Malignant Pleural Effusion with Metastasis to the Pericardial Sac
Abstract
Malignant pericardial effusion is commonly linked to occult carcinoma,
most frequently discovered as disease progression from primary lung cancer.
Pericardial involvement typically remains undiagnosed, with only 1-20% of
tumor-related autopsies documenting invasion of the pericardium. We present a
56-year-old male with massive pericardial effusion, uncovering a diagnosis of
primary adenocarcinoma of the lung. Initially, he presented to the emergency
department with shortness of breath. Past medical history includes chronic
obstructive pulmonary disease and a 45 pack-year smoking history. A
computerized tomography angiography of the chest was performed, revealing a
possible left lower lobe pulmonary embolism. A large pericardial effusion was
also found, with evidence of cardiac tamponade. He was emergently taken to the
cardiac catheterization lab, where a pericardiocentesis was performed and 850
ml of hemorrhagic fluid was evacuated. Cytology of fluid from the left pleural
cavity was performed. This specimen was found to contain numerous cohesive
groups of metastatic adenocarcinoma cells. Pericardial sac tissue biopsy
demonstrated non-small cell lung carcinoma (nsclc) adenocarcinoma.
Immunohistochemical staining demonstrated ttf-1, ck7, and ck20 positivity,
which confirmed primary pulmonary adenocarcinoma. Oncology was consulted and
the patient was diagnosed with stage iva nsclc. Systemic chemotherapy was
started with carboplatin, pemetrexed and pembrolizumab. Malignant pericardial
effusions, although relatively rare, carry grave implications for patients,
often manifesting with varied clinical presentations that demand prompt medical
interventions. The patient's journey through diagnosis and treatment serves as
a reminder of the challenges posed by advanced stage malignancies and their
propensity to affect multiple organ systems, necessitating a multidisciplinary
approach.
Keywords: malignant pericardial effusion; metastasis; pericardial sac
Introduction
Malignant pericardial effusion is commonly
linked to occult carcinoma, most frequently discovered as disease progression
from primary lung cancer. Pericardial involvement typically remains
undiagnosed, with only 1-20% of tumor-related autopsies documenting invasion of
the pericardium. Presentation of malignant pericardial effusions vary and can
range from patients experiencing difficulty breathing, chest pain, cardiac
arrhythmias, to life threatening cardiac tamponade1.
Although emergent procedures such as pericardiocentesis and surgical procedures
such as pericardial window can correct cardiac tamponade, prognosis for these
patients remains guarded due to a primary malignancy being the cause of these
complications. Non-small cell lung cancer (nsclc) has been reported as the most
frequent malignancy related to malignant pericardial effusion; this case report
will describe a patient with primary lung adenocarcinoma presenting with
significant pericardial effusion.
Case presentation:
A 56-year-old male
presented to the emergency department (ed) with shortness of breath. He
reported that his shortness of breath has been on-going from the past 3 months,
and recently progressively worsened. One day prior to his ed visit, he reported
that he could not walk up one flight of stairs without becoming severely short
of breath. At this time, he reported having mild lightheadedness, but denied
chest pain. Past medical history includes chronic obstructive pulmonary disease
(copd) and a 45 pack-year smoking history. Abnormal laboratory findings are
represented in (table 1).
Table 1. Patient’s abnormal lab values; all other values were within normal limits.
|
Lab being evaluated: |
Patient’s lab values: |
Reference values: |
|
Sodium |
134 mmol/l |
135-144 mmol/l |
|
Glucose |
159 mg/dl |
70-99 mg/dl |
|
Alt (sgpt) |
58 unit/l |
7-52 unit/l |
|
Lipase |
9 u/l |
0-160 u/l |
|
Hematocrit |
38.0 % |
39-50% |
|
Neutrophils, relative |
80.7% |
45-80% |
|
Lymphocytes, relative |
8.5% |
12-45% |
|
Lymphocytes, absolute |
0.79 k/mcl |
1.00-3.50k/mcl |
While being examined in the ed, the patient's pulse oximetry declined to 60% and decreased breath sounds were noted to the lung bases bilaterally. Initial chest x-ray (cxr) seen in (figure 1) revealed a mild patchy alveolar airspace opacity in the posterior left lower lobe, suspicious for pneumonia. The patient’s respiration rate was 23 breaths per minute, however, he was speaking comfortably on a nonrebreather mask; endotracheal intubation was not indicated at this time due to preserved respiratory effort. He began having right-sided chest pain. A cardiac rhythm strip at that time revealed a tachycardic rate of 110 beats per minute. Arterial blood gas (abg) was performed; the patient's oxygen level was 99%, po2 of 260, pco2 of 25, and lactic acid level of 4.0. Given his worsening status, additional imaging was ordered. While in the ed, a computerized tomography angiography (cta) of the chest was performed. Revealing sub segmental right upper lobe and possible segmental left lower lobe pulmonary embolism, see (figure 2). A large pericardial effusion was also found, with evidence of cardiac tamponade. Subsequently, the patient developed cardiogenic shock with a secondary acute kidney injury requiring the administration of vasopressors.
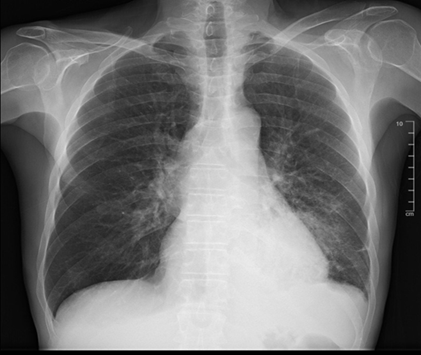
Figure 1. Initial cxr in ed; a mild patchy alveolar airspace opacity in the posterior left lower lobe, suspicious for pneumonia.
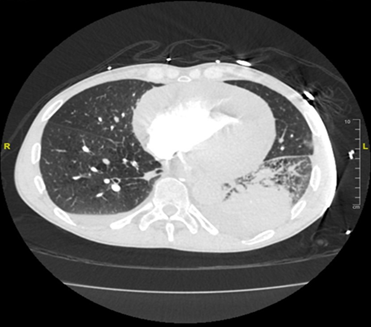
Figure 2. Cta of the chest; an acute pulmonary embolism was noted in the right upper segmental branch and the left lower segmental branch. The main pulmonary artery appeared to be non-dilated. A large area of consolidation was found within the left lower lobe, along with a small left pleural effusion and a trace effusion on the right, likely representing pneumonia. A 1-cm ill-defined focal ground glass nodule was found in the right upper lobe. Trace mucosal layering along the left main pulmonary artery was noted, otherwise the central pulmonary arteries were deemed clear. A large pericardial effusion measuring approximately 4.2 cm was found along the inferior border with moderate reflux of contrast into the hepatic veins.
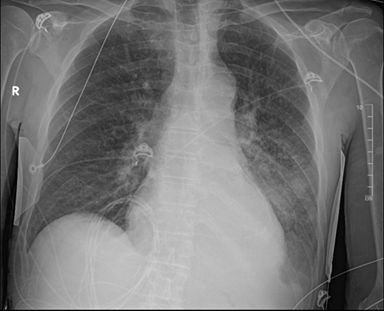
Figure 3. First cxr after drain placement; pericardial drain is overlying the cardiac silhouette. No significant change in the large left lower lobe area of airspace opacification, likely due to pneumonia or underlying neoplasm. Interval development of a left pleural effusion, and mild diffuse interstitial opacities, which could be reflective of mild interstitial pulmonary edema was noted. No pneumothorax or right pleural effusion was appreciated.
He was emergently taken to the cardiac catheterization lab, where a pericardiocentesis was performed and 850 ml of hemorrhagic fluid was evacuated. A pericardial drain was placed at this time, and vitals remained stable. The internal medicine service became concerned that the pericardial drain may be clotted as it was not draining output as expected. Initial cxr post drain placement demonstrated no significant change to the left lower lobe, likely reflecting pneumonia or an underlying neoplasm; see (figure 3). Cardiothoracic surgery was then consulted for evaluation of a pericardial window. The patient was admitted to the cardiac care unit (ccu) and started on intravenous heparin.
Cardiothoracic surgery was then consulted for evaluation of a pericardial window. The patient was admitted to the cardiac care unit (ccu) and started on intravenous heparin. Over the next 48 hours, the patient reported feeling persistent chest discomfort, which worsened with inspirations and coughing. At this time, the patient was breathing unassisted, on room air. Although a pericardial drain was still in place, repeat echocardiogram showed evidence of residual pericardial effusion. A pericardial window was performed, and postoperative pericardial drainage was recorded; see (table 2).
Table 2. Pericardial fluid drainage from drain; drainage recorded up to six days postoperative pericardial window.
|
Postoperative
day from pericardial window |
Pericardial
fluid drainage collected |
|
Day 1 |
160 cc |
|
Day 2 |
110 cc |
|
Day 3 |
275 cc |
|
Day 4 |
180 cc |
|
Day 5 |
160 cc |
|
Day 6 |
105 cc |
While performing thoracentesis of the left pleural cavity, 10
ml of translucent orange pleural fluid was collected. This specimen was found
to contain numerous cohesive groups of metastatic adenocarcinoma cells. A
pericardial sac tissue biopsy was also performed. Immunohistochemically
staining of this specimen demonstrated ttf-1, ck7, and ck20 positivity, which
differentiate primary pulmonary adenocarcinomas from extra pulmonary
adenocarcinomas that have metastasized to the lung.
The patient was consulted by hematology-oncology and a brain
mri was ordered, which was negative for brain metastasis. Our patient was
subsequently diagnosed with stage iva nsclc. Whole-body pet scan was ordered
upon discharge from the hospital which showed an enlarging, hypermetabolic mass
like consolidation in the left lower lobe, hypermetabolic cervical, left
supraclavicular, mediastinal, hilar and sub carinal lymphadenopathy, likely
metastatic, and multiple discrete foci of increased anechoic activity in the
pelvis and cervical spine which that were also like metastatic. Systemic
chemotherapy was started with carboplatin and pemetrexed, in combination with
pembrolizumab. Cyanocobalamin 1000 mcg was given intramuscularly prior to
starting chemotherapy. This regimen was adjusted accordingly following a brief
elevation of liver transaminases.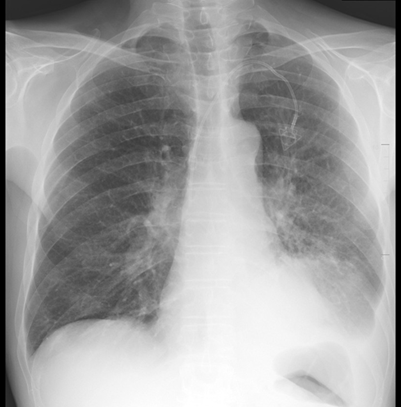
Figure 4. First cxr
post-discharge; left subclavian chest port with the tip in the svc. The heart
is stable in size. There is pulmonary venous congestion without overt edema.
Redemonstrated is a large mass like opacity in the left lower lobe with a small
left pleural effusion. No pneumothorax. The right lung is clear.
Upon discharge, he was educated on the importance of taking
his medications for copd, specifically his albuterol and ipratropium
nebulizers. Four days after discharge from our facility, he received a
single-lumen power port vascular catheter through the left subclavian vein for
chemotherapy. An outpatient cxr showed mild pulmonary venous congestion without
overt edema with a stable, large, mass-like opacity in the left lower lobe. Our
patient was given close follow up with hematology-oncology, pulmonology, and
primary care to optimize his management for stage iva nsclc.
Discussion
Pericardial effusion is defined as a collection of fluid in
the pericardial sac, located around the heart. In healthy patients, the
pericardial sac contains approximately 15 to 50 ml of serous fluid which serves
to reduce friction between the pericardial membranes allowing them to glide
smoothly among each other while the heart is contracting and relaxing2. The
management of pericardial effusions depends on size of the effusion,
hemodynamic compromise, and the primary etiology of the effusion. In developed
countries, most pericardial effusions are idiopathic, however, in
underdeveloped countries, tuberculosis (tb) has been identified as the most
common cause of this problem. Disease states that have been identified to cause
pericardial effusions include neoplasm, systemic lupus erythematosus (sle),
bacterial and viral infections such as chagas disease, dressler syndrome, and chronic
renal failure requiring dialysis. Hydralazine and procainamide have been
identified as medications that can also cause this problem1.
Fluid accumulation, inflammation, and fibrous constriction
are among the most common forms of pericardial disease. These conditions are
usually associated with other cardiac pathologies or systemic disease.
Normally, less than 50 ml of thin, clear, straw-colored fluid is present within
the pericardial sac. In conditions that permit long-standing cardiac
enlargement or allow for a slow accumulation of fluid, the pericardium has
adequate time to remodel and adjust to the larger volume. This process permits the
development of a slow accumulating pericardial effusion to be very substantial
without restricting or compromising cardiac function. Most cases of chronic
effusion (usually less than 500 ml of fluid) only become clinically significant
once identified as globular enlargement of the heart shadow on chest
radiography. However, rapidly developing fluid collections of only
approximately 200-300 ml due to hemopericardium caused by ruptured myocardial
infarction or aortic dissection, can present with significant compression of
the atria and venae cavae. Rapid developing pericardial effusion can be much
more clinically devastating by compromising cardiac filling, leading to life-threatening
cardiac tamponade3.
Malignant pericardial effusion remains a rare presentation in
patients with undiagnosed malignancy. The most prevalent cancer identified as a
cause of malignant pericardial effusions is lung cancer, and breast cancer as
the second most prevalent. Other common malignancies such as melanoma,
mesothelioma, mediastinal lymphoma, and renal cell carcinoma have been seen to
cause malignant pericardial effusion. It is believed that the pathogenesis of
malignant pericardial effusion is due to local lymphatic invasion. Direct
invasion into the mediastinum is thought to occur less frequently and believed
to be due to hematogenous spread1.
Pathologic examination of the pericardial and pleural fluid
showed similar findings, both revealing numerous groups of metastatic
adenocarcinoma cells. Immunohistochemistry (ihc) testing then redemonstrated
ttf-1, ck7 and ck20 positivity which are known to be specific for primary
pulmonary adenocarcinoma4. These ihc results ruled out
extrapulmonary sources for primary malignancy, therefore reinforcing the
diagnosis of metastatic adenocarcinoma of the lung; see (table 3) for details.
Adenocarcinoma of the lung, a prevalent subtype of non-small
cell lung cancer, originates in the lung's epithelial cells and is
characterized by glandular structures. Commonly associated with genetic
mutations like egfr, alk rearrangements, and kras mutations, adenocarcinoma
often presents in nonsmokers or former light smokers, predominantly in the
lung's outer regions. Its diverse histological patterns, including acinar,
papillary, and solid formations, contribute to its varied clinical
manifestations such as persistent cough, shortness of breath, and weight loss.
Diagnosis involves imaging, biopsy, and molecular testing to determine
treatment options, which span surgery, chemotherapy, radiation, targeted
therapy, and immunotherapy, with personalized strategies becoming increasingly
essential3.
Table 3.
Immunohistochemical details; our patients ihc results showed positive staining
for ttf-1, ck7, and ck205,6.
|
Stain |
Definition |
Associated
pathologies |
|
Ttf-15 |
Thyroid transcription
factor 1, also known as nkx2-1, homeodomain containing transcription factor |
Preferential
expression in thyroid, lung and brain structures of diencephalic origin |
|
Ck76 |
Cytokeratin 7, type
ii keratin of nonkeratinized epithelium |
Adenocarcinoma of
lung, breast, thyroid |
|
Ck206 |
Cytokeratin 20,
epithelial marker |
Lung-mucinous
bronchioloalveolar carcinomas, urothelial carcinomas |
Previously reported estimates of the incidence of cardiac metastasis detected in vivo were less than 1% of patients. However, most recent cadaver studies showed an increase of incidence to 2-18%. Incidence of metastasis to the heart and pericardium greatly exceeds the incidence of primary cardiac neoplasms, approximately 40:17. Metastatic tumors of the heart have been identified in approximately 5% of terminally-ill cancer patients. The most frequent cancers seen to have metastatic cardiac tumors are lung cancer, breast cancer, melanoma, leukemia, and lymphoma. Metastases reach the heart and pericardium via retrograde lymphatic extensions from the mediastinum; this process can occur via hematogenous seeding, direct continuous extension, or by venous extension. The clinical symptoms of these conditions present with pericardial effusion, indicating that pericardial spread has successfully occurred. Myocardial metastases are relatively clinically silent, but can cause nonspecific structural heart problems such as generalized defects in ventricular contraction and compliance, usually resulting in an arrhythmia. Bronchogenic carcinoma and malignant lymphoma tend to infiltrate the mediastinum, leading to compression and invasion into the superior vena cava. This causes an obstruction to blood coming from the head and upper extremities, resulting in superior vena cava syndrome. Lastly, renal cell carcinoma typically invades the renal vein and continues to grow upward into the inferior vena cava lumen directly into the right atrium, resulting in a blockage of venous return to the heart3.
Conclusion
In conclusion, this case of the 56-year-old male with primary lung adenocarcinoma presenting as a significant malignant pericardial effusion underscores the intricate interplay between oncology and cardiology. Malignant pericardial effusions, although relatively rare, carry grave implications for patients, often manifesting with varied clinical presentations that demand prompt and precise medical interventions. This case highlights the importance of recognizing the potential cardiac complications of underlying malignancies, particularly in patients with risk factors such as a history of smoking and chronic obstructive pulmonary disease. The patient's journey through diagnosis and treatment serves as a reminder of the challenges posed by advanced stage malignancies and their propensity to affect multiple organ systems, necessitating a multidisciplinary approach. Diagnostic tools, including imaging techniques and immunohistochemically staining, played a pivotal role in confirming the primary source of metastatic adenocarcinoma, thus guiding treatment decisions. The initiation of systemic chemotherapy, coupled with careful monitoring and adjustments based on the patient's response and tolerability, demonstrates the dynamic nature of oncology treatment regimens. Furthermore, the case highlights the critical role of pericardiocentesis and pericardial window procedures in managing cardiac tamponade, providing immediate relief and stabilizing the patient's condition. The interaction between medical specialties, evident through consultations with internal medicine, oncology, and cardiothoracic surgery, exemplifies the collaborative effort required to address the complexities of such cases comprehensively. As medical knowledge and technology continue to advance, the insights gained from cases like this contribute to a deeper understanding of the complexities of malignancy-related cardiac complications and inform the ongoing refinement of treatment strategies.
Conflicts of interest
References
1. babu rs, lanjewar a, jadhav u, wagh p, aurangabadkar g, upadhyay p. A case series of malignant pericardial effusion. J family med prim care 2022;11(10):6581-6585.
2. vogiatzidis k, zarogiannis sg, aidonidis i, et al. Physiology of pericardial fluid production and drainage. Front physiol 2015;6:62.
3. Vinay k, abbas ak, aster jc. Robbins pathologic basis of disease. 10th ed. Elsevier 2023:577-580.
4. su yc, hsu yc, chai cy. Role of ttf-1, ck20, and ck7 immunohistochemistry for diagnosis of primary and secondary lung adenocarcinoma. Kaohsiung j med sci 2006;22(1):14-19.
5. ulici v, wang lj. Stains & cd markers: ttf1. Pathology outlines. Published september 16, 2020. Accessed august 1, 2023.
6. pernick n. Stains & cd markers: cytokeratin 20 (ck20, k20). Pathology outlines. Published july 1, 2013. Accessed august 1, 2023.
7. lichtenberger jp, reynolds da, keung j, keung e, carter bw. Metastasis to the heart: a radiologic approach to diagnosis with pathologic correlation. Am j roentgenol 2016;207(4):764-772.