Candida Albicans Candidemia In The Intensive Care Setting: Epidemiological Profile, Risk Factors And Antifungal Susceptibility
Abstract
Introduction
Candidemia, a common invasive fungal infection in hospital settings, remains associated with high mortality despite medical advancements. Candida albicans is the predominant species in intensive care units, promoted by several risk factors such as prolonged hospitalization, immunosuppression and the use of broad-spectrum antibiotics. Early diagnosis and appropriate antifungal treatment are crucial. This study aims to describe the epidemiological profile of Candida albicans candidemia in the intensive care unit, identify associated risk factors and assess susceptibility to commonly used antifungal agents.
Materials and methods
This is a retrospective descriptive
cross-sectional study conducted over a 5-year period from January 2020 to
January 2024, involving 480 blood culture samples from the intensive care units
of the Hassan II University Hospital in Fez, whose mycological analysis (direct
examination, culture and identification) revealed the presence of Candida
Albicans. All patients admitted to the intensive care unit for a medical or
surgical pathology, hospitalized for more than 48 hours and presenting a
clinical or biological infectious syndrome or clinical signs suggestive of a
Candida sp infection and having a risk factor for the development of a fungal
infection were included in the study.
Antifungal susceptibility was then tested by MIC measurement using 4 antifungal agents with the ETEST technique (fluconazole, flucytosine, voriconazole, caspofungin).
Results
During the study period, 480 blood culture
requests were sent out, 93 of which were positive; 29 patients were diagnosed
with Candida Albicans candidemia, a percentage of 31.18%. The average age of
the patients was 28 years, with extremes ranging from 4 months to 73 years. A
slight male predominance was noted, with a M/F sex ratio of 1.07.
Sensitivity to antifungal agents revealed resistance to fluconazole and flucytosine and particularly to voriconazole, with a 72% resistance rate, compared with only 18% sensitivity. Caspofungin, on the other hand, showed a better balance between sensitivity and resistance, with almost half the strains being sensitive, i.e. a rate of 45.45%.
Discussion and conclusion
Candidemia is an invasive form of
candidiasis. Most cases occur in intensive care units, where the incidence is
generally higher than in other departments. They rank fourth among
healthcare-associated infections (HCAI) (10-15%) and have been on the rise in
recent years with the emergence of non-albicans Candida.
Although Candida Albicans remains a
predominant cause of candidemia in intensive care units, the increasing
prevalence of non-Albicans species and associated resistance patterns pose
significant challenges. Continuous monitoring of epidemiological trends and
patient-specific factors is essential to optimize treatment and improve
outcomes in critically ill patients.
Keywords: Antifungus; Candidemia; Candida albicans; Risk factor
Introduction
Candidemia
has become an important cause of bloodstream infections worldwide, particularly
in vulnerable populations such as those with hematological malignancies,
pediatric patients and the critically ill. The prevalence of candidemia varies
according to region and patient demographics, with a notable increase in
species other than Albicans. Understanding the epidemiology, species
distribution and antifungal resistance patterns is essential for effective
management and treatment1. Candidemia
is a leading cause of nosocomial bloodstream infections, with mortality rates
ranging from 5% to over 80% in pediatric populations1.
The prevalence of candidemia in intensive care unit (ICU) patients is higher than in other hospital wards. Early diagnosis of candidemia in these patients is essential for better management of the disease1.
Candida Albicans is a predominant pathogen of candidemia in intensive care unit (ICU) patients, with a significant impact on morbidity and mortality rates and is the main species in a variety of settings2,3. Despite advances in antifungal therapy, mortality associated with candidemia remains high, reaching up to 51% in critically ill patients4.
Materials
and Methods
This
is a retrospective descriptive cross-sectional study conducted over a 5-year
period from January 2020 to January 2024, involving 480 blood culture samples
from the intensive care units of the Hassan II University Hospital in Fez,
whose mycological analysis (direct examination, culture and identification)
revealed the presence of Candida Albicans. All patients admitted to the
intensive care unit for a medical or surgical pathology, hospitalized for more
than 48 hours and presenting a clinical or biological infectious syndrome or
clinical signs suggestive of a Candida sp infection and having a risk factor
for the development of a fungal infection were included in the study.
Data collected for each patient were: Age, sex, reason for hospitalization, length of hospital stay, risk factors for the development of Candida infection, concomitant bacterial infections, treatment, mycological results of the various samples received and patient outcome.
Blood
cultures were inoculated into mycotic flasks, each time in the presence of an
infectious syndrome and incubated in Bactec 9240-type automats. In the event of
a positive result and after a fresh state, the broths were transferred to
culture medium (Sabouraud Chloramphenicol ± Actidone medium) and incubated at
37°C for 5 days.
Identification of the Candida Albicans species was based on morphological criteria (colony appearance), phenotypic criteria (filamentation test revealing the presence of germ tubes specific to Candida Albicans), immunological criteria (Latex albi-dubliniensis*) and confirmed by the API Candida* biochemical gallery (BioMérieux).
Antifungal sensitivity was then tested by measuring the Minimum Inhibitory Concentration (MIC) using 4 antifungals: Voriconazole, Fluconazole, Caspofungin and Flucytosine by the ETEST* method. The MIC of the antifungal agent is read after 48 hours at the point of intersection of the ellipse and the strip.
Statistical analysis of the data was carried out using Epi info software. Qualitative variables were expressed as headcount and percentage and quantitative variables as mean and standard deviation or median and quartiles.
Results
Patient characteristics
During
the study period, 480 blood culture requests were sent out, 93 of which were
positive; 29 patients were diagnosed with Candida Albicans candidemia, a
percentage of 31.18%.
The
annual distribution of Candida Albicans candidemia cases showed a progressive
increase over the study period. There was a clear increase from 10.34% in 2020
to 41.66% in 2024 (Figure 1).
Figure
1: Distribution of cases by year
The mean age of patients was 28 years, with extremes ranging from 4 months to 73 years. A slight male predominance was noted, with a M/F sex ratio of 1.07.
In terms of reasons for hospitalization, trauma was the most frequent cause, accounting for 24.13% of cases, followed by respiratory distress, shock and neurological and metabolic disorders, each with a frequency of 17.24% (Table 1).
Table
1: Breakdown of patients by reason for
hospitalization
|
Reason for hospitalization |
Number |
Percentage |
|
Trauma |
7 |
24,13 % |
|
Respiratory distress |
5 |
17,24 % |
|
State of shock |
5 |
17,24 % |
|
neurological disorders |
5 |
17,24 % |
|
Metabolic disorders |
5 |
17,24 % |
|
Other |
3 |
10,34 % |
Figure
2: Distribution of patients according to
risk factors
Mycological results
In
our study, direct examination was positive in 100% of cases, showing the
presence of yeasts and pseudo filaments (Figure 3) and culture was
positive in 100% of cases, revealing smooth, whitish, shiny colonies.

Figure 3: Direct examination showing yeasts with pseudo filaments (objective 40)
Identification of the Candida Albicans species was based firstly on the filamentation test, which was positive for all 29 samples, showing the presence of the germ tube, which is a fine, elongated filamentous projection emerging directly from the yeast mother cell, specific to Candida Albicans (Figure 4).
Figure 4: Germ tube after filamentation test identifying Candida Albicans species (target 40)
A
latex particle agglutination test also demonstrated 100% sensitivity for all
samples, revealing the presence of red agglutinates on a green background,
specific to Candida Albicans (Figure5).
 Positive
agglutination test
Positive
agglutination test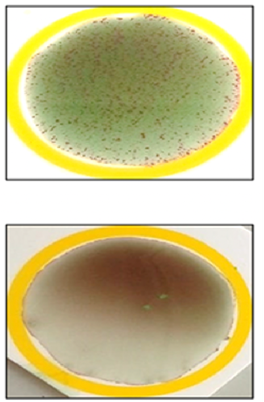
Figure 5: Agglutination test on latex particles: Parasitology - Mycology Department, Hassan II University Hospital, Fez

Confirmation
was provided by the biochemical gallery: API Candida* (Figure 6).

Figure 6: API Candida* biochemical galler
Once the Candida albicans species had been identified, an antifungal sensitivity study was carried out by measuring MICs using 4 antifungals using the ETEST technique.
For
fluconazole, flucytosine and voriconazole, Candida Albicans species show marked
resistance, particularly for voriconazole, with a 72% resistance rate, compared
with only 18% sensitivity. Caspofungin, on the other hand, shows a better
balance between sensitivity and resistance, with almost half the strains being
sensitive, i.e. a rate of 45.45% (Figure7).

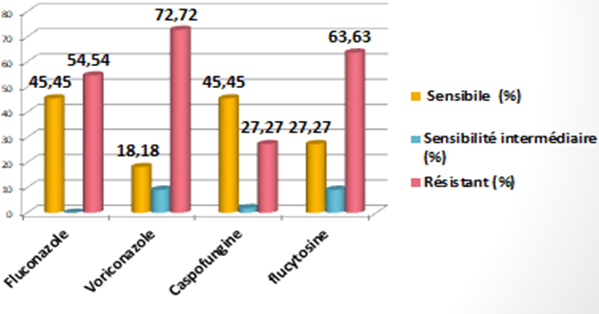
Figure7: Antifungal susceptibility
Discussion
Prevalence
Candidemia is an invasive form of candidiasis. Most cases occur in intensive care units, where the incidence is generally higher than in other departments1. They rank fourth among healthcare-associated infections (HCAI) (10-15%) and have been on the rise in recent years with the emergence of non-albicans Candida5.
is important to prospectively monitor the epidemiology of candidemia, as significant differences may exist between different regions6,7. Prevalence is higher worldwide: in Europe 57% of cases8, in the Americas 37.6%9.
During the study period conducted by Bassetti M et al. in Europe, 570 episodes of ICU-acquired invasive Candidiasis were observed, with a cumulative incidence of 7.07 episodes per 1000 ICU admissions, with significant variability between centers. The separate, non-mutually exclusive cumulative incidences of candidemia and invasive Candidiasis were 5.52 and 1.84 episodes per 1000 ICU admissions respectively. Crude 30-day mortality was 42%8.
During the study period conducted by Nucci M et al. In America 672 episodes of candidemia, 297 (44.2%) occurred in children, 36.2% in adults aged 19-60 and 19.6% in elderly patients. The overall incidence was 1.18 cases per 1,000 admissions and varied from country to country, with the highest incidence in Colombia and the lowest in Chile. Candida Albicans (37.6%), C. parapsilosis (26.5%) and C. tropicalis (17.6%) were the main agents, with great variability in species distribution between countries. Overall survival at 30 days was 59.3%9
In our
study, 31.18% of patients hospitalized in the intensive care unit had proven C.
albicans candidemia. Albicans candidemia. In Morocco, a retrospective study
conducted at the CHU Ibn Rochd in Casablanca over a two-year period showed that
Candida Albicans was the second most common pathogen responsible for
candidemia, with a rate of 32%5,
which concurs with our results. In addition, a previous study carried out in
our parasitology laboratory at the Hassan II University Hospital in Fez between
2015 and 2020 revealed a Candida Albicans candidemia rate of 29%10.
andidemia remains a hospital-acquired infection par excellence. Their frequency in this study is similar to that found in some European countries and has not changed over time. Theistribution of species responsible for candidemia depends on the ecology of each country and each hospital
Epidemiological studies have shown that Candida albicans has emerged as the most common fungal species among Candida in several studies1,11-13, which is consistent with our own. However, in recent years we have increasingly seen an increase in the prevalence of non-albicans Candida species from 59.4% to 68.8% in the Makni et all series14 and from 16.5% to 46.4% in the Fez series (2015-2020)10, which may be due to improved identification techniques, notably culture media as well as chromogenic media and biochemical galleries.
Risk factors and clinical features
Common
risk factors for candidemia include intravenous catheter use, mechanical
ventilation, urinary catheters, prolonged hospital stay and parenteral
nutrition1,15,16
In our study, several risk factors predictive of the development of candidemia were identified. The main factors were central venous catheters and mechanical ventilation, both present in 89.65% of patients, followed by urinary catheters and the use of broad-spectrum antibiotics (72.41%)
These results are in line with a Moroccan study on the analysis of risk factors for systemic candidiasis in the ICU at the Mohammed V Military Training Hospital in Rabat, which also identified urinary catheters, venous catheters, mechanical ventilation and the use of broad-spectrum antibiotics as key factors .
Another study by Fabio et al. found 22 cases of candidemia in a sample of 18,442 patients, with risk factors including renal replacement therapy (40.9% vs. 3.3%), mechanical ventilation (63.6% vs. 29.6%) and parenteral nutrition (40.9% vs. 4.8%). In addition, the length of hospital stay was significantly longer for patients with candidemia, with an average of 42 days versus 8 days for the others15
Predisposing risk factors frequently implicated in candidemia in a study by Selda et al [13] included use of broad-spectrum antibiotic therapy (71.3%), urinary catheterization (56.3%), central venous catheterization (50.3%), parenteral nutrition (47.9%), subjacent hemopathies (46%), recent surgery (48.6%), chemotherapy (37%), steroid treatment (25.5%). The crude mortality rate was 52.7%13
Support
The
management of candidemia caused by Candida Albicans involves a multidimensional
approach that includes rapid diagnosis, appropriate antifungal therapy and
consideration of patient-specific risk factors. Increasing resistance to
antifungal agents and the complexity of candidemia in critically ill patients
necessitate careful selection of treatment strategies17. The main aspects of management are as
follows:
Diagnosis: Candidemia has become a real problem in intensive care patients, with an attributable mortality rate of up to 51%. Numerous global surveillance studies have shown an increasing incidence of candidemia17. Despite their limited sensitivity (21-71%), cultures remain the gold standard for the diagnosis of candidemia. There are many complementary laboratory tests that help to rule out or confirm the diagnosis, each with its own indications and limitations, including procalcitonin, 1,3-β-D-glucan, anti-mannan antibodies and Candida Albicans germ tube antibody. In addition, molecular biology methods could accelerate species identification in positive blood cultures, helping to guide early empirical antifungal therapy17,18.
In our
study, culture is an indispensable tool for confirming the diagnosis of
candidemia, for which it was 100% positive.
Antifungal therapy: The management of candidiasis in critically ill patients can be divided into two categories prophylactic, empirical and targeted treatment of fungal infections. There is no consensus on the benefits of prophylactic treatment in critically ill patients. However, early initiation of appropriate therapy in the case of confirmed candidemia is an important determinant of survival18. The increasing resistance to antifungal agents and the complexity of candidemia in critically ill patients necessitate a rigorous selection of treatment strategies17.
Azole antifungals are the therapeutic class most frequently used to treat Candida Albicans infections19,20. Fluconazole is often the preferred treatment for this species, as it is inexpensive, has limited toxicity and can be administered orally. However, there is abundant literature on intrinsic and developed resistance to azole antifungals in several Candida species. As the frequency of azole-resistant Candida isolates in the clinical setting increases, it is essential to elucidate the mechanisms of this resistance in order to preserve and improve the class of azole antifungals for the treatment of Candida Albicans infections20
In our study, we noted a remarkable resistance of Candida Albicans strains to azole derivatives. Several studies have explored this mechanism of resistance and found mutations in the ERG11 gene, which codes for 14α-demethylase, a target of azoles, to be the main mechanism, leading to reduced efficacy through decreased affinity for their target19,20
The Candida Albicans ERG11 gene codes for lanosterol demethylase, which is targeted by azole antifungals. Mutations in ERG11 can lead to resistance by altering azole binding. In a study by Flowers SA et al. of 63 fluconazole-resistant clinical isolates, 55 had ERG11 mutations and 26 distinct amino acid substitution positions were identified. A total of 31 distinct ERG11 alleles were recovered, including 10 with unique substitutions. Tests revealed that certain single substitutions significantly increased resistance to fluconazole, while strains with multiple substitutions showed even greater resistance. These results highlight the prevalence of ERG11 mutations in azole-resistant isolates and their significant impact on antifungal susceptibility19.
International guidelines recommend the use of echinocandins, including caspofungin, as the treatment option of choice for candidemia in intensive care patients, due to their efficacy against C. Albicans and their favorable safety profiles21,17.
In our study, caspofungin appears to offer the best sensitivity profile, in line with the literature20
Rezafungin, a new long-acting echinocandin approved by the US Food and Drug Administration to treat candidemia and invasive candidiasis, proved effective with a safety profile similar to that of caspofungin in clinical trials21.
Conclusion
Although
Candida Albicans remains a predominant cause of candidemia in intensive care
units, the increasing prevalence of non-Albicans species and associated
resistance patterns pose significant challenges. Continuous monitoring of
epidemiological trends and patient-specific factors is essential to optimize
treatment and improve outcomes in critically ill patients
References
1. 1.
Gugnani
HC. Pediatric candidemia in the Indian subcontinent and parts of the Middle
East, Africa and South America. Eur J Med Health Sci 2022:138-44.
2.
Mazzanti S, Brescini L, Morroni G, et al. Candidemia
in intensive care units over nine years at a large Italian university hospital:
comparison with other wards. PLoS One 2021.
3.
Kim EJ, Lee EY, Kwak HG, et
al. Trends in the epidemiology of candidemia in intensive care units from 2006
to 2017: results from the Korean National
Healthcare-Associated Infections
Surveillance System. Front Med 2020.
4.
El Zakhem A, Istambouli R,
Jabbour J-R, Hindy A, Gharamti A, Kanj SS. Diagnosis and management of invasive
Candida infections in critically ill patients. Semin
Respir Crit Care Med 2022.
5.
Abid
R, El Mabrouki J, Soussi Abdallaoui M. Candidemias at the CHU Ibn Rochd in
Casablanca (Morocco). J Mycol Med 2017;27(3):e26.
6.
Talarmin JP, Boutoille D, Tattevin P, et al. Epidemiology
of candidemia: a one-year prospective observational study in western France.
Med Mal Infect
2009;39:877-885.
7.
Eggimann
P, Bille J, Marchetti O. Diagnosis of invasive candidiasis in the ICU. Ann
Intensive Care 2011;1:37.
8.
Bassetti M, Giacobbe DR, Vena A, et al. Incidence
and evolution of invasive candidiasis in intensive care units in Europe:
results of the EUCANDICU project. Crit
Care 2019;23:219.
9.
Nucci M, Queiroz-Telles F, Alvarado-Matute T, et
al. Epidemiology of candidemia in Latin America: a laboratory-based
survey. PLoS One 2013;8(3):e59373.
10.
Ghmammad
W, Berrada S, Ben-Saghroune H, Tlamcani Z. Le profil épidémiologique des
candidoses au sein du service de parasitologie-mycologie du CHU
Hassan II de
Fès. IOSR J Pharm Biol Sci 2023;18:36-41.
11.
Özel AS,
Altunal LN, Ertürk B, Süzer M, Aydın M. Predictors of outcomes in patients with
candidemia in an intensive care unit. Marmara Med J 2023.
12.
Qiao
Y, Zha T, Hao F, et al. Epidemiological characteristics, antifungal
susceptibility, risk factors and outcomes of Candida bloodstream infection: a
ten-year
surveillance in a teaching hospital in China. Infect Drug Resist 2023.
13.
Aydin S, Derin O, Şahin M, et
al. Epidemiology of nosocomial candidemia, mortality and antifungal resistance,
7-year experience, in Turkey. Jpn J Infect Dis
2022;75(6):597-603.
14.
Makni
F, Sellami A, Trabelsi H, Sellami H, Fatma C. Évolution de la flore des levures
isolées au CHU de Sfax, Tunisie. J Mycol Med 2010;20:42-47.
15.
Hohmann FB, Chaves RCF,
Olivato GB, et al. Characteristics, risk factors and outcomes of bloodstream
Candida infections in the intensive care unit: a
retrospective cohort study. J
Int Med Res 2023.
16.
Massou
S, Ahid S, Azendour H, et al. Les candidoses systémiques en réanimation
médicale : analyse des facteurs de risque et intérêt de l'index de
colonisation. Pathol Biol 2013;61(3):108-112.
17.
El Zakhem A, Istambouli R,
Jabbour JR, et al. Diagnosis and management of invasive Candida infections in
critically ill patients. Semin Respir Crit Care Med
2022.
18.
Tokimatsu
I. Management of candidemia. Jpn J Infect Prev Control 2020.
19. Flowers SA, Colón B,
Whaley SG, et al. Contribution of clinically derived mutations in ERG11 to
azole resistance in Candida albicans. Antimicrob Agents Chemother
2015;59(1):450-460.
20.
Whaley
SG, Berkow EL, Rybak JM, et al. Azole antifungal resistance in Candida albicans
and emerging non-albicans Candida species. Front Microbiol
2017;7:2173.
21.
Thompson GR, Soriano A, Honore
PM, et al. Efficacy and safety of rezafungin and caspofungin in candidemia and
invasive candidiasis: pooled data from two prospective randomized controlled
trials. Lancet Infect Dis 2024;24(3):319-328.