Anaplastic Thyroid Carcinoma with Retained TTF-1 Expression
Abstract
Anaplastic
thyroid carcinoma (ATC) is a rare, highly aggressive malignant neoplasm,
accounting for only 2-3% of all thyroid gland tumors. We present a 78 year-old
female with a past medical history of papillary thyroid carcinoma, diagnosed in
2011. She received a total thyroidectomy and post-operative radioactive iodine
with ablation of remnant right-sided thyroid tissue. In June 2023, she
presented to the emergency department for right-sided neck pain and painful
swallowing. She also reported an unintentional weight loss of 25 lbs. Upon
physical examination, an immobile right sided neck mass was appreciated between
the sternocleidomastoid and the head of the clavicle. Computerized tomography
(CT) demonstrated a new 2.8 cm mass to the right thyroid bed. Tissue
examination showed evidence of anaplastic thyroid carcinoma.
Immunohistochemical staining demonstrated TTF-1 positivity. The hallmark of ATC
on histopathology is a highly infiltrative neoplasm, with invasion to vascular
and adjacent tissues. In approximately 75% of cases, PAX8 is positive and TTF-1
markers are usually negative. Thyroid transcription factor-1 or TTF-1 has a
38kDa homeodomain containing transcription factor with 371 amino acids. TTF-1
is essential for thyroid morphogenesis and gene regulation and is believed to
play a crucial role in oncogenic and inhibitory activities in cancer
development. Pathological uses related to TTF-1 include serving as a tumor
marker for primary and metastatic thyroid cancers. However, in ATC, TTF-1
expression is usually completely lost. Our objective is to clarify this
distinctive presentation of a rare neoplasm, fostering enhanced management and
advocacy for patients affected by this malignancy.
Keywords: Anaplastic
thyroid carcinoma; Thyroid transcription factor; Computerized tomography
Introduction
Anaplastic thyroid carcinoma (ATC)
accounts for approximately 1.3 to 9.8% of all thyroid cancers globally. The
age-adjusted annual incidence of ATC is 1 to 2 cases per 1 million. At
diagnosis, patients are usually older than 65 years of age, with 60-70% of
cases affecting female patients1.
Unlike follicular thyroid carcinoma which is the most common thyroid malignancy
and holds the best overall prognosis, ATC has an extremely guarded prognosis2. In some cases, ATC can arise as a result of
anaplastic transformation of differentiated papillary, follicular, or Hurthle
cell thyroid carcinoma3. We present a
78-year-old female with anaplastic thyroid carcinoma with retained TTF-1
expression.
Case presentation
This is a 78-year-old
female with a history of coronary artery disease (CAD), congestive heart failure (CHF), diabetes mellitus
type 2 (DMII), hypertension (HTN), myocardial infarction (MI) and deep venous thrombosis (DVT). Her medications include
Eliquis, Plavix, and nonsteroidal
anti-inflammatory drugs (NSAID) as needed. Past medical history includes papillary
thyroid carcinoma in 2011,
for which she received a total thyroidectomy and post-operative
radioactive iodine with ablation of remnant right-sided thyroid tissue. In
2014, a follow up 1-131 scan demonstrated that this thyroid
remnant was successfully ablated. Surgical history includes a coronary artery stent
placement, left knee arthroplasty, bilateral hip arthroplasty, thyroidectomy, transverse colon resection, and
salivary gland extension. Family history
includes breast cancer in her sister diagnosed in the 5th decade of life. She
is a former smoker, quitting over 20 years ago, she does not
drink alcohol or use illicit drugs.
In October
2022, she noticed a suspicious lump to the midline of the neck. At this time, she received a computerized tomography
(CT) of the neck, which demonstrated a 1.8 cm mass in the left anterior lower neck. In March 2023, she was seen by
ENT, biopsies of this mass were performed
and demonstrated papillary thyroid carcinoma.In May 2023, she received a
transverse colon resection
for a tubulovillous adenoma and incisional hernia repair; her postoperative course was uncomplicated.
Table 1. Patient’s Abnormal Lab Values;
All other laboratory tests were within
normal limits.
|
Lab Ordered: |
Patient’s Value: |
Reference Value: |
|
Potassium |
3.2 mmol/L |
3.5-5.3 mmol/L |
|
Free T4 |
1.62 ng/dL |
0.8 - 1.8 ng/dL |
|
TSH |
0.375 mclU/mL |
0.45 - 5.33 mclU/mL |
|
Hemoglobin |
10.6 g/dL |
12.0 - 16.0 g/dL |
|
Hematocrit |
33.5% |
35.0 - 46.0% |
|
Platelets |
49.2% |
12.0 - 15.0% |
|
Eosinophils Relative |
10.8 cells/mcL |
30 - 350 cells/mcL |
|
Eosinophils Absolute |
.80 K/mcL |
0.00-0.50 K/mcL |
|
Magnesium |
1.6 mg/dL |
1.7 - 2.5 mg/dL |
In June 2023 she presented to the emergency department (ED) for right-sided neck pain radiating to the left shoulder and headache. She also reported an unintentional weight loss of 25 lbs and painful swallowing. Upon physical examination, an immobile right sided neck mass was appreciated between the sternocleidomastoid muscle and the head of the clavicle. Abnormal laboratory results at this time are demonstrated in (Table 1). At this time, CT of the neck demonstrated a new 2.8 cm mass to the right thyroid bed with inflammatory changes. Core needle biopsies were taken for histopathological examination. Pathology reviewed the specimen, determining that anaplastic thyroid carcinoma was present. Invasive carcinoma with marked cytologic atypia with squamoid features were noted, as demonstrated in (Figure 1). In comparison to the prior tissue exam in March 2023, this specimen did not have well differentiated characteristics or have features of papillary thyroid carcinoma. Immunohistochemical staining was also performed and demonstrated TTF-1 and PAX-8 positivity; (Figure 2) demonstrates IHC results. Our patient was seen by hematology-oncology after surgical resection was determined not appropriate. She was placed on Taxol in combination with radiotherapy.
Figure 1. Anaplastic Thyroid Carcinoma, High Power
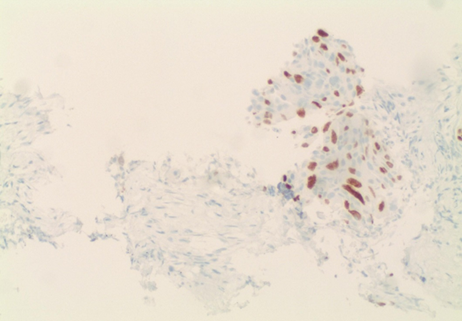
Figure 2. Immunohistochemical Stain, TTF-1 Positivity
Discussion
Anaplastic thyroid carcinoma, also referred to as undifferentiated thyroid carcinoma, typically occurs in elderly patients, peaking around the seventh decade of life. This neoplasm rarely occurs in patients younger than 50 years old and has a female predominance with a 4:1 female to male ratio. The location of anaplastic thyroid carcinoma is in the thyroid gland, often paired with its extension into surrounding structures and tissues. Patients usually present with a rapidly enlarging mass, associated with dyspnea, dysphagia, and hoarseness4. Most anaplastic carcinomas occur from well-differentiated thyroid carcinomas, most frequently from papillary and follicular thyroid carcinoma3.
The hallmark of ATC on histopathology is a highly infiltrative neoplasm, with invasion to vascular and adjacent tissues. Common histological features of ATC include three distinct patterns: squamoid, spindle cell, and giant cell. These patterns have been seen to coexist within the same tumor. This neoplasm typically does not show lobular growth patterns, nor lymphoepithelial appearance. Marked nuclear pleomorphism, increased mitotic rates, and extensive tumor necrosis are usually present. Such cellular changes are accompanied by infiltration of inflammatory cells, specifically neutrophils. In the squamoid form of ATC, squamous markers such as p40, p63, and CK5/6 are present. In approximately 75% of cases, PAX8 is positive, however TTF-1, thyroglobulin, c-kit, and CD5 markers are usually negative.
Treatment for ATC includes complete surgical resection, paired with adjuvant radiotherapy and chemotherapy. The prognosis for this condition is extremely poor and fatality is usually related to airway obstruction or widespread metastases4.
Thyroid transcription factor-1 or TTF-1, also referred to as NKX2-1 and thyroid specific enhancer binding protein. TTF-1 has 38kDa homeodomain containing transcription factor with 371 amino acids. It is encoded by a gene located on chromosome 14q13 and is a member of the NKX2 gene family. It is expressed in thyroid, lung, and brain tissues and is associated with structures from diencephalic origin. TTF-1 is essential for thyroid morphogenesis and gene regulation and is believed to play a crucial role in oncogenic and inhibitory activities in cancer development and progression. Pathological uses related to TTF-1 include serving as a tumor marker for primary and metastatic thyroid cancers. However, in ATC, TTF-1 expression is usually completely lost. TTF-1 stains are usually positive in papillary, follicular and medullary thyroid carcinomas, as well as follicular adenomas5.
Treatment options for ATC usually include surgery, radiotherapy, chemotherapy, and immunotherapy. Typically for patients with localized ATC, surgical treatment is appropriate. However, given the rapid progressive nature of this malignancy, radiotherapy and systemic therapy are also recommended after any surgical interventions6. Patients with ATC without targeted gene mutations, chemoradiotherapy is recommended; combinations usually include paclitaxel, doxorubicin, or combined regimens7. Recent studies have shown promising results when treating ATC with combined radiotherapy and immunotherapy. Since PDL1 expression in tumor cells has been shown to increase after radiotherapy, immunotherapy can be used to combat radiation resistance. With the release of immune stimulators after radiation, the tumor can become immunogenic, leading to enhancement of the effects of immunotherapy. One example of this synergistic antitumor effect with these therapies is pembrolizumab8.
Conclusion
Conflicts of interest
We do not have any conflict of interest.
References