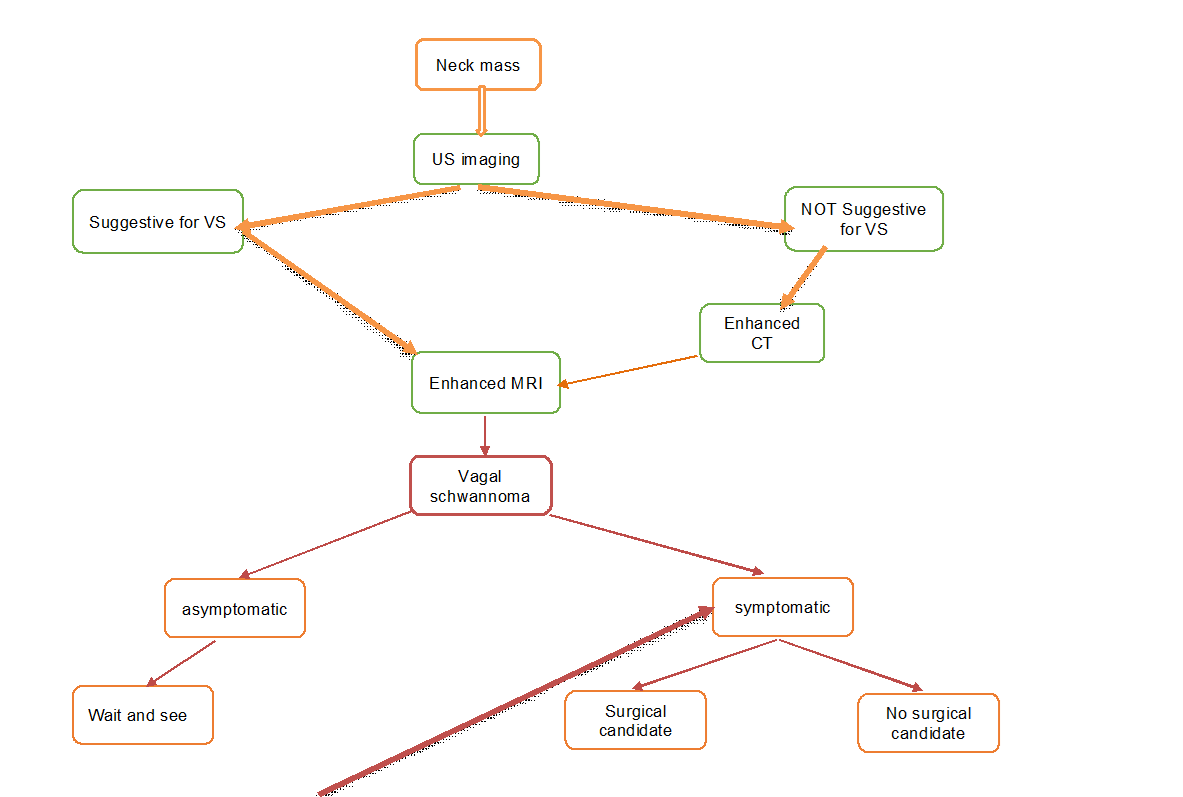
It is
difficult to control anesthesia during the excision of a vagal nerve
schwannoma. There have been reports of severe bradycardia resulting in
hypotension and anomalies in the ECG5.
Mukherjee et al.'s patient went into cardiac arrest while having a sizable
vagal schwannoma removed6. Direct
vagal stimulation was assumed to be the most likely cause of cardiac arrest. In
addition, our patient had two episodes of bradycardia with hypotension, which
made injecting atropine necessary because the mass release performed by the
surgeon had not been very helpful. When inducing anesthesia, every effort was
made to avoid employing anesthetics that could cause bradycardia. With the bulk
removed, there was no more bradycardia episode. To stop a recurrence, the tumor
capsule must be completely removed4.
Postoperative
reports indicate that hoarseness occurs in nearly all instances, while 85% of
cases result in spinal cord paralysis4.
It is crucial to evaluate the voice cords' range of motion prior to surgery.
Therefore, as part of the preoperative evaluation, the anesthetist must also
advise the patient about potential postoperative neurological problems.
Conclusion
Vagus nerve schwannomas are rarely occurring neck
masses. The identification and treatment of vagal schwannomas are undoubtedly
difficult in the clinical setting. In actuality, symptoms are typically
non-specific, which could cause delays in diagnosis. Imaging is a very helpful
modality for diagnosis and surgical planning. Furthermore, it should be
carefully addressed because post-operative sequelae, such as vagal nerve
injury, still represent important issues, even though complete surgical
resection is the standard therapeutic method. We have offered a vagal schwannomas
care algorithm based on a combination of scientific data from the literature
and our clinical expertise.
References
1.Behuria
S, Rout TK, Pattanayak S. Diagnosis and management of schwannomas originating
from the cervical vagus nerve. Ann R Coll Surg Engl 2015;97:92-97.
2.Kanatas
A, Mücke T, Houghton D, Mitchell DA. Schwannomas of the head and neck. Oncol
Rev 2009;3:107-111.
3.Cavallaro
G, Pattaro G, Iorio O, Avallone M, Silecchia G. A literature review on surgery
for cervical vagal schwannomas. World J Surg Oncol 2015;13:130.
4.Chiofalo MG, Longo
F, Marone U, Franco R, Petrillo A, Pezzullo L. Cervical vagal schwannoma. A case report. Acta Otorhinolaryngol
Ital 2009;29:33-35.
5.Wood BM, mcneil WT. Schwannoma of the vagus nerve.
Anaesthesia 1986;41:1130-1132.
6.Mukherjee
DK. Neurilemmoma of the vagus nerve, A case report. J Laryngol Otol 1979;93(2):187-192.
7.Sandler
ML, Sims JR, Sinclair C, et al. Vagal schwannomas of the head and neck: a
comprehensive review and a novel approach to preserving vocal cord innervation
and function. Head Neck 2019;41(7):2450-2466.
8.Kim
SH, Kim NH, Kim KR, Lee JH, Choi HS. Schwannoma in head and neck. Preoperative
imaging study and intracapsular enucleation for functional nerve preservation.
Yonsei Med J 2010;51(6):938-942.
9.Dosemane
D, Kabekkodu S, Jaipuria B, Sreedharan S, Shenoy V. Extracranial non-vestibular
head and neck schwannomas. A case series with the review of literature. Braz J
Otorhinolaryngol 2022;88:9-17.
10.Crist
J, Hodge JR, Frick M, Leung FP, Hsu E, Gi MT, Venkatesh SK. Magnetic Resonance
Imaging Appearance of Schwannomas from Head to Toe. A Pictorial Review. J Clin Imaging
Sci 2017;7:38.
11.Gibber
MJ, Zevallos JP, Urken ML. Enucleation of vagal nerve schwannoma using
intraoperative nerve monitoring. Laryngoscope 2012;122(4):790-792.
12.
Akheel
M, Mohd Athar I, Ashmi W. Schwannomas of the head and neck region: A report of
two cases with a narrative review of the literature. Cancer Res Stat Treat 2020;3(3):517-525.
13.
Wang
B, Yuan J, Chen X, Xu H, Zhou Y, Dong P. Extracranial non-vestibular head and
neck schwannomas. Saudi Med J 2015;36(11):1363-1366.
14.
Valentino
J, Boggess MA, Ellis JL, Hester TO, Jones RO. Expected neurologic outcomes for
surgical treatment of cervical neurilemmomas. Laryngoscope 1998;108(7):1009-1013.
15.
Lin J, Martel W. Cross-sectional imaging of peripheral nerve sheath tumors:
Characteristic signs on CT, MR imaging, and sonography. Am J Roentgenol 2001;176(1):75-82.
16.
Xu
T, Liu Y, Li S, Zheng L. Pre-operative embolization and excision of vagal
schwannoma with rich vascular supply: A case report and literature review.
Medicine 2022;101(4):28760.
17.
Ijichi
K, Kawakita D, Maseki S, Beppu S, Takano G, Murakami S. Functional Nerve
Preservation in Extracranial Head and Neck Schwannoma Surgery. JAMA Otolaryngol
Head Neck Surg 2016;142(5):479-483.
18.
Gilmer-Hill
HS, Kline DG. Neurogenic tumors of the cervical vagus nerve: Report of four
cases and review of the literature. Neurosurgery 2000;46(6):1498-1503.
19.
Khafif
A, Segev Y, Kaplan DM, Gil Z, Fliss DM. Surgical management of parapharyngeal
space tumors: A 10-year review. Otolaryngol Head Neck Surg 2005;132(3):401-406.
20.
Kumar KP, Alam MS. “Collateral Damage”: Horner’s
Syndrome Following Excision of a Cervical Vagal schwannoma. Int. J. Appl. Basic
Med. Res 2018;8:190-192.
21.
Pankratjevaite L, Dreyer NS, Dauksa A, Sarauskas V. Challenging diagnosis of
cervical vagal nerve schwannoma. J Surg Case Rep 2022;2022(4).
22. Allen
E, Pongpaopattanakul P, Chauhan RA, Brack KE, Ng GA. The Effects of Vagus Nerve Stimulation on
Ventricular Electrophysiology and Nitric Oxide Release in the Rabbit Heart.
Front Physiol 2022;13:867705.
23.
Yafit
D, Horowitz G, Vital I, Locketz G, Fliss DM. An algorithm for treating
extracranial head and neck schwannomas. Eur Arch Otorhinolaryngol 2015;272:2035-2038.
24.
Fionda
B, Rembielak A. Is There Still a Role for Radiation Therapy in the Management
of Benign Conditions? Clin Oncol 2023;35(11):698-700.
25.
Kim
IY, Kondziolka D, Niranjan A, Flickinger JC, Lunsford LD. Gamma Knife surgery
for schwannomas originating from cranial nerves III, IV, and VI. J Neurosurg
2008;109:149-153.
26.
Li Y, Lou J, Qiu S, Guo Y, Pan M. Hypofractionated stereotactic radiotherapy
for dumbbell-shaped hypoglossal schwannomas: Two cases of long-term follow-up
and a review of the literature. Mol Clin Oncol 2016;5(2);371-374.
27.
Kim
YG, Park CK, Jung NY, Jung HH, Chang JH, Chang JW, Chang WS. Early-onset
adverse events after stereotactic radiosurgery for jugular foramen schwannoma:
A mid-term follow-up single-center review of 46 cases. Radiat Oncol 2022;17(1):89.
28.
Merzouqi
B, El Bouhmadi K, Oukesou Y, et al. Head and neck paragangliomas: Ten years of
experience in a third health center. A cohort study. Ann Med Surg 2021;66:102412.
29.
Lahoti
BK, Kaushal M, Garge S, Aggarwal G. Extra vestibular schwannoma: A two year
experience. Ind J Otolaryngol Head Neck Surg 2011;63(4):305-309.