Case Report, Erythroderma and Dactylitis in Mycosis Fungoides
ABSTRACT
MF is a type of mature T cell non-Hodgkin lymphoma
that typically appears in the skin, but it can also affect the blood, viscera,
and nodes. Tumors, erythroderma, patches and plaques that might be localized or
widespread are examples of skin lesions. Although the exact origin of MF is
unknown. Common characteristics include epigenetic changes, aberrant RNA
splicing, altered JAK-STAT signaling, and T cell receptor (TCR)/T cell
activation.
We
present the case of a 65-year-old male with many episodes of Erythroderma in addition to moderate pruritus and dactylitis, in the last 2 years. At
Hospital Carlos Andrade Marin in Quito, Ecuador.
Keywords:
Erythroderma; Mycosis fungoides; Dactylitis
Abbreviations:
CTCL:
Cutaneous T cell lymphoma
MF:
Mycosis Fungoides
SS:
Sézary Syndrome
INTRODUCTION
Erythroderma, also known as exfoliative dermatitis,
is a severe and sometimes fatal disorder that manifests as diffuse erythema and
scaling over 90% of the skin's surface area. Erythroderma can be the clinical
manifestation of many different systemic and cutaneous disorders (such as
atopic dermatitis and psoriasis), medication hypersensitivity reactions, and,
less frequently, Sézary syndrome, a leukemic subtype of cutaneous T cell
lymphoma1-3. Within cutaneous T cell
lymphoma (CTCL), Mycosis fungoides (MF) and Sézary syndrome (SS) are the most
prevalent subtypes3.
MF is a type of mature T cell non-Hodgkin lymphoma
that typically appears in the skin, but it can also affect the blood, viscera,
and nodes. Tumors, erythroderma, and patches or plaques that might be localized or
widespread are examples of skin lesions.
Patients with poikilodermatous
skin abnormalities, generalized erythroderma, or chronic nonspecific dermatitis
may be suspected of having MF. A skin biposy hast he highest yield to help the
physician diagnose MF; several skin biopsies are frequently needed. The
lesional skin with the highest degree of induration should be included in the
single biopsy, if one is taken. A 4 mm punch biopsy at the very least is
advised. Biopsies can usually be preserved in formalin before being stained
with hematoxylin and eosin or subjected to further pathological testing.
Individuals from endemic areas should have serologic testing for HTLV-I3,5,6.
The European Organization for Research and Treatment
of Cancer (EORTC) cutaneous lymphoma task force and the International Society
for Cutaneous Lymphoma (ISCL) have proposed an algorithm for diagnosing and
staging early MF based on clinical, histopathologic, molecular, and
immunopathologic criteria. It is advised that the ISCL/EORTC algorithm be used
to confirm the diagnosis of every patient included in MF trials or databases3,7,8 (Table 1).
CASE PRESENTATION
We
present a 65-year-old male with medical history of mixed anxiety disorder,
gastritis, chronic deep vein thrombosis. He had presented progressive pruritus
since approximately 63 years of age, had had two skin biopsies, for which he
had been diagnosed of psoriasis. 6 months ago, our patient presented
erythroderma that required hospitalization at other hospitals. The differential
diagnoses included MF, psoriasis, atopic dermatitis, adverse drug reaction. He
had been managed with: topical corticosteroids, methotrexate, or cyclosporine,
with partial improvement.
In
February 2024, he
presented to the emergency room at Hospital Carlos Andrade Marín with
generalized, intense pruritus, erythroderma and eczematous plaques covered with
thin crust or scales. The nails were thick, as were the palms and soles and
presented painful f. The 3rd left finger presented indurated edema.
The eyes and ears discharged yellowish fluid (Figure
1 and Figure 2). Laboratory exams were
within normal range, aside from a mildly elevated eosinophilia of 1500 per cc3.
Infectious diseases were ruled out (HIV, CMV and Anti-HTLV antibodies 1 and 2
were negative). Auto antibodies including ANA, Anti-DNA, Anti-Histone,
Anti-Ro/La were negative). Paraneoplastic syndromes were ruled out and included
full-body CT scan, upper endoscopy, colonoscopy, occult blood in stool; and
they were all negative.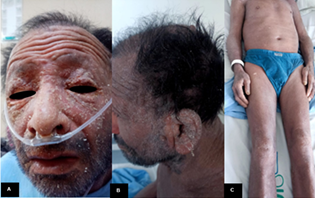
Figure 1. A-C. Generalized erythroderma
and desquamation, affecting more than 90% of the body surface.
A,
B.
Infiltration of the entire facial mass, auricular pavilions.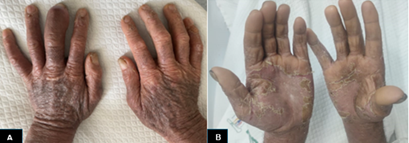
Figure 2. A. Marked dactylitis of the 3rd finger of the
left hand. B. Bilateral palmar hyperkeratosis with
significant scaling.
Two
skin biopsies were taken, and previous biopsies were requested for comparison.
The previous biopsies
revealed psoriasiform and spongiotic dermatitis, and a diagnosis of psoriasis
was precluded. However, immunohistochemistry was performed, and showed:
epidermis with foci of parakeratosis, spongiosis and irregular hyperplasia of
the ridge network, the dermis with lymphocytic infiltrate with nuclear atypia,
hyperchromasia, located in the superficial dermis, some extending into the
epidermis to form occasional pautrier's microabscesses.”
Immnohistochemistry
Results:
LCA CD3, CD4, CD5 positive in dermal and
intraepidermal lymphocytes.
CD8: positive in dermal t-lymphocytes.
CD4/CD8 ratio: 4:2
CD2, CD7: scantly positive in some dermal
lymphocytes.
CD30: positive greater than 25%, in this sample,
60%.
KI67: 20%.
CD20 and Granzyme B: negative.
With the results of cutaneous Atypical Lymphoid
proliferation, a diagnosis of Mycosis Fungoides with CD30 greater than 25%
(suggests transformation) was made.
Upon his hospitalization and due to previous
treatment failure with methotrexate cyclosporine was started at a dose of
3mg/kg daily, as well as loratadine 10mg QID, hydroxyzine 10mg HS, and topical
steroids and emollients. The patient presented marked improvement of his
clinical manifestations, including his dactylitis at one-month review (Figure 3). At the two month review,
the patient started gaining weight and the skin continued to improve (Figure 4). 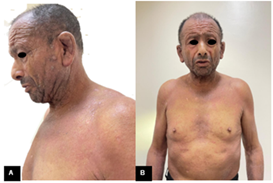
Figure
3. A, B.
Patient one month after the last hospitalization, with improvement of lesions.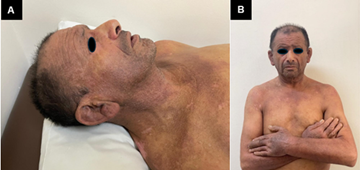
Figure
4. A, B.
Patient at present, two months after the last hospitalization, with improvement
of lesions and even weight gain.
DISCUSSION
Patients with MF commonly present with
persistent and/or slowly progressive skin lesions of varying size and shape.
Skin lesions may be localized or widespread patches or plaques, tumors, and/or
generalized erythroderma. The skin is often pruritic and the patient's quality
of life can be profoundly affected9. Other clinical manifestations include opportunistic infections, alopecia,
and, less commonly, involvement of other organs. Some of these clinical
features are used in the point-based diagnostic algorithm (Table
1).
Table 1. Diagnosis of early
mycosis fungoides7
A total of 4 points is required for the
diagnosis of mycosis fungoides based on any combination of points from the
clinical, histopathologic, molecular biologic, and immunopathologic criteria.
* Lymphoid atypia is defined as cells with
enlarged hyperchromatic nuclei and irregular or cerebriform nuclear contours.
¶ T cell antigen deficiency confined to the
epidermis.
A definitive diagnosis of MF is often
preceded by a "premycotic" period ranging from months to decades,
during which the patient may have pruritus and nonspecific, slightly scaling
skin lesions and nondiagnostic biopsies for months to years. These lesions may
wax and wane over years, and a diagnosis of parapsoriasis en plaque or
nonspecific dermatitis is often made3-8.
EVALUATION
The diagnosis of MF is
suspected in patients who present with chronic nonspecific dermatitis,
poikilodermatous skin findings, or generalized erythroderma. Skin biopsy with
routine histology is the single most important laboratory tool that will assist
the clinician in establishing the diagnosis of MF(5). Often,
multiple skin biopsies are required. If only one biopsy is performed, it should
include the lesional skin with the most induration3,10.
As we present this case report, it is important to
emphasize the negativity of HTVL-1, reported in the clinical history, because
the histologic differential diagnosis is leukemia/lymphoma and HTLV-1 positive
adult T-cell Leukemia/Lymphoma3,11.
CONCLUSIONS
The diagnosis of this
case of Mycosis Fungoides is based on
clinical, histopathologic, immunopathologic findings. The patient had had two
years of dermatologic manifestations, as we know we must do a good
anamnesis and the necessary studies for a good diagnosis and treatment.
Also, we can’t forget the psychological and social impact for a holistic
management12,5.
Conflict of
Interest: The authors declare no conflicts of
interest.
REFERENCES
1. Campo
E, Jaffe ES, Cook JR, et al. The international consensus classification of
mature lymphoid neoplasms: a report from the clinical advisory committee. Blood
2022;140(11):1229-1253.
2. Alaggio R, Amador
C, Anagnostopoulos I, et al. The 5th edition of The world health organization
classification of haematolymphoid tumours: Lymphoid neoplasms. Leukemia
2022;36(7):1720-1748.
3. Hoppe
RT, Kim YH. Clinical manifestations, pathologic features, and diagnosis of
mycosis fungoides. UpToDate 2024.
4. Bolognia
JL, Schaffer JV, Cerroni L. Dermatology 5th edition Vols 2. Elsevier -
Saunders, Mosby, Churchill 2024.
5. Fung MA, Murphy
MJ, Hoss DM, Grant-Kels JM. Practical evaluation and management of cutaneous
lymphoma. J Am Acad Dermatol 2002;46(3):325-357.
6. Cuellar-Barboza A,
Ocampo-Candiani J, Herz-Ruelas ME. A Practical Approach to the Diagnosis and
Treatment of Adult Erythroderma. Actas Dermosifiliogr (English Edition)
2018;109(9):777-790.
7. Pimpinelli N,
Olsen EA, Santucci M, et al. Defining early mycosis fungoides. J Am Acad
Dermatol 2005;53(6):1053-1063.
8. Ryu HJ, Kim S Il,
Jang HO, et al. Evaluation of the international society for cutaneous lymphoma
algorithm for the diagnosis of early mycosis fungoides. Cells 2021;10(10):2758.
9. Demierre MF, Gan
S, Jones J, Miller DR. Significant impact of cutaneous T-cell lymphoma on
patients’ quality of life: Results of a 2005 National Cutaneous Lymphoma
Foundation Survey. Cancer 2006;107(10):2504-2511.
10. Lee H. Mycosis fungoides and Sezary
syndrome. Blood Res 2023;58.
11. Karube K, Takatori
M, Sakihama S, et al. Clinicopathological features of adult T-cell
leukemia/lymphoma with HTLV-1-infected Hodgkin and Reed-Sternberg-like cells.
Blood Adv 2021;5(1):198-206.
12. Walia
R, Yeung CCS. An Update on Molecular Biology of Cutaneous T Cell Lymphoma. Front
Oncol 2020;9:1558.