Heterotopic Ossification Induced Autonomic Dysreflexia in Spinal Cord Injury: A Case Report and Review of the Literature
ABSTRACT
Spinal cord injury is a devastating neurological
condition that results in numerous complications. We present a case of a
17-year-old male who sustained a gunshot wound to the neck, resulting in a
complete spinal cord injury at T1 and a left brachial plexopathy. He developed
sudden onset of hypertension and facial flushing, consistent with autonomic
dysreflexia. Further evaluation
suggested that his autonomic dysreflexia was triggered by severe bilateral hip
heterotopic ossification (HO). This case report emphasizes the importance of
recognizing HO as a trigger for autonomic dysreflexia. Raising awareness about
HO and its imaging findings can broaden the differentials for potential
triggers of autonomic dysreflexia in individuals with spinal cord injury.
Keywords:
Spinal cord; Autonomic dysreflexia; Plexopathy; Heterotopic ossification
INTRODUCTION
Spinal cord injury (SCI) is a devastating neurological
condition that results in impaired motor and sensory functions at and below the
level of injury. Following SCI, numerous medical complications can develop,
including heterotopic ossification (HO). HO refers to the pathological
formation of bone within muscles and adjacent joints1. It is commonly observed as a complication
following SCI, traumatic brain injuries, burns, and major orthopaedic
surgeries. The incidence of developing HO ranges from 15 to 30% in cases of
combined SCI and polytrauma.1 Clinically significant HO, characterized by
restricted ROM that impacting function, occurs in approximately 10-20% of
cases, with 5-8% progressing to ankylosis2.
HO often presents with painful, swollen joints and
limited joint range of motion (ROM). We present a case of thoracic SCI from a
gunshot wound that developed autonomic dysreflexia (AD), and later it was found
his AD was triggered by HO. This case highlights the importance of awareness
and management of complications related to SCI.
CASE PRESENTATION
A 17-year-old male sustained a gunshot wound to the
neck resulting in a C6 transverse process fracture, C7 fracture, and retained
bullet fragments at T1 to T2, leading to a complete spinal cord injury (SCI) at
T1 and a left brachial plexopathy. His injuries were treated conservatively
with a spinal orthosis. He was subsequently transferred to an inpatient
rehabilitation unit, remaining paraplegic with no sensory or motor preservation
at and below T1. 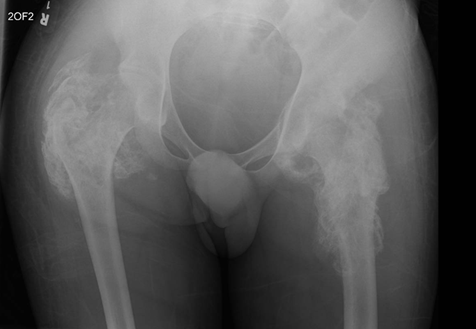
Figure 1.
Pelvic X-ray. Evidence of soft tissue calcification/ossification around
bilateral hip joints, indicating severe heterotopic ossification (HO).
At two months post-injury
during his rehabilitation unit stay, he was noted to have elevated blood
pressure (BP) at 200/120mmHg and facial flushing during the passive ranging of
his lower extremities. These symptoms were suggestive of autonomic dysreflexia
(AD). Common triggers for AD, including bladder distention, urinary tract
infection (UTI), or stool impaction, were excluded. Of note, patient was found
to have bilateral swollen hip joints with limited passive range of motion
(ROM). A pelvic X-ray and pelvic CT were therefore performed (Figure 1 and Figure 2). The pelvic
X-ray (Figure1) showed evidence of
soft tissue calcification/ossification around bilateral hip joints, indicating
severe heterotopic ossification (HO). The pelvic CT (Figure 2) in coronal view similarly depicted heterotopic
ossification involving bilateral hip joints. 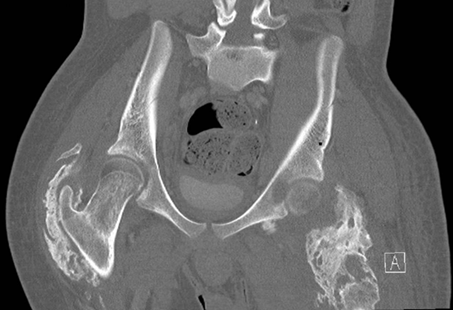
Figure 2.
Pelvic CT. CT in coronal view similarly depicted heterotopic ossification
involving bilateral hip joints.
Further laboratory investigations revealed elevated
alkaline phosphate (ALP) at 213 units/L (normal range 35-126 units/L) and a
mild elevation in C-reactive protein (CRP) at 4.9 mg/dl (normal range: ≤0.5
mg/dl). Calcium and phosphate levels were within normal limits. ALP levels were
regularly monitored, peaking at 3 months post-SCI, and gradually normalizing by
5 months post-SCI. Due to the functional limitations caused by HO and its
association with frequent AD, the patient was referred to Orthopaedic Surgery
for consideration of surgical resection.
DISCUSSION
The underlying mechanisms driving HO formation remain
incompletely elucidated. It is generally understood that the process is
initiated following a simultaneous central and peripheral injuries, which
stimulate bone formation at the site of peripheral injury through endochondral
ossification3. SCI prompts the
release of osteogenic and inflammatory factors. The influx of these factors
instigates the differentiation of osteoprogenitor cells (OPCs) into
fibroblasts, mediated by fibroblast growth factors (FGFs). This influx also
induces angiogenesis, leading to increased oxygen tension, which in turn
prompts OPC differentiation into chondrocytes. These chondrocytes undergo
hypertrophy and generate a cartilage matrix, providing a structural scaffold
for blood vessels formation, osteoblast proliferation and differentiation, and
ectopic bone formation1.
Joints frequently affected in SCI are the hip,
followed by the knee, the shoulder, and the elbow1,2.
Typically, HO most often developes between 1-6 months post SCI, with peak incidence
at 2 months4. Risk factors of HO
formation include male gender, smoking, complete injury, presence of pneumonia,
pressure injuries, urinary tract infections, and severe spasticity5. Clinical signs of HO include joint and muscle
pain, paraesthesia, tissue swelling in the involved region, fever, and
restricted ROM in the affected joints. AD is a common emergency complication of
SCI characterized by a sudden increase is blood pressure triggered by
peripheral stimulation, such as bladder distention6.
This case is distinguished by the notable presence of severe bilateral hip HO,
emphasizing the importance of recognizing HO as a potential trigger for
autonomic dysreflexia (AD).
Plain radiographs are frequently used for detecting
HO. The advantages of radiographs include the low cost and relative ease of
acquisition. However, they have limitation in visualizing the full extent of
ectopic bone deposition, especially in the early stages of the disease2. In the initial phases of HO, plain
radiographs may yield negative results. Triphasic bone scan offer earlier
detection capability, with positive uptake typically observed in the third
phase (static bone/ossification phase)7.
Bone scan is also used as a reliable indicator to determine the maturity of HO7. CT scans enhance preoperative planning by
providing three-dimensional visualization of HO in relation to important
anatomic landmarks2. ALP,
specifically bone alkaline phosphatase (BAP), is commonly utilized as a bone
turnover marker for monitoring HO progression after SCI8. Other biomarkers, such as C-reactive
protein(CRP), erythrocyte sedimentation rate (ESR), and creatine kinase (CK),
are also utilized. However, the above biomarkers lack specificity for HO, and
they are not effective for monitoring HO maturity9.
Several pharmacological agents have been investigated
for their efficacy in managing HO. Previous studies have demonstrated that
non-steroidal anti-inflammatory drugs (NSAIDs) can reduce the incidence of HO
when administered early after SCI.410,
but the side effects of NSAIDS need to be taken into consideration before
initiation of the treatment. Bisphosphonates have been tried in several studies
but there have been conflicting results with regards to the effectiveness in
treating HO11. Palovarotene, a
retinoic acid receptor (RAR-) agonist, has received approval by the US Food
and Drug Administration (FDA) for reducing the volume of new heterotopic
ossification in both adult and pediatric patients with fibrodysplasia
ossificans progressiva (also known as stone man disease)1. Research on the efficacy of palovarotene in
treating HO remains limited. Currently, there is no pharmacological treatment
available to reserve established HO formation. Surgical excision is considered
the most effective treatment approach once HO has developed1,3. However, a careful assessment of the risks
and benefits of surgery needs to be carried out when managing each individual
with this condition.
CONCLUSION
In conclusion, this unique case is distinguished by
the notable presence of severe bilateral hip HO, emphasizing the importance of
recognizing HO in imaging. Raising awareness about this condition can broaden
the differentials of potential triggers for AD identification in patients with
SCI.
REFERENCES
1. Wong
KR, Mychasiuk R, O'Brien TJ, Shultz SR, McDonald SJ, Brady RD. Neurological
heterotopic ossification: novel mechanisms, prognostic biomarkers and prophylactic
therapies. Bone Res 2020;8:20201209.
2. Ranganathan K, Loder S,
Agarwal S, et al. Heterotopic ossification: Basic-science principles and
clinical correlates. J Bone Joint Surg Am 2015;97(13):1101-1111.
3. Brady RD, Shultz SR,
McDonald SJ, O’Brien TJ. Neurological heterotopic ossification: Current
understanding and future directions. Bone 2018;109:35-42.
4. Banovac K, Williams JM,
Patrick LD, Haniff YM. Prevention of heterotopic ossification after spinal cord
injury with indomethacin. Spinal Cord 2001;39:370-374.
5. Yolcu YU, Wahood W, Goyal
A, et al. Factors associated with higher rates of heterotopic ossification
after spinal cord injury: A systematic review and meta-analysis. Clin Neurol
Neurosurg 2020;195:105821.
6. Calderon-Juarez M, Miller
T, Samejima S, et al. Heart rate variability-based prediction of autonomic
dysreflexia after spinal cord injury. J Neurotrauma 2024.
7. Campagnolo DI, Kirshblum S, Nash MS, et al. Spinal
cord medicine. Lippincott Williams & Wilkins 2011.
8. Ponzano M, Wiest MJ,
Coleman A, et al. The use of alkaline phosphatase as a bone turnover marker
after spinal cord injury: A scoping review of human and animal studies. J
Spinal Cord Med 2023;46(2):167-180.
9. Citak M, Grasmücke D,
Suero EM, et al. The roles of serum alkaline and bone alkaline phosphatase
levels in predicting heterotopic ossification following spinal cord injury.
Spinal Cord 2016;54:368-370.
10. Banovac K, Sherman AL,
Estores IM, Banovac F. Prevention and treatment of heterotopic ossification
after spinal cord injury. J Spinal Cord Med 2004;27:376-382.
11. Rizvi
S, Sharaf J, Williams KD, et al. Effectiveness of Prophylactic Interventions in
Neurogenic Heterotopic Ossification (NHO): A Systematic Review. Cureus 2022;14:e27683.