Effects of GastriSan on gastritis symptoms - a randomized, single-blind, placebocontrolled study
Keywords: gastritis; gastritis herbal remedy; cabbage; cabbage extract; kale
Materials and methods
This was a randomized, single-blind,
placebo-controlled study conducted at fundeni clinical institute, bucharest,
romania. A total of 50 patients were randomly divided into two groups: test (n
= 25) and placebo (n = 25) in a randomization ratio of 1:1. Test product was a
tablet containing dehydrated powder of brassica oleracea 450 mg, and the
placebo was an identical capsule containing starch. Both treatments were given
twice daily with water for 28 days. We asked the patients to complete a visual
analog scale investigating the influence of gastritis symptoms on the health
before and after 4 weeks of treatment. After the treatment we compared the vas
scores.
Patients. We selected 50 patients admitted between 2022 and 2023 to the fundeni clinical institute (bucharest, romania). 26 male and 24 females were included in the present study. The medium age for men was 52 (min 27 max 81) and the medium age for females was 54 (min 23, max 87). The inclusion criteria was a diagnosis of gastritis obtained by an upper digestive endoscopy at screening and a gastric biopsy was obtained and we performed a rapid urease helicobacter pylori from the gastric mucosa biopsy. Exclusion criteria were: recent treatment with any medication for peptic disease in the last month prior to enrolment, endoscopic evidence or history of neoplasia, known diagnosis of gastric cancer, any other concomitant pathology that require treatment and the presence of other concomitant treatments or pathology that can influence the study results. We obtained the informed consent from all patients.
Measurement of scales. We used a 100 millimetres scale printed on a a4 sheet of white paper with black ink. We explained every patient how to use the visual analogue scale. The patients were in a silent environment without distractions. We collected the completed scales at visit 1 and visit 2 in the patient file. We used the same ruler for all the patients. The question was: “we would like to know how good or bad your health is today related to gastritis symptoms”.
Statistical analysis. Statistical analysis and database management were performed using jasp 0.16.3 software for windows11. Descriptive results were presented as the mean ± standard deviation (sd) or number (percentage) of patients. We used multiple comparisons for the anova test to compare the mean data. The tests were two-tailed and p<0.05 was considered to indicate a statistically significant difference. Results.
Patient characteristics. A total of 50 patients mean age 52.96±16.03 years were included. 26 patients were male with a mean age of 52±15,37 years and 24 females with a mean age of 54±16,98 years. The main demographic data of the patients are summarized in table 1 and the clinical and laboratory data in (table 1)
Table 1: demographic characteristics of the study population
|
Descriptive
statistics |
|
valid missing meanstd.
Deviation minimum maximum |
|
Age (years) 50 0 52.96 16.03 23.00 87.00 |
Table 2: demographic characteristic splited
by sex
|
Descriptive
statistics |
|
valid
missing meanstd. Deviation minimum maximum |
|
Age (years)
female 24
0 54.00 16.98 23.00
87.00 |
|
Age (years) male 26 0 52.00 15.37
27.00 81.00 |
Variation of vas score before and after treatment. The medium value of vas1 was 72,96 (min 56 max 88) for all patients. The value of vas2 (on day 29) was 75,76 (min 55 max 95). The medium difference delta was 2,8 (min -7 max 21). The results indicated that all the patients had a small increase of vas after 28 days (table 2)
Variation of vas score before and after treatment for placebo group. In the placebo group were 25 patients, 17 women and 8 men. The medium value of vas1 was 71,76 (min 56 max 87) for placebo group. The value of vas2 (on day 29) was 69,12 (min 55 max 80). The medium difference delta was -2,6 (min -7 max 2). The results indicated that vas had a decrease after 28 days (table 3).
Variation of vas score before and after treatment for gastrisan group. In the gastrisan group were 25 patients, 7 women and 18 men. The medium value of vas1 was 74,16 (min 58 max 88) for gastrisan group. The value of vas2 (on day 29) was 82,4,12 (min 63 max 95). The medium difference delta was 8,23 (min -1 max 21). The results indicated that all the patients had an increase of vas after 28 days in the gastrisan group. We applied t-student on the vas2-vas1 (delta) and treatment groups with the hypothesis that the gastrisan group has an superior increase in vas compared with the placebo group and the result was significant (table 4, table 5, figure 2).
Table 3: independent samples t-test
|
T |
df |
p |
|
Delta
8.39 |
48 |
2.83×10-11
ᵃ |
Note. For all
tests, the alternative hypothesis specifies that group gastrisan is greater than group placebo.
Note. Student's t-test.
ᵃ levene's test is significant (p <
.05), suggesting a violation of the equal variance assumption
|
|
|
|
Group |
N |
Mean |
Sd |
Se |
Coefficient of variation |
|
Delta |
Gastrisan |
25 |
8.24 |
5.98 |
1.20 |
0.73 |
|
|
Placebo |
25 |
-2.64 |
2.50 |
0.50 |
-0.95 |
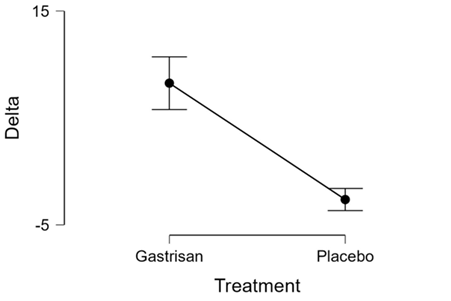
Figure 1: descriptives plots the evolution of delta in gastrisan and placebo groups
after 28 days of treatment
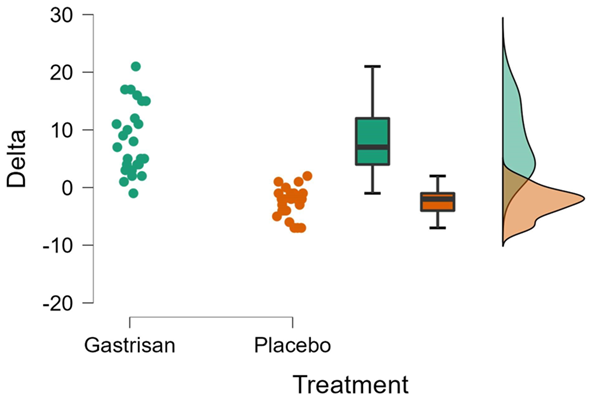
Figure 2: raincloud plots the graphic variation of delta in the gastrisan and placebo groups before and after 28 days of treatment
Variation of vas score before and after treatment for helicobacter pylori negative patients. 35 patients (16 women and 19 men), 18 on gastrisan group and 17 in placebo group had helicobacter pylori negative result in the gastric biopsy urease test. The medium value of vas1 was 73,31 (min 56 max 88). The value of vas2 (on day 29) was 77,4 (min 55 max 95). The medium difference delta was 4,11 (min -7 max 21). The results indicated that all the patients helicobacter pylori negative had an increase of vas after 28 days. We applied t-student on the vas2-vas1 (delta) and helicobacter pylori urease test results with the hypothesis that the negative helicobacter pylori group has an superior increase in vas compared with the placebo group and the result was significant (table 5).
Table 5: independent samples t-test
|
t
|
df |
p |
|
Delta
2.06
|
48 |
0.02
ᵃ |
Note. For all
tests, the alternative hypothesis specifies that group negative is greater than group positive.
Note. Student's t-test.
ᵃ levene's test is significant (p < .05), suggesting a violation of the equal variance assumption
Variation of vas score before and after treatment for helicobacter pylori positive patients. 15 patients (8 women and 7 men), 7 on gastrisan group and 8 in placebo group had helicobacter pylori positive result in the gastric biopsy urease test. The medium value of vas1 was 72,13 (min 63 max 80). The value of vas2 (on day 29) was 71,86 (min 64 max 85). The medium difference delta was -0,27 (min -7 max 8). The results indicated that all the patients helicobacter pylori positive had a decrease of vas after 28 days. We applied t-student on the vas2-vas1 (delta) and helicobacter pylori urease test results with the hypothesis that the negative helicobacter pylori group has an superior increase in vas compared with the helicobacter pylori positive and the result was significant (table 5, table 6, figure 3).
Table
6: group descriptives
|
|
Group |
N |
Mean |
Sd |
Se |
coefficient of variation |
|
Delta
|
Negative
|
35 |
4.11
|
7.65 |
1.29 |
1.86
|
|
|
Positive
|
15 |
-0.27
|
4.59 |
1.19 |
-17.21
|
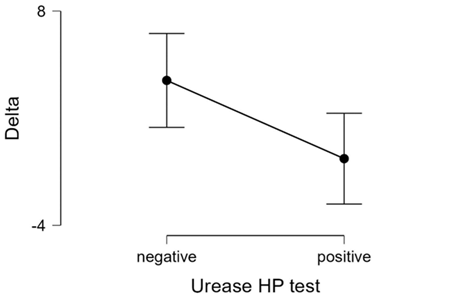
Figure 3: descriptives plots values of delta in the patients with or without helicobacter pylori
Variation of vas score before and after treatment for helicobacter pylori pozitive patients on gastrisan arm. 7 patients (3 women and 4 men), had helicobacter pylori positive result in the gastric biopsy urease test. The medium value of vas1 was 70,85 (min 63 max 80). The value of vas2 (on day 29) was 74,42 (min 64 max 85). The medium difference delta was 3,57 (min -1 max 8). The results indicated that all the patients helicobacter pylori positive had a mild increase of vas after 28 days.
Variation of vas score before and after treatment for helicobacter pylori negative patients on gastrisan arm. 18 patients (4 women and 14 men), had helicobacter pylori negative result in the gastric biopsy urease test in the gastrisan group. The medium value of vas1 was 75,44 (min 58 max 88). The value of vas2 (on day 29) was 85,5 (min 63 max 95). The medium difference delta was 10,05 (min 1 max 21). The results indicated that all the patients helicobacter pylori negative had a high increase of vas after 28 days (table 7).
Variation of vas score before and after treatment for different types of gastritis patients on gastrisan arm. The digestive endoscopy identified 5 types of gastritis on our patients: gave, atrophic gastritis, biliary reflux gastritis, erosive gastritis and portal hypertensive gastritis (table 8, figure 4). The gave category had only one patient and the data couldn’t be compared with placebo. All the other types of gastritis showed an increase in the vas2 value.
|
|
level |
Counts |
Total |
Proportion |
Bf₁₀ |
|
Treatment
|
Gastrisan
|
25 |
50 |
0.50
|
0.17
|
|
|
Placebo
|
25 |
50 |
0.50
|
0.17
|
|
Sex |
Female
|
24 |
50 |
0.48
|
0.18
|
|
|
Male
|
26 |
50 |
0.52
|
0.18
|
|
Urease
hp test |
Negative
|
35 |
50 |
0.70
|
9.81
|
|
|
Positive
|
15 |
50 |
0.30
|
9.81
|
|
Digestive
endoscopy |
Gave
|
1 |
50 |
0.02
|
4.42×10+11 |
|
|
Atrophic
gastritis |
5 |
50 |
0.10
|
1.04×10+7
|
|
|
Biliary
gastritis |
17 |
50 |
0.34
|
2.24
|
|
|
Erosive
gastritis |
22 |
50 |
0.44
|
0.25
|
|
|
Portal
gastropathy |
5 |
50 |
0.10
|
1.04×10+7
|
Tabel 7: bayesian binomial test
note.
Proportions tested against value: 0.5. The shape of the prior
distribution under the alternative hypothesis is specified by beta(1, 1).
Discussion
Improvement
in the vas score was observed in all patients in the gastrisan group compared
with placebo p=2.83×10-11(table 4).
The presence of helicobacter pylori has a negative influence on the results
p=0,02 (table 6). All types of
gastritis identified at digestive endoscopy had an increase of vas after
treatment with gastrisan (table 8,
figure 4). The anova analysis demonstrated the efficacity of gastrisan
treatment on gastritis symptoms (table 9, table 10, table 11).
Table 8: anova - delta and treatment arms
|
Cases |
sum of squares |
df |
Mean square |
f |
p |
|
Treatment |
1479.68
|
1 |
1479.68
|
70.44
|
5.65×10-11 |
|
Residuals |
1008.32
|
48 |
21.01
|
|
|
Note. Type iii sum of squares
Table
9: descriptives
|
Descriptives delta and the
treatment arms | |||
|
Treatment
|
Mean |
Sd |
N |
|
gastrisan |
8.24 |
5.98 |
25
|
|
placebo |
-2.64 |
2.50 |
25
|
Table 10: descriptives - delta and sex
|
Sex |
Urease
hp test |
Mean |
Sd |
N |
|
Female
|
negative |
0.44 |
6.93 |
16
|
|
|
positive |
-1.88 |
3.87 |
8 |
|
Male
|
negative |
7.21 |
6.96 |
19
|
|
|
positive |
1.57 |
4.93 |
7 |
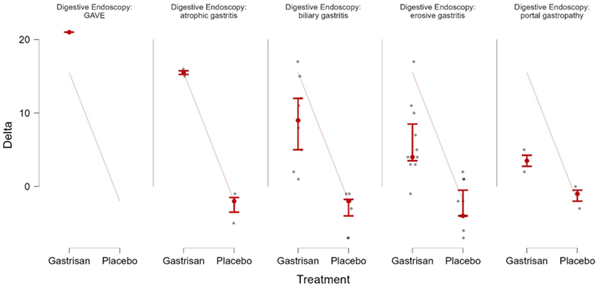
Figure 4: effects of gastrisan on types of gastritis flexplot
Conclusion
In our study, we found significant
improvement in reduction of gastritis symptoms. All the patients receiving
gastrisan had an improvement in the symptoms vas scale after 28 days of
treatment. So gastrisan can be used as a treatment option for these patients.
The patients should be tested for helicobacter pylori. The presence of
helicobacter pylori amplifies the gastritis simptoms and it should be treated
with triple therapy. Gastrisan can be used concomitant with other gastritis
treatments.
Gastrisan can be used for chronic biliary reflux gastritis, atrophic gastritis, portal hypertensive gastritis and autoimmune gastritis. However, more research is needed to confirm the effectiveness and safety of cabbage juice for gastritis.
References
1.
cheney g. Vitamin u therapy
of pepculcer. Calif med 1952;177(4):248-252.
2.
šamec d, urlić b, salopek-sondi b. Kale (brassica oleracea
var. Acephala) as a superfood: review of the scien fic evidence behind the
statement. 2018 apr 24, crit rev food sci nutr 2019;59(15):24112422.
3.
Marivane lemos, josé roberto san n, luiz
carlos klein júnior, rivaldo niero, sérgio faloni de andrade. Gastroprotec ve ac
vity of hydroalcoholic extract
obtained from the leaves of brassica oleracea
var. Acephala dc in different animal models, journal of ethnopharmacology 138.
4.
cheney g, rapid healing of pepɵc
ulcers in paɵents receiving fresh cabbage juice. Calif med 1949;70(1):10-5.