Advancements in Cancer Immunotherapy: A Comprehensive Review of Immune Checkpoint Inhibitors with a Focus on Pembrolizumab and Emerging Strategies
Abstract
The introduction of immune checkpoint inhibitors (ICIs) has marked a significant breakthrough in oncology, fundamentally altering cancer treatment paradigms.
This review examines the transformative impact of ICIs, including PD-1
inhibitors (Nivolumab, Pembrolizumab, Cemiplimab), PD-L1 inhibitors
(Atezolizumab, Durvalumab, Avelumab), and CTLA-4 inhibitors (Ipilimumab), all
of which have received FDA approval for various malignancies. These agents
enhance survival outcomes by reactivating the immune system to target cancer
cells. Focused particularly on Pembrolizumab, a prominent PD-1 inhibitor, the review
details its mechanism of action, which involves blocking the PD-1/PD-L1
interaction to restore T-cell activity against tumors. Pembrolizumab's efficacy
is highlighted through clinical trials in non-small cell lung cancer, melanoma,
and other cancers, demonstrating its broad-spectrum efficacy and safety
profile. Predictors of response, such as PD-L1 expression and tumor mutational
burden, are discussed alongside the associated immune-related adverse events
(irAEs) and their management. Future directions include refining patient
selection criteria, improving irAE management, and leveraging computational
algorithms for personalized therapy. Emerging research on fecal microbiota
transplantation (FMT) suggests the potential for enhancing ICI efficacy and managing side effects. Continued
innovation and research
are crucial for optimizing
ICI therapy and addressing existing
challenges to fully realize their potential in cancer treatment.
Keywords: Immune checkpoint inhibitors; Fecal microbiota transplantation; Autoimmunity
Introduction
The
advent of immune checkpoint inhibitors (ICIs) has significantly transformed the
treatment landscape of various cancers. This class of drugs, comprising PD-1
inhibitors (Nivolumab, Pembrolizumab, Cemiplimab), PD-L1 inhibitors
(Atezolizumab, Durvalumab, Avelumab), and CTLA-4 inhibitors (Ipilimumab), has
been approved by the US Food and Drug Administration (FDA) for several types of
cancer. ICIs have demonstrated remarkable efficacy in improving survival outcomes
for patients with advanced and metastatic cancers
by stimulating the immune
system to recognize and destroy cancer cells. This review explores the
mechanisms of action, clinical applications, predictors of response, side
effects, management strategies, and future directions of ICIs, with a
particular focus on Pembrolizumab, a notable PD-1 inhibitor1.
Mechanism of action
Immune Checkpoints and Their Role
Immune
checkpoints are essential components of the immune system that help maintain
self-tolerance and prevent
autoimmunity. These checkpoints are regulatory pathways
that modulate the immune response to ensure it is appropriately targeted and not overly aggressive. They primarily function
through interactions between immune checkpoint receptors on T-cells and their
corresponding ligands on antigen-presenting cells or tumor cells.
Two key immune checkpoint pathways relevant to cancer therapy
are the PD-1/PD-L1 and CTLA-4 pathways:
1. PD-1/PD-L1 Pathway:
· PD-1 (Programmed Cell Death Protein
1): A receptor
expressed on the surface
of T-cells.
· PD-L1 (Programmed Death-Ligand 1): A ligand that binds to PD-1, expressed on tumor cells and
antigen-presenting cells.
· PD-L2 (Programmed Death-Ligand 2): Another
ligand for PD-1,
found on some antigen-presenting cells.
Under normal circumstances, the interaction between
PD-1 and PD-L1/Pd-L2 acts as a brake on T-cell activity, reducing
immune responses and promoting tolerance. This mechanism helps prevent
autoimmunity by inhibiting excessive immune reactions against self-antigens.
2. CTLA-4 (Cytotoxic T-Lymphocyte Antigen
4):
· CTLA-4:
A receptor expressed on T-cells that competes with the costimulatory receptor CD28 for binding
to B7 molecules (CD80/CD86) on antigen-presenting
cells.
· CD80/CD86: Ligands
on antigen-presenting cells that provide
necessary signals for T-cell
activation.
CTLA-4 engagement with CD80/CD86 inhibits
T-cell activation and promotes T-cell exhaustion, contributing to immune
evasion by tumors2.
Mechanism of Action of Pembrolizumab
Pembrolizumab is a humanized monoclonal antibody that specifically targets
PD-1. The detailed mechanism of action is as
follows:
1. Binding to PD-1:
• Pembrolizumab binds with high affinity to PD-1 receptors on T-cells, thereby blocking the interaction between PD-1 and its ligands, PD-L1 and PD-L2 (Figure 1).
• This inhibition prevents the PD-1/PD-L1 and PD-1/PD-L2 interactions, which are crucial for downregulating T-cell responses.
2. Reactivation of T-Cells:
• By blocking PD-1, Pembrolizumab removes the inhibitory signals that would otherwise dampen T-cell activity.
• This reactivation of T-cells enhances their ability to recognize and attack tumor cells, effectively boosting the anti-tumor immune response.
3. Lack of Direct Cytotoxic Effects:
• Unlike cytotoxic antibodies that directly kill target cells through mechanisms like complement activation or engagement of Fc receptors, Pembrolizumab does not induce direct cell death.
• It operates through immunomodulation rather than cytotoxicity, altering the immune environment to enhance T-cell-mediated tumor destruction.
4. Effective Inhibitory Concentration:
· The 50% effective
inhibitory concentration (IC50) of Pembrolizumab in T-cell activation assays,
which measures its potency, ranges
from 0.1 to 0.3 nM. This
indicates its strong efficacy in blocking the PD-1 pathway and reactivating T-cells2
In
summary, Pembrolizumab functions by blocking the PD-1 receptor on T-cells,
thereby disrupting the inhibitory signals that tumors use to evade immune
surveillance. This action restores and enhances
T-cell activity against
cancer cells, making
it a powerful tool in cancer
immunotherapy.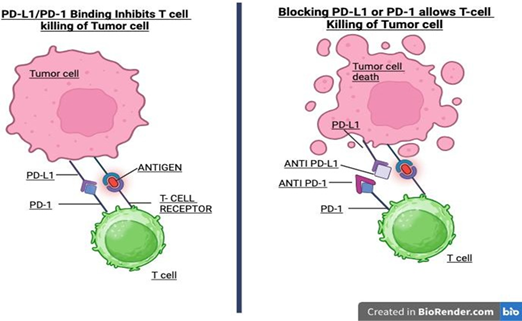
Figure
1: Mechanism of action of pembrolizumab
Clinical applications
Non-Small Cell Lung Cancer (NSCLC)
Survival Advantage and Clinical Trials: Pembrolizumab has significantly altered the treatment landscape for advanced NSCLC, particularly in patients with high PD-L1 expression. The KEYNOTE-024 trial, a landmark study, evaluated Pembrolizumab as a first-line treatment for patients with NSCLC and a PD-L1 tumor proportion score (TPS) of 50% or greater.
· KEYNOTE-024
Trial Findings: The trial demonstrated that
Pembrolizumab provided a substantial survival benefit compared to
platinum-based chemotherapy. Patients treated with Pembrolizumab had a median
overall survival (OS) of 20.0 months versus 12.2 months for those receiving chemotherapy. Furthermore, Pembrolizumab was associated with
a higher progression-free survival (PFS) rate, highlighting its effectiveness
as a first-line therapy3.
· Adverse
Events: The trial also revealed a favorable safety
profile for Pembrolizumab compared to chemotherapy. Pembrolizumab resulted in fewer severe treatment-related
adverse events (31.2%)
compared to chemotherapy (53.3%), underscoring its relative
safety advantage3.
Melanoma
The KEYNOTE-001 trial assessed Pembrolizumab, a PD-1 inhibitor, in advanced melanoma patients. It demonstrated:
• Sustained Effectiveness: Pembrolizumab provided durable tumor responses over five years, with high objective response rates in both treatment-naive and previously treated patients.
• Safety Profile: The drug maintained a favorable safety profile with manageable immune-related adverse events and comparable or improved quality of life outcomes.
· Clinical Implications:
Effective as both a first-line and subsequent treatment, Pembrolizumab showed long-term benefit
and flexibility in managing advanced melanoma4
Real-World Data: Real-world studies further supported the trial findings:
• Durability of Response: Pembrolizumab offered long-lasting responses similar to trial results, with benefits extending across various patient populations.
• Clinical Outcomes: Enhanced overall survival and improved quality of life in clinical practice were reported.
· Safety and Tolerability: Consistent with trial data,
the safety profile
was favorable, confirming
Pembrolizumab's role as a long-term therapeutic option5
Overall, both the KEYNOTE-001 trial and real-world evidence affirm Pembrolizumab as a highly effective and safe treatment for
advanced melanoma.
Pembrolizumab,
a leading PD-1 inhibitor, has proven highly effective across various cancers
beyond melanoma and non-small cell lung cancer. In head and neck squamous cell
carcinoma (HNSCC), Pembrolizumab has shown significant efficacy in both
recurrent and metastatic cases, as demonstrated by the KEYNOTE-012 and
KEYNOTE-040 trials, offering notable survival benefits, especially for patients
with high PD-L1 expression. For urothelial carcinoma, Pembrolizumab has
exhibited strong performance in both first-line and second-line treatments,
with trials such as KEYNOTE-052 and KEYNOTE-045 highlighting its impact on overall survival and progression-free survival. Similarly, in gastric cancer,
Pembrolizumab has shown promising
results in advanced stages, with trials like KEYNOTE-059 and KEYNOTE-061
confirming its durable responses and safety profile (Tab1e 1). Overall, Pembrolizumab's versatility across these
diverse cancers, coupled with its manageable safety profile, underscores
its significant role in modern cancer immunotherapy6.
Table 1: Clinical Applications of Pembrolizumab
|
Cancer Type |
Trial/Study |
Outcome |
Reference |
|
Non-Small Cell Lung Cancer (NSCLC) |
KEYNOTE-024 |
Improved survival compared to chemotherapy;
fewer severe adverse events. |
Reck et al., 2016 |
|
Melanoma |
KEYNOTE-001 |
Sustained tumor response and safety over five
years. |
Larkin et al., 2015 |
|
Head and Neck Squamous Cell Carcinoma |
- |
Demonstrated efficacy and safety. |
- |
|
Urothelial Carcinoma |
- |
Effective across various stages; ongoing
trials. |
- |
|
Gastric Cancer |
- |
Broad-spectrum efficacy; ongoing research on
survival benefits. |
- |
Side Effects Profile
While
immune checkpoint inhibitors (ICIs) like Pembrolizumab generally exhibit a
favorable safety profile compared to traditional chemotherapy, they are not
without their own set of adverse effects. Pembrolizumab is associated with a variety
of immune-related adverse
events (irAEs) due to its mechanism of enhancing immune system activity
against cancer cells.
Common
irAEs include dermatologic reactions such as lichenoid reactions and eczema,
gastrointestinal issues like colitis and hepatitis, and endocrine disorders,
including thyroiditis and adrenalitis. A meta-analysis comparing Pembrolizumab
with chemotherapy found no significant difference in the risk of fatal adverse
events (FAEs) between the two treatments. However, certain adverse events,
such as infections and pneumonitis, are particularly notable due to their potential severity10,11.
Severe
hematologic adverse effects, although relatively rare, have also been
documented with Pembrolizumab. These include autoimmune anemia, such as
autoimmune hemolytic anemia (AIHA), and immune thrombocytopenia, which can lead
to significant health concerns. Case reports have highlighted instances of
pancytopenia, a condition characterized by reduced levels of red blood cells,
white blood cells, and platelets, associated with Pembrolizumab treatment12,13. Furthermore, clinical trials have
frequently observed adverse events like neutropenia and anemia, which require
careful monitoring and management14,15.
Thus, while Pembrolizumab offers substantial therapeutic benefits, its
associated adverse effects underscore the importance of ongoing vigilance and proactive management strategies to ensure patient safety and optimize
treatment outcomes (Table 2).
Table 2: Side Effects Profile of Pembrolizumab
|
Management of side effects
The management of ICIs involves
early recognition and prompt intervention of immune-related
adverse events (irAEs). Standard management strategies include corticosteroids
and other immunosuppressive agents for severe irAEs. Hematologic complications
may require specific treatments such as transfusions, steroids, or
immunoglobulins, depending on the severity and type of adverse event16.
Early Recognition and Monitoring
The management of ICIs involves
early recognition and prompt intervention of immune-related
adverse events (irAEs). Standard management strategies include corticosteroids
and other immunosuppressive agents for severe irAEs. Hematologic complications
may require specific treatments such as transfusions, steroids, or
immunoglobulins, depending on the severity and type of adverse event16.
· Early
Detection: Early recognition of irAEs is crucial
for effective management. Regular monitoring, including clinical assessments,
laboratory tests, and imaging studies, aids in identifying irAEs at an early stage. Patients should be educated
about the potential
side effects of ICIs and
encouraged to report any new symptoms promptly.
· Tapering
and Discontinuation: Once the irAE is under
control, corticosteroids should be gradually
tapered to avoid rebound inflammation. The tapering schedule
depends on the initial
dose and duration of corticosteroid therapy16.
· Patient
Education: Patients should be informed about the
signs and symptoms of common irAEs. Educating patients
on the importance of early reporting can lead to timely intervention and prevent the progression
of irAEs.
General Management Strategies
· Mild irAEs: Managed with supportive care and, if necessary, low-dose corticosteroids.
· Moderate irAEs: Often require higher doses of corticosteroids (e.g., prednisone 0.5-1 mg/kg/day).
· Moderate irAEs: Often require higher doses of corticosteroids (e.g., prednisone 0.5-1 mg/kg/day).
Immunosuppressive Agents: In cases where corticosteroids are ineffective or not well-tolerated, additional immunosuppressive agents such as infliximab, mycophenolate mofetil, or tacrolimus may be used. The choice of agent depends on the specific irAE and the patient’s overall health status
Future directions
The field of ICIs is rapidly evolving, with ongoing research focusing on optimizing treatment outcomes and mitigating adverse effects. Key future directions include refining patient selection criteria, improving management of side effects, identifying predictive biomarkers, and advancing computational algorithms and personalized medicine.
Refining Patient Selection Criteria
Future research aims to enhance patient selection criteria to improve ICI efficacy and safety. Current strategies involve identifying patients most likely to benefit from ICIs based on tumor and patient characteristics. Research is increasingly focused on tumor mutational burden (TMB) and microsatellite instability (MSI) as predictive biomarkers for ICI response17. High TMB and MSI are associated with increased neoantigen load, which may enhance the likelihood of a positive response to ICIs. Advanced imaging technologies and molecular profiling are being explored to identify suitable candidates for ICI therapy more precisely18.
Improving Management of Side Effects
Managing irAEs remains critical to optimizing ICI therapy. Future research aims to develop more effective strategies for early detection and management of irAEs. Understanding the pathophysiology of irAEs could lead to targeted interventions and preventive measures19. For example, biomarker-driven approaches to predict and prevent severe irAEs are being actively pursued20. Developing standardized treatment protocols for managing common irAEs, such as pneumonitis and colitis, is essential for improving patient outcomes21.
Identifying Predictive Biomarkers
Efforts are focused on discovering and validating biomarkers that can accurately predict response and adverse effects. While PD-L1 expression is a well-known biomarker, its predictive value can vary. Research is underway to identify additional biomarkers, such as circulating tumor DNA (ctDNA) and immune cell profiling, that may provide a more comprehensive understanding of ICI efficacy and safety22,23.
Advances in computational algorithms and personalized medicine
Advances in computational algorithms and personalized medicine are expected to enhance the precision of ICI therapy. Computational models and machine learning techniques are being developed to integrate diverse data sources, including genomic, proteomic, and clinical data, to predict treatment outcomes and optimize patient management24. Personalized approaches are also being explored to tailor ICI therapy based on individual genetic and molecular profiles25. These approaches include developing algorithms to predict the most effective therapies and guiding the development of combination therapies to enhance ICI efficacy and reduce resistance26.
Fecal Microbiota Transplantation
Fecal microbiota transplantation (FMT) is an emerging area of interest in ICI therapy. Recent studies suggest that gut microbiota may influence the efficacy of ICIs and the occurrence of irAEs. FMT, which involves transferring fecal material from a healthy donor to the patient, aims to restore a healthy gut microbiota (Table 3). Preliminary studies suggest that FMT may enhance the efficacy of ICI therapy and mitigate some adverse effects27. Ongoing research is investigating how gut microbiota influence ICI outcomes and the potential benefits of FMT28.
Table 3: Future Directions in ICI Therapy
|
Area of Focus |
Description |
Reference |
|
Refining Patient Selection |
Enhancing criteria based on TMB and MSI,
advanced imaging, and molecular profiling. |
Rizvi & Hellmann, 2015 |
|
Improving Management of Side Effects |
Developing effective strategies for early
detection and management of irAEs. |
Dougan & Shulman, 2020 |
|
Identifying Predictive Biomarkers |
Discovering biomarkers like ctDNA and immune
cell profiling to predict responses. |
Cohen & Chung, 2021 |
|
Advances in Computational Algorithms |
Integrating diverse data sources for
predicting outcomes and personalizing therapy. |
Lee & Lee, 2021 |
|
Fecal Microbiota Transplantation |
Investigating FMT to enhance efficacy and
manage irAEs. |
Mazzoni & D'Alessio, 2020 |
Conclusion
The emergence of immune checkpoint inhibitors (ICIs) has revolutionized the management of various cancers, offering new hope through enhanced survival rates and durable responses. Pembrolizumab, a prominent PD-1 inhibitor, exemplifies this transformation with its proven efficacy across multiple malignancies, including non-small cell lung cancer, melanoma, and gastric cancer. Its ability to restore immune surveillance by blocking the PD-1/PD-L1 interaction underscores a pivotal shift from traditional cytotoxic therapies to immunotherapy, leveraging the body's immune system to target and destroy cancer cells.
Despite their significant benefits, ICIs are not without challenges. Pembrolizumab, like other ICIs, is associated with a range of immune-related adverse events (irAEs), such as dermatologic, gastrointestinal, and hematologic issues. While these side effects are generally manageable, their early detection and prompt management are critical to optimizing patient outcomes. The use of corticosteroids remains the cornerstone of treatment for severe irAEs, but the development of more targeted and personalized management strategies is essential.
Looking forward, the field of ICI therapy is poised for substantial advancements. Enhancing patient selection through refined biomarkers, such as tumor mutational burden and microsatellite instability, will likely improve treatment precision and efficacy. Additionally, ongoing research into predictive biomarkers, including circulating tumor DNA and immune cell profiling, holds promise for more accurately forecasting patient responses and adverse effects.
The integration of advanced computational algorithms and personalized medicine approaches represents another exciting frontier. By harnessing machine learning and genomic data, future strategies aim to tailor treatments more precisely, predict responses, and mitigate resistance. Furthermore, novel approaches such as fecal microbiota transplantation offer intriguing possibilities for enhancing ICI efficacy and managing irAEs, potentially paving the way for more effective and personalized cancer therapies.
In summary, while Pembrolizumab and other ICIs have ushered in a new era of cancer treatment, continued research and innovation are crucial to overcoming current limitations and fully realizing their potential. The future of ICI therapy will depend on our ability to refine patient selection, manage adverse effects effectively, and leverage cutting-edge technologies to deliver personalized and optimized care.
References
1. Kwok G, Yau TC, Chiu JW, Tse E, Kwong YL. Pembrolizumab (Keytruda). Human Vaccines Immunotherapeutics 2016;12(11):2777-2789.
2. Reck M, Rodríguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Eng J Medicine 2016;375(19):1823-1833.
3. Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Eng J Med 2015;373(23):23-34.
4. Topalian SL, Sznol M, McDermott DF, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. Lancet Oncol 2012;16(4):435-445.
5. Hanna K, Ruttan T, Makarov V, Patil S. Pembrolizumab in advanced melanoma: Real-world experience. J Clin Oncol 2017;35:e19107-e19107.
6. Fehrenbacher L, Spira A, Ballinger M, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (POPLAR): A multicentre, open-label, phase 2 randomised controlled trial. Lancet 2017;389(10066):255-265.
7. Patel SP, Kurzrock R. PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther 2015;14(4):847-856.
8. Sacher AG, Gandara DR, Felip E. Tumor mutation burden and response to immune checkpoint inhibitors. J Clinical Oncol 2016;34:e17552-e17552.
9. Gopalakrishnan V, Spencer CN, Nezi L, Helmink BA. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 2018;359(6371):97-103.
10. Johnson DB, Sullivan RJ, Makhoul I. Immune checkpoint inhibitors in the treatment of melanoma: Safety and efficacy. J Clin Oncol 2016;34: e18153-e18153.
11. Motzer RJ, Escudier B, McDermott DF, et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Eng J Med 2015;373(19).
12. Wang L, Shi Y. Autoimmune hemolytic anemia induced by Pembrolizumab. J Hematol Oncol 2020;13(1):8.
13. Valpione S, Kotecha R. Immune-related thrombocytopenia with Pembrolizumab. Cancer Immunol Immunoth 2020;69(10):1927-1935.
14. Marabelle A, Le DT, Ascierto PA. Efficacy of Pembrolizumab in patients with microsatellite instability-high colorectal cancer: Updated results from the KEYNOTE-164 trial. J Clin Oncol 2020;38:4508.
15. Atkinson R, Shaw JW, Alarcon T. Management of anemia and neutropenia in cancer patients receiving Pembrolizumab. Oncol Nursing Forum 2019;46(2):127-135.
16. Haanen JB, Carbonnel F. Management of adverse events following treatment with anti-PD-1/PD-L1 therapy. Ann Oncol 2018;29.
17. Rizvi NA, Hellmann MD. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 2015;348(6230):124-128.
18. Yang Y, Xu J. Advances in imaging technologies for predicting and monitoring immune checkpoint inhibitor therapy. Current Oncology Rep 2020;22(11):106.
19. Dougan M, Shulman H. Management of immune-related adverse events from immune checkpoint inhibitors. Current Opinion Oncol 2020;32(5):632-638.
20. Wang H, Zhang H. Predictive biomarkers for immune checkpoint inhibitor therapy: A systematic review and meta-analysis. Front Oncol 2021;11:736738.
21. Cortellini A, Bersanelli M. Management of immune-related adverse events from immune checkpoint inhibitors: A review of current guidelines. Expert Rev Anticancer Ther 2019;19(9):761-774.
22. Cohen JV, Chung CH. Circulating tumor DNA and immune checkpoint inhibitors: A promising combination for precision oncology. Clinical Cancer Res 2021;27(7):1780-1788.
23. Sato T, Hoshida Y. Advances in immune cell profiling for predictive biomarkers of immune checkpoint inhibitor therapy. Immuno-Oncology Technol 2022;9:100074.
24. Lee MJ, Lee H. Machine learning algorithms for predicting immune checkpoint inhibitor responses. Cancer Immunol Res 2021;9(8):848-859.
25. Saad A, Moin D. Personalized medicine in immune checkpoint inhibitor therapy: Current progress and future directions. Molecular Med 2022;28(1):64.
26. Yaguchi T, Nishio M. Advances in combination therapies involving immune checkpoint inhibitors. Front Oncol 2021;11:644406.
27. Mazzoni A, D'Alessio S. Fecal microbiota transplantation in cancer immunotherapy: A new approach to enhance efficacy and mitigate adverse effects. Gastroenterology Res Practice 2020.
28. Gopalakrishnan V, Spencer CN. The impact of fecal microbiota transplantation on immune checkpoint inhibitors: Evidence and future perspectives. J Immunother 2021;44(2):46-54.