Involvement of the Central Nervous System in Plasma Cell Leukemia: Case Report
Abstract
Plasma
cell leukemia (PCL) is a rare malignant hematologic disease with an aggressive
and poor prognosis characterized by the uncontrolled proliferation of plasma
cells in the bone marrow and peripheral blood. Some clinical and laboratory
manifestations resemble multiple myeloma (MM), so the treatment of PCL is based
on therapies similar to those for MM. We present a case of a 47-year-old male
patient diagnosed primary plasma cell leukemia. The treatment was initiated
with VDT-PACE, despite the occurrence of complications and relapses, the
patient showed a good response to the proposed treatment, however after some
symptoms, the involvement of the disease in the central nervous system was
confirmed. Despite adequate monitoring and treatment, this type of complication
in patients with neurological symptoms still requires further investigation to
support therapeutic decisions.
Keywords: Plasma cell leukemia; Central nervous system
Introduction
Plasma cell leukemia (PCL) is a rare and highly aggressive hematological disease that is characterized by uncontrolled proliferation of plasma cells in the bone marrow and peripheral blood. PCL can be categorized as primary in most cases, or it can arise as a progression from pre-existing multiple myeloma (MM), designated as secondary plasma cell leukemia. Some clinical and laboratory manifestations resemble MM, such as genetic profile, proportion of cytogenetic abnormalities, appearance of tumor mass, impaired renal function, anemia, increased lactate dehydrogenase, and β2-microglobulin1-3. Worldwide, the annual incidence of PCL is around 0.04 cases per 100,000 individuals. Among patients with acute leukemia, the incidence of PCL reaches rates of approximately 0.9% and between 2-4% among patients with MM. According to the World Health Organization (WHO), among the plasma cell neoplasms, primary PCL represents 3-5% worldwide2,4. Recently, a significant increase in the number of cases of secondary PCL has been observed, which may be related to better survival of MM cases, as the longer the disease progresses, the greater the probability of developing PCL, for example, in the late and advanced stages of the initial disease5,6. PCL treatment has been adapted from MM, and it is based on bortezomib-like therapies, such as VDT-PACE (bortezomib, dexamethasone, thalidomide, cisplatin, adriamycin, cyclophosphamide and etoposide), VDT (bortezomib, thalidomide and dexamethasone), VRD (bortezomib, lenalidomide and dexamethasone), VCD (bortezomib, cyclophosphamide and dexamethasone), VAD (bortezomib, doxorubicin and dexamethasone) or VMP (bortezomib, melphalan and prednisone)6-8.
Case
report
A 47-year-old male patient was
admitted at the Oncology Center Oswaldo Leite at the Emergency Hospital of
Sergipe, on October 19th, 2021, presenting tachycardia, eupnea and
evident bruises on the upper and lower limbs. Patient was also found to have
palpable liver, weight loss of 10 kg in 2 months, bone pain, nausea, vomiting,
fever and sweating. On admission, the blood count demonstrated a hemoglobin of
11.5 g/dL and leukocytosis of 98,000/uL. Additionally, peripheral blood and
bone marrow smears revealed plasma cell morphology compatible with PCL, as seen
in (Figure 1).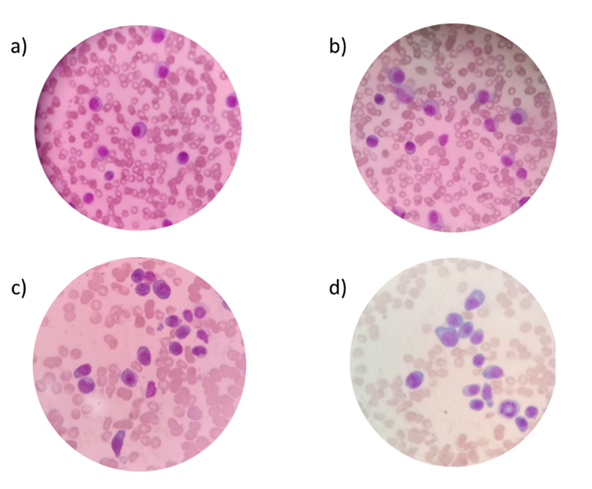
Figure 1: Morphological
analyzes of peripheral blood (a, b, c) and bone marrow smears (d) of the
patient showing malignant plasma cells. Photomicrograph of peripheral blood and
bone marrow cells on slides stained with May-Grünwald-Giemsa. Common optical
microscope. (100X magnification).
Immunophenotypic flow
cytometrics studies revealed 78.1% of monoclonal plasma cells with CD45-/partial CD38/partial
CD138/CD13+/CD28+/CD81+/partial CD117 phenotype, with Lambda light chain
expression being observed in 89.3% of the plasma cells, as seen in (Figure 2).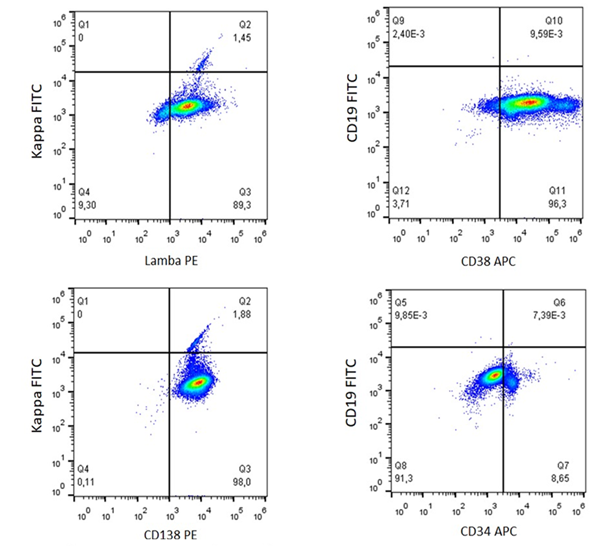
Figure 2: PCL with monoclonal plasma cells. Immunophenotyping by
flow cytometry demonstrates plasma cells with CD45-/partial CD38/partial
CD138/CD13+/CD28+/CD81+/partial CD117 phenotype and clonality observed through
lambda chain positivity in 89.3% of plasma cells.
The patient underwent a bone screening, which revealed no evidence of lytic lesions, but cervical spine radiography showed signs of bone rarefaction. An echocardiogram was performed, indicating preserved systolic function in the right ventricle, mild diastolic dysfunction and increased left atrium volume. Serological tests for hepatitis B and C, HIV and VDRL were all negative.
The patient underwent the
first of three cycles of the DV-PACE (dexamethasone, bortezomib, cisplatin,
cyclophosphamide, doxorubicin and etoposide) on October 26th, 2021.
Febrile neutropenia was corrected with cefepime and G-CSF. On March 16th,
2022, 2% of plasma cells were observed in the bone marrow, indicating complete
remission. Two months later, a new bone marrow examination confirmed recurrence
of the disease with the presence of 39% of plasma cells. Treatment was followed
with three cycles of CED-TAL (cyclophosphamide, etoposide, dexamethasone and
thalidomide), achieving complete remission again. In September 2022, the
patient was referred to collect peripheral stem cells for autologous
transplantation at the Real Hospital Português de Beneficência in Pernambuco
state, Brazil.
After the collection of the
cells, a new recurrence of the disease was detected, with 18% of plasma cells
in the peripheral blood and suspected infiltration in the central nervous
system (CNS), manifesting through peripheral facial paralysis, deviation of the
labial commissure to the right, and eyelid flaccidity. The patient's siblings
were invited to be evaluated as donors for allogeneic bone marrow
transplantation. On September 29th, 2022, the patient began
treatment with cyclophosphamide 300 mg/m2 and dexamethasone 20 mg/IV for
cytoreduction. On October 4th, 2022, CNS infiltration was confirmed,
with cerebrospinal fluid (CSF) showing 426 atypical plasma cells. On October 6th,
2022, the patient underwent cycle 1 (28/28 days) of DRD treatment (daratumumab,
lenalidomide and dexamethasone), followed by cycle 2 in November, cycle 3 in
December, cycle 4 in January, with concomitant administration of cytarabine,
dexamethasone and methotrexate, in four sessions of intrathecal chemotherapy.
After 16 months of overall survival, the patient died in February 2023.
Discussion
The case described in this
paper refers to primary plasma cell leukemia, a rare malignant hematologic
disease with an aggressive and poor prognosis. Despite new treatments
implemented in recent decades, there has still been no improvement in survival
rates when compared to other plasma cell neoplasms, such as multiple myeloma9,10. In addition to the classic features of PCL
diagnosis, patients commonly present anemia, leukocytosis and thrombocytopenia,
as well specific morphology of plasma cells, with a high nucleus/cytoplasm
ratio, basophilic cytoplasm, eccentric nucleus and cytoplasmic projections8,11.
Similar to the case cited by
Tuazon and collaborators (2021), the morphology of this case indicated similar
conditions in the peripheral blood (78.1% of clonal plasma cells), emphasizing
the need and importance of morphological evaluation of peripheral blood and
bone marrow as the initial laboratory assessment approach12. In addition to the characteristic
morphology, the immunophenotypic profile of B cell neoplasms definitely
contributes to the characterization of the cell lineage and maturation stage,
showing the expression of CD38 and CD138, and clonality of light chains, such
as the lambda chain in this case.
PCL has a very poor prognosis,
with an average overall survival of 4 months, and average rates of 28%, 14%,
and 6% at four months, one year, two years and five years, respectively9. In this case, despite the occurrence of
complications and relapses, the patient showed a good response to the proposed
treatment, with an overall survival of 16 months. Regardless of the therapeutic
advances, there is still no specific treatment protocol for PCL, and
therapeutic protocols used in the treatment of MM have been adapted, with the
aim of eradicating neoplastic plasma cells and reversing organ damage. In this
case, the patient's treatment was initiated with 4 cycles of VDT-PACE due to
the severe state of the disease, resulting in the patient's first complete
remission. Subsequently, there was the first relapse of the disease, with 38%
of plasma cells in the bone marrow, followed by a change to the CED-TAL
protocol. Finally, after proving the involvement of the disease in the central
nervous system, the patient began the DRD protocol with satisfactory initial
prospects for a therapeutic response.
Conclusion
Although the literature
describes few cases of CNS involvement in PCL, this case report demonstrated
that, despite adequate monitoring and rapid modification of the treatment
regimen, this type of complication in patients with neurological symptoms and rapid
progression of the disease still requires further investigation to support
therapeutic decisions. However, limited data to guide the treatment of relapsed
or refractory PCL demonstrate the need for prospective multicenter clinical
trials exploring more aggressive regimens. This may include chemotherapy
combination, immunomodulatory drugs, proteasome inhibitors, and other novel
compounds.
Acknowledgements: The authors would like to thank the professionals at
the Hospital de Urgências de Sergipe, involved in this study.
Conflicts of Interests:
This
manuscript has not been published, is not being considered for publication
elsewhere, and we have no conflicts of interest to disclose.
Funding: This study was supported by the Coordenação de
Aperfeiçoamento de Pessoal de Nível Superior- Brasil (CAPES).
Ethical Approval: The Human Ethics
Committee of the Federal University of Sergipe study approved protocol:
3.225.938.
References
1. Papadhimitriou
SI, Terpos E, Liapis K, et al. The cytogenetic profile of primary and secondary
plasma cell leukemia: etiopathogenetic perspectives, prognostic impact and
clinical relevance to newly diagnosed multiple myeloma with differential
circulating clonal plasma cells. Biomedicines 2022;10(2):209.
2. Gundesen
MT, Lund T, Moeller HEH, Abildgaard N. Plasma cell leukemia: Definition, presentation,
and treatment. Curr Oncol Rep. 2019;21(1):8.
3. Op Bruinink DH,
Kuiper R, van Duin M, et al. Identification of high-risk multiple myeloma with a
Plasma cell leukemia-like transcriptomic profile. J Clin Oncol
2022;40(27):3132-3150.
4. McKenna RW, Kyle RA, Kuehl WM, Gtogan
TM, Harris NL, Coupland RW. Plasma cell neoplasm. In: Swerdlow SH, Campo E,
Harris NL, et al. World Health Organization classification of tumors of
haematopoietic and lymphoid tissues 4th Edition. IARC Press 2008;2:200-213.
5. Fernández de
Larrea C, Kyle R, Rosiñol L, et al. Primary plasma cell leukemia: Consensus
definition by the International Myeloma Working Group according to peripheral
blood plasma cell percentage. Blood Cancer J 2021;11(12):192.
6. Rojas EA,
Gutiérrez NC. Genomics of plasma cell leukemia. Cancers (Basel) 2022;14(6):1594.
7. Jung
SH, Lee JJ. Update on primary plasma cell leukemia. Blood Res 2022;57:62-66.
8. Chauhan S, Jaisinghani
P, Rathore J, et al. Plasma cell leukemia. J Family Med Prim Care
2018;7(2):461-465.
9. Ramsingh G,
Mehan P, Luo J, Vij R, Morgensztern D. Primary plasma cell leukemia: A surveillance,
epidemiology, and end results database analysis between 1973 and 2004. Cancer
2009;115(24):5734-5739.
10. Nandakumar B,
Kumar SK, Dispenzieri A, et al. Clinical characteristics and outcomes of
patients with primary plasma cell leukemia in the era of novel agent therapy.
Mayo Clin Proc 2021;96(3):677-687.
11. Swaminathan
N, Varadi G. Secondary plasma cell leukemia: A case report. Cureus 2020;12(6):8693.
12. Tuazon SA,
Holmberg LA, Nadeem O, Richardson PG. A clinical perspective on plasma cell
leukemia; Current status and future directions. Blood Cancer J 2021;11(2):23.