A Rare Case of Meningococcemia with Disseminated Intravascular Coagulation in an 11-Year-Old Boy
Abstract
Invasive Meningococcal
Disease; a rare life-threatening bacterial infection caused by Neisseria
meningitidis, can manifest itself as meningitis or meningococcemia or a
combination of both. This case report details a case of fulminant
meningococcemia with DIC in an 11 year old unvaccinated boy, presenting with
high grade fever, palpable purpuric rash with inter spread petechiae and
ecchymosis primarily over the extremities and signs of circulatory collapse
that recovered after getting timely antibiotic therapy, aggressive fluid
resuscitation, intensive care monitoring and multidisciplinary management,
highlighting the critical role of rapid diagnosis and timely management of
meningococcal disease by recognizing the characteristic rash in a febrile, ill
looking child in shock. This case report also shows the importance of
meningococcal vaccination and post exposure prophylaxis of close contacts.
Keywords: Invasive meningococcal disease; Neisseria meningitidis;
Fulminant meningococcemia; Meningococcal vaccination
Case
Presentation
An 11-year-old
unvaccinated boy, previously healthy, presented to the emergency department on
12/12/24 5:21 am with one-day history of high-grade fever, multiple episodes of
vomiting and loose stools, two episodes of generalized seizures, severe
muscular pain especially in legs associated with rapidly progressing rash
starting from face, trunk and extending to extremities. On arrival, patient was
drowsy but arousable, toxic looking, irritable with the oral temperature of
102.7-degree Fahrenheit. He was tachycardiac (HR 138) with poor peripheral
pulses, CRT (>4 seconds) and BP below 50th centiles (B.P 95/55). He was
tachypneic and was maintaining oxygen Saturation level at 94% at room air.
Physical examination revealed a palpable purpuric rash with inter spread
petechiae and ecchymosis, primarily on the trunk and extremities associated
with B/L small subconjunctival hemorrhages. Neurological examination revealed
GCS of 9/15 with no focal neurological deficits but positive signs of
meningismus. Motor examination showed power of 3/5 in all limbs.
Investigations
|
Investigations
|
Findings
|
|
Total
Leucocyte Count (TLC) (/mm3) |
17000/mm3
|
|
Platelets
(/mm3) |
64000/mm3
|
|
Urea
(mg/dl) |
58
|
|
Creatinine
(mg/dl) |
1.12
|
|
Serum
Potassium (mEq/L) |
2.5
|
|
Prothrombin
Time (PT) (seconds) |
17.3
|
|
International
Normalized Ration (INR) |
1.63
|
|
Activated
Partial Thromboplastin time (APTT) |
47.1
|
|
D
Dimers (ng/ml) |
>10,000
|
|
Arterial
Blood Gases (ABGs) |
Metabolic
acidosis with elevated lactate |
|
Blood
for Culture and Sensitivity (Blood CS) |
Neisseria
Meningitidis (serogroup B) |
|
Chest
X ray (CXR) |
B/L
clear lung fields |
|
CT
Scan head |
Unremarkable
|
|
2
D Echocardiography |
Normal
|
Diagnosis
The clinical
presentation of fever, hypotension, petechial rash, with positive blood
cultures and positive associated lab findings confirmed the diagnosis of
fulminant meningococcemia with DIC.
Management
Patient was admitted in
intensive care unit. Ionotropic support with aggressive resuscitation with
crystalloids and vasopressors was initiated with Injection dopamine started
(12/12/24 - 16/12/24) and then tapered off gradually. Due to Disseminated Intravascular
Coagulation (DIC), Lumber Puncture (LP) was withheld. Coagulopathy was
corrected with Fresh Frozen Plasma (FFPs) and platelets. Patient was transfused
8 FFPs and 4 platelets in total. Empiric antibiotics; Inj. Ceftriaxone
(Rocephin) and Inj. Vancomycin were commenced, later Inj. Meropenem and Inj.
Linezolid (Nezkil) were added due to blood cultures revealing bacterial
sensitivity to these antibiotics. Seizure prophylaxis was done with Syrup
Levetiracetam (Lerase). Inj. Nalbuphine (Nalbin) was given in the early course
of admission due to severe myalgias and arthralgias that patient had.
Strict vital monitoring
was done throughout course of admission and patient was observed for
development of complications. Patient developed grade 3 bed sores in
sacrococcygeal region due to necrotic patch of rash and inactivity despite air
mattress provided, surgical consultation was done and wound was regularly
cleaned, debrided and dressed (Figure 1).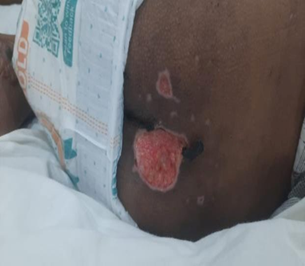
Figure 1: Grade three bed sores in the
Sacrococcygeal region of the patient
Discussion
Invasive
Meningococcal disease, manifesting itself as meningitis or meningococcemia or a
combination of both1 has an
annual incidence in the Europe and United States of 1 case per 100,000 and 0.35
cases per 100,000 respectively with fluctuations in its incidence in some
epidemic regions, like Sub-Saharan Africa, where case fatality rates are
recorded as high as 70%2. Highest
incidence rates are observed in infants and young children aged 1 to 4. It is
even rarer (≤0.1)3 for those aged 11-15
as in our case.
Twelve
distinct serogroups of N. meningitidis, a human specific gram negative
encapsulated diplococcus causing meningococcemia, transmitted via droplet
aerosols or secretions from the nasopharynx of colonized contacts have been
identified1, with serogroups W
(40.2%), B (31.7%) and C (10.4%)being the most common4. In our case serogroup B was isolated from blood
cultures. Asymptomatic pharyngeal colonization is the initial step of infection
and when the organism gains access to the systemic circulation, it causes
meningococcemia with clinical features including fever, hemorrhagic rash (often
petechial or purpuric) and signs of circulatory collapse (deranged capillary
refill time, hypotension) often progressing rapidly to septic shock in an
ill-looking child favoring suspicion of meningococcal disease similar to the
case of meningococcemia reported in an 11 month old infant in kathmandu5. An observational study done on 233 children up to
15 years of age also showed that most children with meningococcal infection are
ill looking, have a purpuric rash, fever and delayed capillary refill time6. Our case also presented with similar features
along with positive signs of meningismus but confirmation of meningitis
couldn’t be made as LP was not performed due to DIC.
Diagnosis
should be clinically made while awaiting organism identification via
Cerebrospinal Fluid (CSF) analysis and blood cultures (meningococcemia) due to
rapid progression and high fatality rates of the disease and immediate
empirical antibiotic therapy should be commenced along with aggressive fluid
resuscitation and vasopressor support for maintaining blood pressures and shock
management3. Our case was also
managed with intensive care monitoring, seizure prophylaxis, symptomatic
management, empirical antibiotics, fluids, vasopressors and hemodynamic support
like the case reported in kathmandu5.
Recommended first line antibiotics are Cephalosporins or penicillin G3 but due to high bacterial resistance rates of
these antibiotics in our area, Vancomycin and Ceftriaxone were commenced as
first line antibiotics while awaiting blood cultures. Blood cultures are
positive in up to 3/4th cases of meningococcemia5. In our
case also blood cultures came out to be positive. In rare cases, surgical
intervention may be needed to manage complications due to tissue ischemia,
which may require debridement or even amputation of necrotic tissued3 as seen in a case report of a 5-month-old girl
from Poland where hemorrhagic lesions of the extremities evolved to necrosis
leading to hands and feet amputation6,7. In our
case patient’s rash on sacrococcygeal region got necrotic and was debrided.
The
patient in our case report was not immunized with meningococcal vaccine similar
to the case reported in Kathmadu5,
highlighting the importance of meningococcal vaccination. Post-exposure
antibiotic prophylaxis (e.g., rifampin or ciprofloxacin) (PEP) of close
contacts of individuals diagnosed with meningococcal disease should be done to
reduce the risk of transmission3 and was
done in our case with Ciprofloxacin.
Conclusion
This case underscores the
critical role of rapid diagnosis of meningococcal disease by recognizing the
characteristic rash and maintaining a high index of suspicion in febrile
patients with mentioned systemic symptoms as well as practicing emergent interventions
including timely antibiotic therapy, aggressive fluid resuscitation, intensive
care monitoring , vaccination programs, post exposure prophylaxis and
multidisciplinary management for meningococcal disease to prevent further
outbreaks and reduce the mortality associated with this rare but life
threatening condition.
References
1. Rausch-Phung EA, Siddiqui
JA, Gulick PG. Meningococcemia. Infectious
Diseases in Critical Care 2025:341-371.
2. Bouneb R,
Mellouli M, Regaieg H, Majdoub S, Chouchène I, Boussarsar M.
Meningococcemia complicated by myocarditis in a 16-year-old young man: a case report. Pan Afr Med J
2018;29:149.
3. Hulays Alharbi S, Ayad Αnazi AM,
Abdulaziz Alfaleh T, et al. Meningococcemia: An emergent medical
condition-updated review article.
4. Bobde S, Sohn WY,
Bekkat-Berkani R, et al. The Diverse Spectrum of Invasive Meningococcal
Disease in Pediatric and Adolescent Patients: Narrative Review of Cases
and Case Series. Infect Dis
Ther 2024;13(2):251.
5. Shrestha R, Karki S,
Khadka M, et al. Meningococcemia in an 11 Months Old Infant. Case Rep Infect Dis
2023;2023(1):8951318.
6. Wells LC, Smith JC,
Weston VC, Collier J, Rutter N. The child with a non-blanching rash: how
likely is meningococcal disease? Arch Dis Child 2001;85(3):218-222.
7. Medeiros I, Reis Melo A,
Baptista V, Ribeiro A. Meningococcemia: rare but life-threatening. Case Reports
2018;2018:2018-226914.