Case Report: A Rare Case of an Atypical Lipomatous Tumor
ABSTRACT
Liposarcomas are the most
common histologic subtype of soft tissue sarcomas. The World Health Organization (WHO) recognizes five
histological sub-types of liposarcoma: well-differentiated or atypical lipoma,
undifferentiated, myxoid, round cells, and pleomorphic. This histological
differentiation is important since it determines the prognosis and guides the treatment protocol.
As well-differentiated liposarcomas of any type have no potential to
metastasize unless they undergo dedifferentiation. Complete surgical removal of
atypical lipomas is the treatment of
choice. However, these tumors recur repeatedly, can dedifferentiate, and thus
acquire metastatic potential. Radiotherapy may be needed if surgical margins are small or positive, then adjuvant
or neoadjuvant. Here, we report a rare case of atypical lipoma in the
left parotid region of a 57-year-old patient. The patient is HIV positive and
had cervical cancer that was treated solely through hysterectomy but with sign
of progression. The patient could not be operated so radiotherapy alone was
chosen as the sole treatment modality. The patient was prescribed 70 Gy in 2Gy
daily fractions, but the patient stopped treatment at 40 Gy for many health
problems not related to radiotherapy administration. Even with this dose, there
was a significant response with about a 90% reduction in tumor volume.
Keywords: A typical pleomorphic
lipomatous tumor; A typical spindle cell lipomatous tumor; Pleomorphic
liposarcoma.
INTRODUCTION
The
treatment of choice for atypical lipomas is complete surgical removal. However,
despite their benign nature, these
neoplasms can dedifferentiate, acquiring the potential to metastasize. This underscores the importance of early and accurate diagnosis. In cases where surgical margins are small or positive, adjuvant
or neoadjuvant, radiotherapy may be administered. The use of
adjuvant/neoadjuvant chemotherapy on well-differentiated liposarcomas and
undifferentiated liposarcomas remains a topic of debate. The classification of
the previously unexplored group of atypical adipocytic neoplasms with spindle
cell characteristics, now termed atypical lipomatous spindle cell tumor,
presents a significant challenge. Recent research has identified atypical
lipomatous spindle cell tumors as a distinct entities characterized by specific
genetic abnormalities, mainly deletions/losses of 13q14, including RB1 and its adjacent genes,
RCBTB2, DLEU1, and ITM2B. Similar genetic aberrations have been observed
in pleomorphic liposarcomas1,2,3. The microscopic
examination of these tumors reveals a broad spectrum of histological features,
each holding a key to understanding their nature. All cases consisted of
slightly atypical spindle cells in a fibrous or myxoid stroma with a variable
amount of adipocyte components showing variation in adipocyte size, scattered
nuclear atypia, and frequent non- or multivacuolated lipoblasts2,4. The
complete surgical removal of atypical lipomas is the treatment of choice, a
decision backed by extensive research and clinical experience. However, these neoplasms recur
repeatedly, can dedifferentiate, and thus acquire metastatic potential5,6.
THE CASE
The patient is a
57-year-old patient with a history of HIV infection and is under treatment. The
patient previously had cervical cancer treated three years ago by total
hysterectomy, but no chemotherapy or radiation therapy, and there is no sign of
progression. She was admitted to the Cameroon Oncology Center with a painful
left head mass located in the tempo parietal section of the brain. A
biopsy of the mass carried out at another medical institution shows that it is
an atypical lipomatous tumor. On physical examination, the patient has
significant retro auricular swelling going upwards to the left temporal region
and downwards to the mandible. The tumor was painful when touched. The
cervical-facial CT contrast-enhanced scan highlighted mixed attenuation and
bulging left retro-auricular soft tissue mass infiltrating the ipsilateral
frontal, occipital, and parietal scalp. The mass extends
into the cranium along the left temporal,
frontal, and parietal lobes,
where it remains extra-axial and epidural with marked contrast enhancement of
the dura. The left temporal lobe has a mass effect with sulci and Sylvian
fissure effacement and mild subfalcine midline shift. (Figure 1) shows a cross-sectional cut of the brain through the
mass and a 3D rendering of the CT scan showing left sided mass.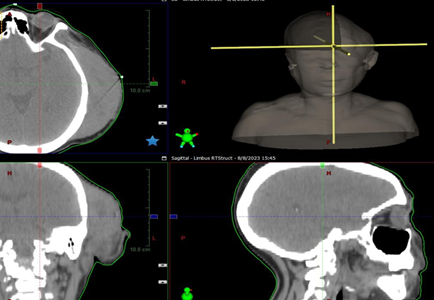
Figure 1.
Cross-sectional cut the head
showing the tumor and 3D reconstruction.
Given the patient’s other
health problems, including frequent infections and hospitalization, and the
fact that the surgeons were not confident of total resection, radiotherapy was
chosen as the monotherapy for this patient. For post-operative radiotherapy,
the radiation prescription dose ranges from 50.4 Gy in 28 to 66 Gy in 33
fractions7. For radiotherapy alone,
the prescription dose ranges from 60
Gy in 30 to 70 Gy in 35 fractions.
As the tumor was not
resectable, after a multidisciplinary consultation
meeting, it was decided to give radiotherapy at a dose of 70Gy in classic
fractionation of 2Gy per session and five sessions per week. The Eclipse
Treatment Planning System (version 15.6.8) was used in developing the plan. The
plan was a five fields IMRT field technique. The reason for using IMRT was to
limit risk organ doses in the brain and neck region. Except for the left parotid gland, which received the full prescription dose,
most organs at risk received
any dose close to the tolerance doses
published by QUANTEC8. The left
lens, about 1 cm from the PTV volume, only received 1.3 Gy (see Figure 3). (Figure 2) shows the color-wash of the
dose distribution in all three planes and the dose volume histogram. The doses
shown in (Figure 4) range from 30Gy
to 75Gy. D95=70Gy for the Planning Target Volume (PTV).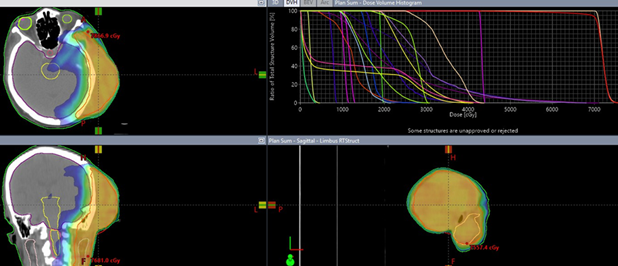
Figure 2. Isodose Distribution displayed as color-wash with a minimum
dose of 30 Gy.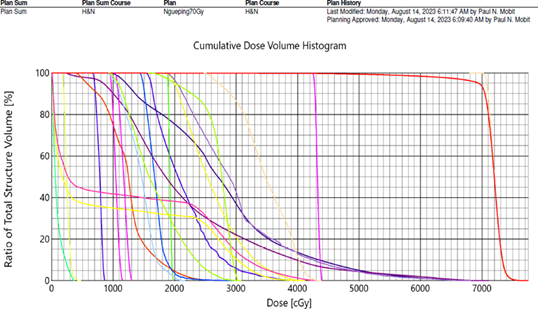
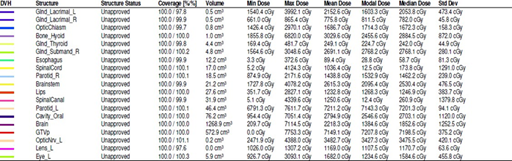
Figure 3.
Dose Volume Histogram Analysis of the critical structures surrounding the tumor
RESULTS & DISCUSSIONS
The patient
received 20 fractions of the 2 Gy per fraction for 40 Gy. The patient had many
infections during the five weeks of treatment and was hospitalized throughout
most her radiotherapy treatment. By the end of the 2nd week of
treatment, the pain was much diminished. Still, given that the patient was not
doing well health-wise, the radiation oncologist decided to discontinue her
radiotherapy and considered the already administered radiotherapy as a good
palliative dose. The patient was discharged from the hospital and returned
home. Four months later, the patient
returned for a post-radiotherapy follow-up, and it was a surprise to see that
the tumor was more than 90% resolved. The patient’s health condition has also improved remarkably during
this period, and the pain associated with the tumor is gone. Figure 4: the
picture of the before the radiotherapy treatment and post-treatment.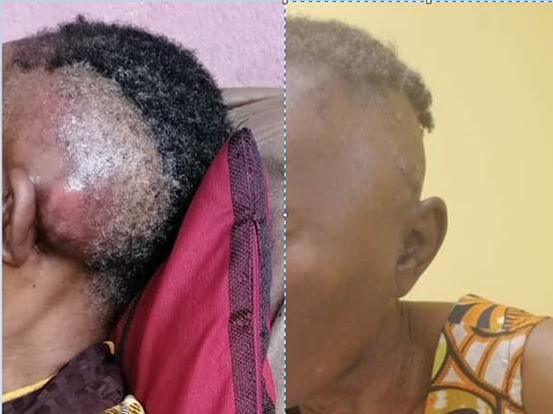
Figure 4. Before the treatment and after the treatment picture
CONCLUSION
Liposarcoma is a rare malignant entity
whose definitive diagnosis is anatomopathological.
The histological differentiation is essential; it determines the prognosis and
guides the treatment. Surgical resection as wide as possible constitutes the
only therapeutic means. Radiotherapy is necessary when surgical limits are
marginal. Despite the lower radiation therapy dose of 40 Gy administered to the
patient rather than 70 Gy planned, there has been a remarkable reduction of at
least 90% reduction in tumor volume. Given that we were significantly below the
risk organ doses for the entire plan of 70 Gy, if the tumor
recur then we can still replan to deliver a dose of 30-40 Gy efficiently without exceeding
risk organ tolerances. So this case demonstrate the effective for radiotherapy
even for lower administered dose of 40 Gy for liposarcoma.
Conflicts of interest: The authors declare no conflict of interest.
Author contributions: All authors read and approved the final version of the manuscript.
REFERENCES
1. Dodd LG. Update on liposarcoma: A review for cytopathologists. Diagn Cytopathol 2012;40(12):1122‑1131.
2. Creytens D, Mentzel
T, Ferdinande L, et al. Atypical
Pleomorphic Lipomatous Tumor: A Clinicopathologic, Immunohistochemical and Molecular Study of 21 Cases, Emphasizing its Relationship to Atypical Spindle Cell Lipomatous Tumor
and Suggesting a Morphologic Spectrum (Atypical Spindle Cell/Pleomorphic Lipomatous
Tumor). Am J Surg Pathol 2017;41(11):1443‑1455.
3. Henze J, Bauer
S. Liposarcomas. Hematol
Oncol Clin North
Am 2013;27(5):939‑955.
4. Bahadir B, Behzatoglu K, Hacıhasanoglu E, Koca SB, Sigirci
BB, Tokat F. Atypical spindle cell/pleomorphic lipomatous tumor: A clinicopathologic, immunohistochemical, and molecular study of 20 cases. Pathol Int 2018;68(10):550‑556.
5. Mentzel T, Fletcher CD. Lipomatous tumours
of soft tissues: An update.
Virchows Arch Int J Pathol
1995;427(4):353‑363.
6. Kim YB, Leem DH, Baek JA, Ko SO. A typical lipomatous
tumor/well-differentiated liposarcoma of the gingiva: a case report
and review of literature. J Am Assoc Oral Maxillofac Surg 2014;72(2):431‑439.
7. Allignet B, Waissi W, Geets X, et al. Longterm outcomes after definitive
radiotherapy with modern techniques for unresectable soft tissue sarcoma.
Radiother Oncol 2022;173:55-61.
8. Marks LB, Yorke ED, Jackson A, et al. Use of normal tissue
complication probability models
in the clinic. Int J Radiat Oncol Biol Phys 2010;76:10-19.