Maculopathy and Interstitial Cystitis: An Underappreciated Association of Pentosan Polysulfate Sodium
Keywords: maculopathy; interstitial cystitis; bladder pain syndrome
1. Introduction
1.1 background
Interstitial
cystitis is common1,2, affecting up to 6.5% of women in the united states
(us).curhan et al3
demonstrated that up to 70 cases per 100,000 women in the us have ic. In europe
and japan, the numbers are approximately 18 and 4 cases per 100,000 women
respectively. This is likely due to differences in diagnostic criteria. In the
uk, as many as 400,000 people are affected, with 90% being women4.
Pps
is an established treatment for ic, especially in the us. It is often used
after the failure of conservative management, antispasmodic/antimuscarincs,
non-narcotic analgesics, tricyclic anti-depressants and h2 antagonists2. Pps is the only medication specific to ic and
is a last line of treatment. Pps related maculopathy is a significant and
irreversible ophthalmicmorbidity. The chances of developing it are cumulatively
dose dependent. This means thatpatients on a standard dose will eventually
reach at leasta 41.7% risk of having maculopathy5-7.
We report a case of maculopathy occurring within 6 weeks of pps initiation. This case demonstrates that whilst evidence suggests maculopathy is generally cumulative dose dependent, maculopathy can still occur early.
2. Case presentation
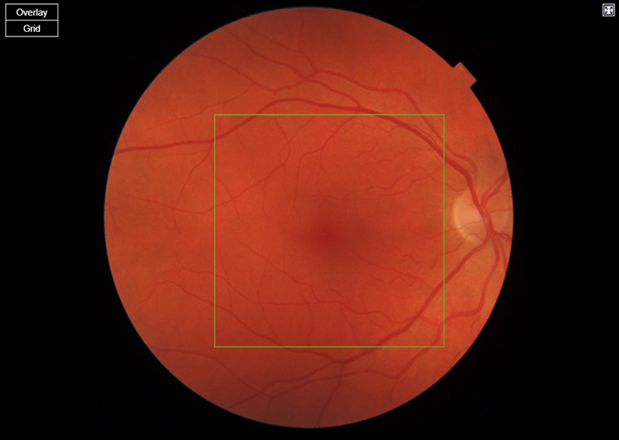
(a) Right eye
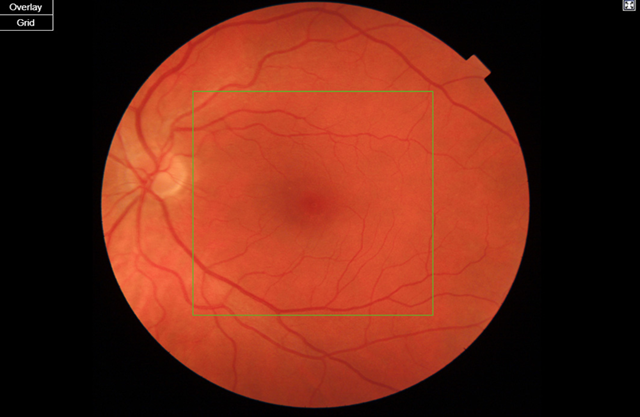
(a) Left eye
figure 1. Standard fundus photos
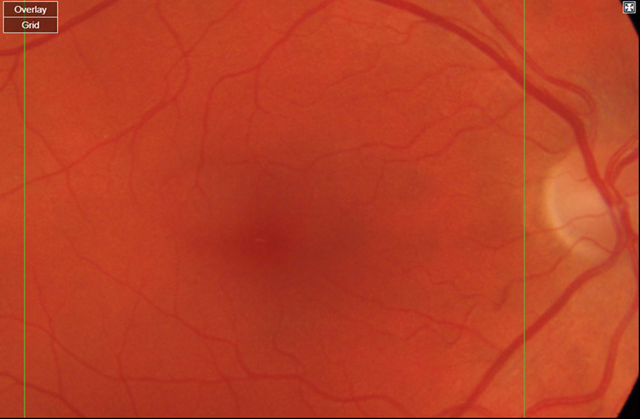
(a) Right eye
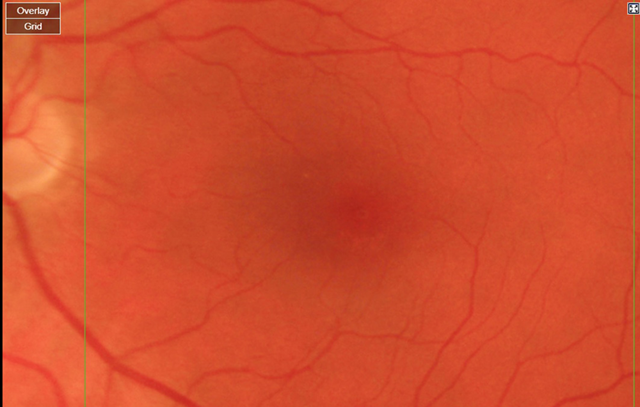
(a) Left eye
Figure 2. Magnified standard fundus photos
2.1 investigations if
relevant
Optical
coherence tomography (oct) is a non-invasive imaging modality that uses
low-coherence light to capture micrometer resolution images of the retina.
Using the information obtained, two and three dimensional images of the retina
are formed which the clinician can use to give an indication of retinal/ rpe
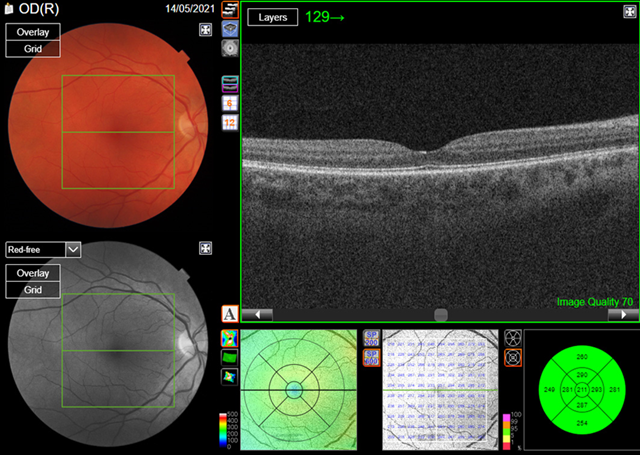
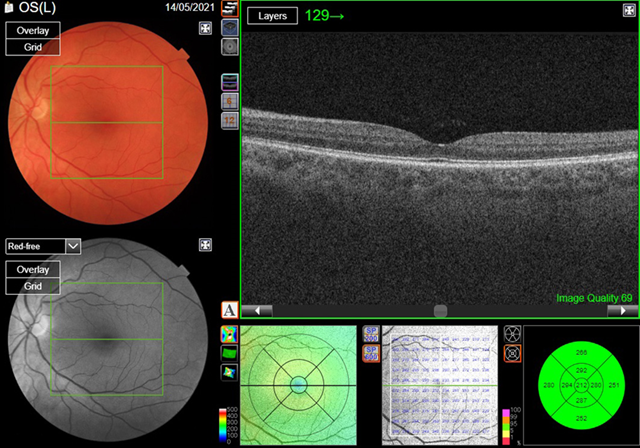
health. The case study patient’s macula oct scans were normal (figures 3a,3b).
(a) Right eye
(a) Left eye
Figure
3.
Optical coherence tomography
(oct)
Fundus
autofluorescence (faf) is a non-invasive imaging modality that captures an
image from ocular endogenous fluorophores found within the retinal pigment
epithelium and the choroid. These are composed mainly of lipofuscin and
melanin. By capturing an image composed
of the distribution of lipofuscin and melanin, the clinician can formulate an
impression of the health of the retina/rpe.
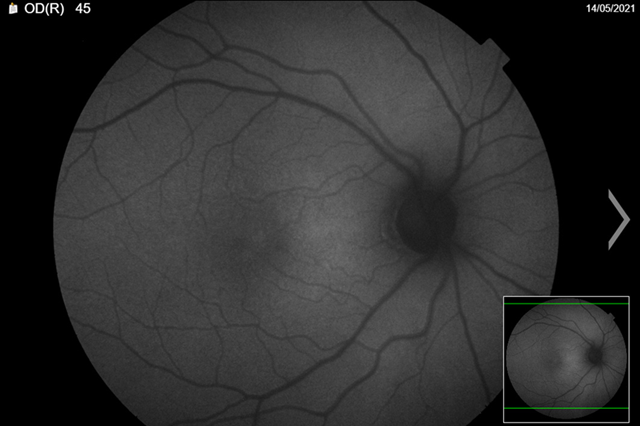
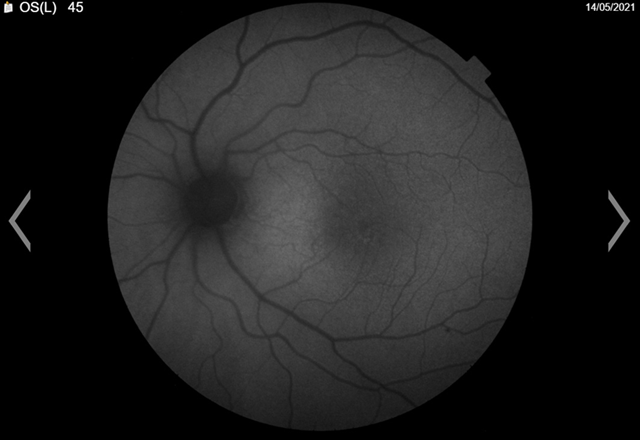
The patient’s faf was abnormal. We could see from the images that there were areas of hyper and hypo-autofluorescent spots circumferentially, with the left more defined than right (figures 4a,4b). Magnified images showing defects more clearly (figures 5a,5b).
(a) Right eye
(a)
Left eye
figure 4. Fundus autofluorescence (faf)
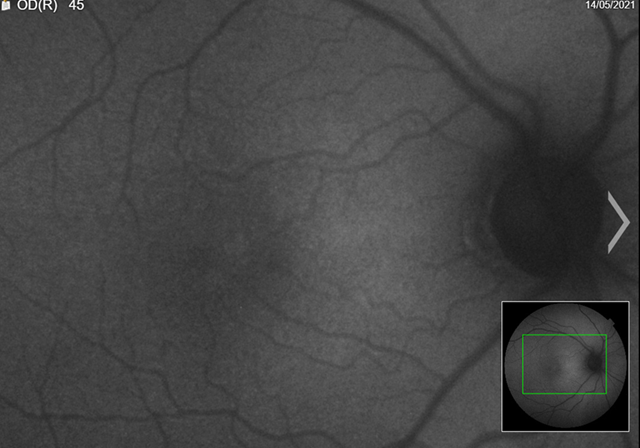
(a) Right eye
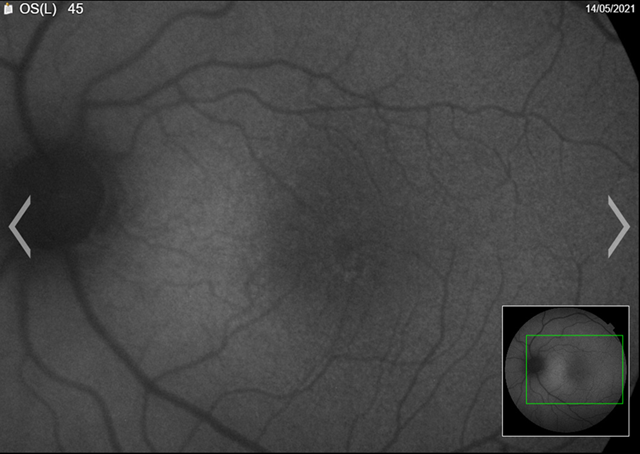
(a) Left eye
Figure 5. Fundus autofluorescence (faf) (magnified)
Visual
field testing was obtained to assess the sensitivity of the macula area. They
demonstrated very mild central field loss (figures
6a,6b).

(a)
Right eye
(a)
Left eye
figure 6. Visual
field testing vf 10-2
1.
Differential diagnosis if relevant
Pps
maculopathy has a few differential diagnoses that we considered. These
aremacular dystrophies, age related macular degeneration (armd), pachychoroid
pigment epitheliopathy and maternally inherited diabetes and deafness syndrome
(midd)18.
It is
quite simple in this case to exclude armd. The patient does not fit the general
age criteria for development, there was no family history of armd and the faf
pattern suggested a toxic maculopathy rather than armd9. Additionally, the hyper-autofluorescent
lesions observed in this patient were distinct from the expected retinal
oedema, drusen and subretinal drusenoid deposits normally seen in armd. The
patient’s lesions wereat the level of the rpe and they caused no interference
when visualising the choroid.
Inherited
macular and pattern dystrophies often present with less densely packed faf
lesions than pps maculopathy10.the
patient’s lesions were not typical of a diagnosis of inherited dystrophy and
there was no family history of this.
Midd
was ruled out because unlike pps, the pigmentary maculopathy characteristics of
midd do not involve the fovea until late stage disease. Also midd with macular
changes most often presents with additional systemic manifestations such as
diabetes and deafness11.the patient
did not have any associated systemic manifestations.
Pachychoroid
diseases can present with similar macular changes to pps maculopathy. However,
the clinician would expect additional findings such as dilated choroidal
vessels, rpe and neurosensory retinal detachments and streaks representing
previous exudation on faf12.
Given
that the patient has no prior history or signs of the above discussed
differential diagnosis, as well as recent initiation of pps therapy and a
symptomatic scotoma despite normal bcva, we can confidently assume a diagnosis
of pps maculopathy.
2.
Treatment if relevant
Currently
there is no treatment for pps maculopathy. Prescribing clinicians are advised
to avoid or minimize cumulative exposure to pps as primary prevention. When
prescribing pps, clinicians should discuss the risks of vision loss associated
with pps and its dose dependant nature with the patient. This will then lead to
an informed decision as to whether the benefits outweigh the risk. Should pps
be recommended and agreed with the patient, the clinician should prescribe the
minimum dose and length of time for ic management.
Additional
to pps maculopathy, pps can also cause cystoid macular oedema (cmo) and macular
neovascularisation (mnv). Cmo can be treated with topical therapy (carbonic
anhydrase inhibitors, corticosteroids or nsaids), oral therapy (diamox) or
intravitreal therapy. Both cmo and mnv can be treated with anti-vascular
endothelial growth factor (anti-vegf).
In
the event of pps maculopathy, cmo or mnv, the diagnosing clinician should make
the prescribing clinician aware. The prescribing clinician should cease pps
then transition to other therapies. Patients with pps maculopathy should
ideally be monitored annually with multimodal imaging (colour fundus
photography, faf and oct) in the event that treatable ophthalmic sequelae of
pps occur. Cessation of pps may not prevent progression of maculopathy13,14.
3.
Outcome and follow-up
This
patient was advised to stop pps and referred back to her prescribing clinician
for alternative management of ic. A medical retina appointment was requested
for 6 weeks with multimodal imaging to assess for progression of her
maculopathy due to the rapidity of onset of her condition.
4.
Discussion
As
mentioned before, pps maculopathy has no established risk beyond cumulative pps
drug exposure. Patients may have normal best corrected visual acuity (bcva)
with symptomatic scotoma.
What
can be observed is a disruption to the rpe/ photoreceptor interface. As seen in
our case, early disease is defined by para-fovealmultifocal atrophy. In time
this may coalesce and involve the centre15.
Rpe atrophy is a sequelae of advanced disease11,15.
There
are no established guidelines for the treatment and monitoring of pps
maculopathy as this is still a novel area.
Cmo
can be treated with both topical and intravitreal therapies11,16. Mnv can be treated with anti-vegf17.
In
terms of prognosis, longer terms studies and more data is needed to predict
this novel condition. However, there is no evidence to suggest pps maculopathy
is reversible. The fact that it can also progress after drug cessation is a
point of concern and any suspected case should be taken seriously13,14.
Early
detection is important to prevent permanent and progressive sequelae of pps
maculopathy.
Wang et al15 recommend an initial exam within 6 months of pps initiation and then annual exams as the patient approaches 500g of cumulative exposure. Based on our case, we would recommend a baseline examination within 1 month of starting pps treatment followed by annual review depending on initial observations.
5.
Learning points/take home messages
•
please take into consideration alternative and safer therapies for ic. We would
recommend only using pps as a last line of treatment in otherwise refractory
cases.
• use the minimum dose and length of time of treatment to achieve
therapeutic value
• to
be extremely sensitive to new onset scotoma despite normal bcva
•
have a low threshold for referral to ophthalmology after commencement of pps
6.
Patients perspective
Shortly
after starting pps i became aware of some disturbance of my central vision,
especially in the left eye. The eye care professionals confirmed a connection
between my medicine and my symptoms. After stopping the treatment my vision did
not worsen.
I am glad i was able to avoid losing any more vision. I hope that my case raises awareness of this condition for other patients and professionals.
References
4.
The urology foundation bladder related
statistics. 2021.
18.
Aaron l, adam m, nierah j. Pentosan
polysulfate maculopathy. Surv ophthalmol 2021;67(1):83-96.