It’s all in the History - Hemiplegic Migraine versus Stroke due to Arterial Dissection: A Case Report
Abstract
A 42-year-old right-handed woman with a history of hypertension and migraine presented with acute onset headache with left-sided weakness. Imaging showed right mca territory infarction with internal carotid artery (ica) dissection with circumferential luminal thickening into the petrous segment. A repeat angiogram five days later revealed new left vertebral artery dissection in v3 and v4 segments. She was managed with dual antiplatelet therapy and her symptoms improved after standard stroke care. We report this case to highlight the importance of taking a good history in patients with previous migraines presenting with acute headaches and focal neurology.
Introduction
Case presentation
A 42-year-old right-handed woman presented with a
gradual onset, persistent headache of one-day duration. She described this
headache as a ‘cold headache’ as if her head had been on ice and this was
different from her usual migraine headaches. This headache was associated with
binocular blurring and greying of vision. She was taken to her local acute
hospital where she was also found to have weakness in her left arm and leg. She
had never experienced limb weakness along with migraine headaches in the past. She
had a non-contrast ct head which was unremarkable. When she was about to be
discharged home with a presumed diagnosis of migraine attack, she developed
sudden onset left facial droop and slurring of speech. She was referred to our
regional specialist stroke center, for further evaluation and management.
Her past medical history included migraine and
hypertension. She was on ramipril and as required sumatriptan. She didn’t
smoke, had a moderate alcohol intake of 5 units per week and was not employed.
Her usual migraines were chronic in nature with a pulsating character and
visual aura of seeing flashing lights. There was no history of recent neck
trauma.
Upon our assessment, she was alert and orientated with
normal vital signs. The neurological deficits on our assessment were left
facial droop and left arm and leg weakness which contributed to the score of
nihss of 8. A ct angiogram (cta) of the aortic arch and carotids (and
intracranial vessels) was performed as all her symptoms were within 6 hours of
time of onset. The cta (figure 1) revealed right internal carotid artery
dissection 17 mm past carotid bifurcation. No large vessel occlusion of both
extracranial and intracranial vessels was found. 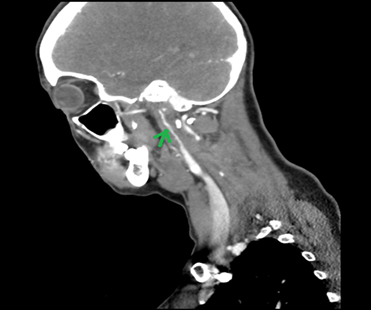
Figure 1: right ica
dissection on ct angio (arrow). Rat tail appearance due to reduced lumen size
of blood vessels because of dissection
She was admitted to the hyperacute stroke unit (hasu)
for further management. An mri head with angiogram (figure 2) was
performed which revealed acute right mca territory infarct in the region of
right insula, centrum semi-ovale and corona radiata in the right frontal lobe
and aneurysmal dilatation of the petrous segment of the right internal carotid
artery leading to possible occlusion of the right extracranial ica into the
petrous segment.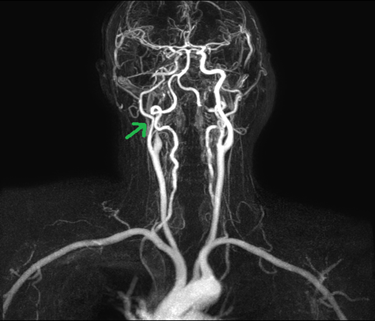
Figure 2: right ica
dissection on magnetic resonance angiogram (green arrow)
She was treated with dual antiplatelet therapy
(aspirin 75mg plus clopidogrel 75mg) with a follow-up plan of monitoring via
scans and optimizing the antiplatelet regimen as per scan findings. A repeat mr
angio 5 days later to monitor “pseudoaneurysm” showed left distal v3 segment
vertebral artery (va) dissection extending up to the border with v4. A
neurovascular mdt took place and it was advised that imaging is not suggestive
of pseudoaneurysm and the area of abnormality is likely an ica dissection flap
instead. The patient had no neck trauma or pain during her stay with us. The
right ica dissection appearances did not change.
Our patient also had a ct angiogram of a renal artery
which did not reveal fibromuscular dystrophy (fmd). The vasculitis screen came
back negative. The exact cause of these dissections remained unclear.
She was discharged home as she showed excellent
recovery. We reviewed her in the stroke outpatient clinic in 3 months and she
was doing very well. A repeat mr angiogram of carotids showed that the right
ica dissection and petrous segment ica dilation had resolved but left vertebral
artery dissection was still persisting. A further follow-up was planned with
repeat imaging and a plan to reduce the antiplatelet therapy to aspirin only if
improvement was found.
Discussion
About 10-25% of strokes in young and
middle-aged patients can be due to carotid artery dissection. The symptoms may
vary and include headache, horner’s syndrome, tinnitus and features suggestive
of a transient ischemic attack1. Incidence2 of spontaneous cad is 2 to 3 in 100,000 per
year and bilateral cad is even less as evidenced by only a few case reports in
literature.
The risk factors3 for cad include minor trauma, migraine, genetic
predisposition, connective tissue disorders like ehlers-danlos syndrome, marfan
syndrome and a recent acute infection4. Outcomes in carotid artery dissection (cad) are usually
good with mortality less than 5%5. Our patient denied any neck trauma, familial features or recreational
drug misuse.
There is no single agreed-upon medical
treatment strategy for cad management. A randomized trial, cadiss6 investigated the difference of outcomes of cad in 250
patients comparing the use of antiplatelet therapy and anticoagulation therapy.
The trial found no difference in efficacy between the two drug groups in terms
of preventing stroke and death in both groups, the trial was however not
without limitations. As per a review article by zafer, et al7, dual antiplatelet therapy is indicated for extracranial cad
for 3 months followed by single antiplatelet therapy for life long. Our patient
was treated with dual antiplatelet therapy with the plan of reducing to single
antiplatelet therapy and later follow-ups in the stroke clinic.
There are only a few similar case reports in
the literature which have indicated patients having cad masked under migraine.
As per a case report by sharif et al which includes a literature review,
features of a migraine which should prompt consideration of the diagnosis of
cad are a painful horner’s syndrome, an incomplete horner’s syndrome (miosis,
ptosis but no anhidrosis), tinnitus, visual scintillations and cranial nerve
palsies8. Our patient had clinical features of a
headache not like her usual migraine headaches, also had left facial droop,
slurring of speech and left arm weakness. Consideration was given to hemiplegic
migraine as a potential diagnosis prior to the scans.
Mr angiography is the preferred choice1 for
diagnosis of cad but ct angiogram can be used if there are any
contraindications for mr scans. Carotid duplex scan (cuss) should only be used
as a screening tool as it has poor detection of cad near the skull base and
transverse foramina9.
An mdt approach is recommended for deciding the
treatment of carotid artery dissection. Patients with cad can present with
focal neurologic deficits due to ischemia (thromboembolism or arterial
occlusion) or subarachnoid hemorrhage (pseudoaneurysm formation and rupture)10.
Overall, our patient showed good improvement
after receiving medical management and rehabilitation. Headache and weakness
resolved significantly and she was followed up in an outpatient clinic11.
Our case study had limitations. There is not
much follow up data available for such patients. There is lack of studies in
literature clarifying use of single vs dual antiplatelet therapy in such
patients. More studies are required to add to the body of knowledge on cad
patient management.
Conclusion
References
1. debette
s and leys d. Cervical-artery dissections: predisposing factors, diagnosis and
outcome. The lancet neurology 2009;8(7):668-678.
2. lee vh, brown jr rd,
mandrekar jn and mokri b. Incidence and outcome of cervical artery dissection:
a population-based study. Neurology 2006;67(10):1809-1812.
3. debette s and leys d.
Cervical-artery dissections: predisposing factors, diagnosis and outcome. The
lancet neurology 2009;8(7):668-678.
4. guillon b, berthet k,
benslamia l, bertrand m, bousser mg and tzourio c. Infection and the risk of
spontaneous cervical artery dissection: a case-control study. Stroke 2003;34(7):79-81.
5. lee vh, brown jr rd,
mandrekar jn and mokri b. Incidence and outcome of cervical artery dissection:
a population-based study. Neurology 2006;67(10):1809-1812.
6. Macleod m,
colam b and salman ras. Antiplatelet treatment compared with anticoagulation
treatment for cervical artery dissection (cadiss): a randomized trial. Lancet
neurology 2015;14(6):566.
7. keser z, meschia jf and
lanzino g. Craniocervical artery dissections: a concise review for clinicians.
In mayo clinic proceedings 2022;97(4):777-783.
8. sharif m, trinick t and
khan kh. Identification of internal carotid artery dissection in patients with
migraine--case report and literature review. Jpma. The journal of the pakistan
medical association 2010;60(2):131-133.
9. provenzale jm. Mri and
mra for evaluation of dissection of craniocerebral arteries: lessons from the
medical literature. Emergency radiol 2009;16:185-193.
10. murai
y, shirokane k, kitamura t, tateyama k, matano f, mizunari t and morita a.
Petrous internal carotid artery aneurysm: a systematic review. Journal of
nippon medical school 2020;87(4):172-183.
11. donnelly
a, sinnott b, boyle r and rennie i. Beware the middle-aged migraine: internal
carotid artery dissection mimicking migraine in the emergency department. Case
reports 2017.