Brain Abscess Secondary to Visceral AVMS in Hereditary Haemorrhagic Telangiectasia
Abstract
Hereditary Hemorrhagic
Telangiectasia (HHT) is an autosomal dominant disorder associated with various
neurological complications arising from vascular malformations.
We present a case of a
49-year-old female who presented with focal seizures. Imaging revealed a brain
abscess. Further investigation, prompted by a history of recurrent epistaxis,
identified multiple pulmonary and liver arteriovenous malformations (AVMs). The
brain lesion was confirmed to be an abscess and the patient was subsequently
diagnosed with HHT based on clinical criteria and family history.
This case highlights
the importance for a high index of suspicion for HHT in patients presenting
with recurrent epistaxis and evidence of systemic AVMs. Prompt diagnosis and
screening for visceral AVMs, particularly PAVMs, is essential for preventing
serious neurological complications like cerebral abscesses thus significantly
improving patient outcomes in this multisystemic disorder.
Keywords: Hereditary hemorrhagic telangiectasia; Vascular
malformations; Arteriovenous malformations
Introduction
Hereditary haemorrhagic
telangiectasia (HHT) is an autosomal dominant condition that is linked to a
myriad of neurologic complications arising from vascular malformations of the
brain, spinal cord and lungs. We present a 49-year-old female who came to our
hospital outpatient department with history of seizures and was found to have a
brain abscess stemming from a pulmonary arteriovenous malformation (PAVM).
PAVMs are associated with intracranial abscesses due to shunting and loss of
the normal filtering effects of the lung capillary bed, predisposing patients
with PAVMs to cerebral abscess. It is because of this association that it is
imperative to treat the AVMs in patients found to have brain abscesses. In our
case, embolization of pulmonary AVMs was done to minimize the risk of
reinfection. PAVMs are often associated with HHT.
Case Presentation
A
49-year old female presented with history of 2 episodes of left sided focal
seizures with secondary generalization that occurred 2 days ago. On examination
we found right hemiparesis with predominant lower limb weakness, truncal ataxia
and subtle right upper limb pronator drift which remained for a few hours. No
facial or speech involvement was seen. She also gave history of repeated
episodes of spontaneous intermittent, painless epistaxis since many years but
denied history of haemoptysis.
On
physical examination, she was hemodynamically stable. Initial investigations
were unremarkable except for a white cell count of 15,000/μL. Renal and liver
function tests were within normal limits. Blood culture was sent, which did not
show any growth till 3rd day of incubation. A Magnetic Resonance Imaging (MRI)
of the brain with contrast was performed in view of seizures. This showed a
ring enhancing lesion in the left paramedian post central gyrus with
surrounding perilesional edema (Figure 1a-d). The
central necrotic part showed restricted diffusion without any enhancement.
Possibilities of Tuberculoma and cerebral abscess were suggested. She was asked
to take antiepileptic drugs– Levetiracetam 500 mg twice a day and was started
on intravenous ceftriaxone 2 gm twice a day.
Figure 1: (A) - T2W coronal image
showing a hyperintense lesion in the left paramedian post central gyrus
with surrounding perilesional
edema.
(B) & (C) - ADC image and
its trace map showing restricted diffusion in the involved area.
(D) - Post contrast Axial
image showing peripherally enhancing lesion.
A
previous chest radiograph done a week back showed large subpleural oval shaped
opacity in the right lower lobe. She underwent Positron Emission Tomography
(PET – CT ) scan in view of the suspicion of this being a neoplastic lesion.
On PET
CT, the right lung showed weakly metabolic enhancing soft tissue lesion in the lower
lobe measuring 33 x 25 mm and 23 x 18 mm & an enhancing pleural based
lesion measuring 36 x 29 mm in the right middle lobe. The brain lesion was
ametabolic, confirming it to be an abscess (Figure 2).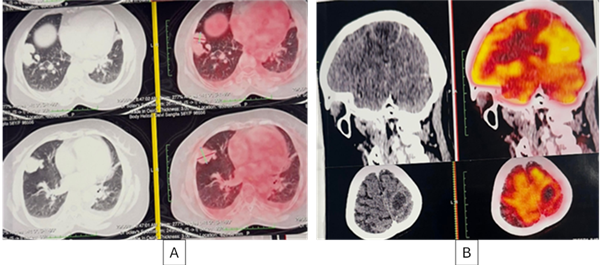
Figure 2: (A)- Axial PET CT image shows weakly metabolic
enhancing soft tissue lesion in right lung lower lobe.
(B) -
Brain lesion appears ametabolic, confirming it to be an abscess
A Computed Tomography (CT)
Pulmonary angiogram (Figure 3) was suggested which confirmed the presence of 4 well defined
pulmonary arteriovenous malformations, three in the right lung, of which 2 were
in the lower lobe largest measuring 5.6 x 2.2 cm and one in the middle lobe
measuring 2.3 x 2.5 cm and one in the left lung. All AVMs were having single
arterial feeders (fistulous point). Two liver AVMs were also identified one
each in the right and left lobes.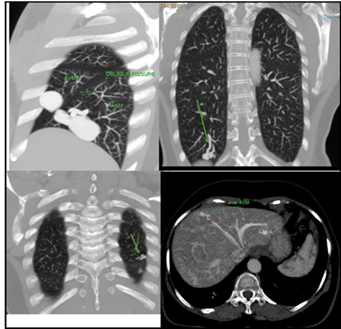
Figure 3: (A):- Reconstructed CT
Sagittal image shows two
pulmonary AVMs in the right lung; one in lower lobe and other in middle lobe.
(B):- Reconstructed CT
coronal image shows third pulmonary AVM in the lower lobe of right lung.
(C):- Reconstructed CT
coronal image shows fourth pulmonary AVM in the lower lobe of left lung.
(D):- Reconstructed CT axial
image shows a hepatic AVM in the right lobe of the liver.
Conservative management of the
brain abscess was planned with a 6-week course of ceftriaxone. The liver AVM
was left untreated as the patient had no abdominal symptoms.
Endovascular embolization of the
pulmonary AVMs was performed under general anaesthesia via a transfemoral
approach.
Follow up MRI brain after a month
showed reduction in size of the abscess.
In view of recurrent epistaxis,
multiple pulmonary AVMs, liver AVMs, she was diagnosed as a case of HHT as her
sister and another relative also had recurrent painless spontaneous epistaxis.
Discussion
This case report
highlights the importance of recognizing Hereditary Haemorrhagic Telangiectasia
(HHT) in patients presenting with seemingly isolated symptoms, even in
resource-limited settings where comprehensive genetic testing may not be
immediately feasible.
Our patient's
history of spontaneous and recurrent epistaxis since childhood, also seen in
her sibling, strongly suggested a syndromic diagnosis. The subsequent
identification of arteriovenous malformations (AVMs) in both the liver and
lungs further suggested a diagnosis of HHT.
A good clinical and
family history, coupled with targeted imaging, can be pivotal in diagnosing HHT
and guiding management.
HHT, also known as
Osler-Weber-Rendu disease, is an autosomal dominant multisystem vascular
disease with a prevalence of 1:5,000 to 1:10,000, which is associated with a
myriad of primary and secondary neurologic complications1,2. The most common
clinical manifestation is spontaneous and recurrent epistaxis beginning on an
average at age 12 years of age3, however the true
burden of HHT lies in its potential for severe, often life-threatening,
visceral complications. A diagnosis of HHT can be made by application of the
Curaçao criteria2 (Table1).
Table 1: Summary of Highlighted
Diagnostic and Surveillance Recommendations in Hereditary Hemorrhagic
Telengiectasia2
|
Consideration |
Recommendation |
|
Diagnostic |
• An underlying diagnosis of HHT should be considered in all persons
with a PAVM. • An underlying diagnosis of HHT should be considered in persons
meeting 2 or more Curaçao criteria' (1) Spontaneous and recurrent epistaxis; (2) Multiple mucocutaneous telangiectasias of the lips oral cavity,
fingers or nose; (3) Visceral lesions (gastrointestinal telangiectasias or
pulmonary/hepatic/cerebral/spinal AVMs); (4) A first-degree relative with definite HHT. |
|
Targeted screening |
• PAVM screening should be performed at the time of HTT diagnosis
(adult/pediatric patients) and repeated at 5-y intervals (pediatric patients
only)." • All adults with possible or definite HHT should receive MRI
screening for brain vascular malformations at the time of diagnosis. • Spinal arteriovenous malformations are not routinely screened. |
Abbreviations:
HHT = hereditary hemorrhagic telangiectasia; PAVM = pulmonary arteriovenous
malformation.
HHT is
considered possible or suspected with 2 criteria presents, definite with 3 or
more positive criteria1.
Primary
neurological complications in HHT are predominantly secondary to cerebral AVMs
(CAVMs), which affect nearly a quarter of individuals with HHT4. Given their
prevalence and potential for catastrophic outcomes such as hemorrhage or
seizures, expert guidelines unequivocally recommend routine brain MRI screening
for all individuals with suspected or definite HHT. In contrast, spinal AVMs
are considerably rarer and thus not routinely looked for5.
PAVMs found in
approximately 50% of HHT patients, represent direct shunts between pulmonary
arteries and veins, bypassing the crucial capillary bed filtration and gas
exchange6. This right-to-left shunt mechanism directly facilitates the
paradoxical embolism of septic micro-emboli, bypassing the lungs' natural
filtering capacity and allowing them direct access to the cerebral circulation.
Consequently,
the risk of developing a cerebral abscess is significantly elevated in HHT
patients with PAVMs, with an incidence as high as 6%6.
While the
causative organism in our patient's cerebral abscess could not be definitively
identified, the strong association between PAVMs and cerebral abscesses in HHT
entailed prompt intervention.
The management
strategy in such cases is therefore two-fold: addressing the acute neurological
insult and mitigating the future risk of recurrence. Embolization of the
identified pulmonary AVMs in our patient was performed to minimize the risk of
subsequent septic emboli and reinfection.
This case
further reinforces a crucial diagnostic paradigm: the identification of PAVMs,
even in isolation, should prompt a comprehensive evaluation for HHT. Given that
nearly all PAVMs are attributable to HHT, screening for HHT is warranted in any
patient found to have PAVMs, irrespective of other overt clinical
manifestations7. The recommended first-line screening for PAVMs in patients with
possible or confirmed HHT is transthoracic contrast echocardiography, with
pediatric patients requiring repeat screening every five years2.
Conclusion
This case
highlights the need for a high index of suspicion for HHT in patients
presenting with recurrent epistaxis, particularly when combined with evidence
of systemic AVMs. Early diagnosis and screening for visceral AVMs, especially
PAVMs, is important to prevent debilitating neurological complications like
cerebral abscesses. Even in resource-constrained environments, a thorough
clinical assessment can guide appropriate investigations and interventions,
significantly improve patient outcomes and prevent life-threatening events
associated with this multisystemic disorder.
References
2. Shovlin
CL, Guttmacher AE, Buscarini E, et al. Diagnostic
criteria for hereditary hemorrhagic telangiectasia (Rendu-Osler-Weber
syndrome). Am J Med Genet 2000;91(1):66-67.
3. Faughnan ME, Mager JJ, Hetts SW, et al. Second International Guidelines for the Diagnosis and Management of
Hereditary Hemorrhagic Telangiectasia. Ann Intern Med 2020 Dec
15;173(12):989–1001.
4. Fulbright
RK, Chaloupka JC, Putman CM, et al. MR of hereditary hemorrhagic
telangiectasia: prevalence and spectrum of cerebrovascular malformations. AJNR
Am J Neuroradiol 1998;19(3):477-484.
5. McDonald
J, Bayrak-Toydemir P, Pyeritz RE. Hereditary hemorrhagic telangiectasia: an
overview of diagnosis, management and pathogenesis. Genet Med Off J Am Coll Med
Genet 2011;13(7):607-616.
6. AAssar
OS, Friedman CM, White RI. The natural history of epistaxis in hereditary
hemorrhagic telangiectasia. The Laryngoscope 1991;101(9):977-980.
7. Shovlin
CL, Condliffe R, Donaldson JW, Kiely DG, Wort SJ, British Thoracic Society.
British Thoracic Society Clinical Statement on Pulmonary Arteriovenous
Malformations. Thorax 2017;72(12):1154-1163.