Management of Congenital Third Branchial Arch Anomalies: About 2 Cases
Abstract
A rare source of lateral neck masses of congenital
origin are branchial abnormalities, which arise from aberrant development
during embryogenesis. The most frequent source of origin is the second
branchial cleft; anomalies resulting from the first, third and fourth clefts
are less common. Even though branchial cleft-derived cysts are rare, it's
crucial to take this condition into account when making a differential
diagnosis for neck masses, especially those that are laterally situated. This
article presents the rare case of a child of 6 years who presented the sudden
appearance of a lateral collection in the neck fistulized to the skin with
notion of recurrent neck infections at the same site. Patient underwent
extensive diagnostic examinations, including radiology, which were consistent
with a left subcutaneous collection measuring17.2*15 mm with irregular,
heterogeneous and hypoechogenic contours. This article presents the rare case
of 2 children aged 6 and 7 who presented with the sudden onset of a lateral
collection in the neck fistulized to the skin with notion of recurrent neck
infections at the same site. The patients underwent extensive diagnostic
examinations, including radiology, which were compatible with a left
subcutaneous collection measuring a cauterization was performed on both
patients. Nine months after surgery, there were no signs of neck infection or
purulent episodes. This clinical example underlines how essential it is to
identify uncommon diseases such as branchial cleft cysts as early as possible
and treat them appropriately.
Keywords:
Branchial apparatus; Cyst; Cleft anomaly
Introduction
During the fourth week of pregnancy, the gill
apparatus, also known as the branchial arches, which are made up of endodermal
pouches and ectodermal clefts, aid in the correct development of the head and
neck. Incomplete obliteration causes congenital malformations of the ectodermal
clefts of the branchial arches, which in most cases (75%) culminate in a cyst
and in 25% in a sinus1.
Roughly 17% of all pediatric neck masses are
abnormalities related to branchial clefts2.
These are typical congenital lesions that are typically identified in the early
years of life in children3. A
cyst, sinus or fistula may occur as a result of a branchial apparatus failing
to involutate4. Less than 1% of
branchial anomalies are fourth branchial arch anomalies, which primarily affect
the left side and manifest as suppurative thyroiditis or recurrent neck
infections5.
Cysts with a fourth branchial cleft parallel to the
recurrent laryngeal nerve are extremely rare. Like third branchial cleft
sinuses, they are most frequently found on the left side (80%) and they
typically form a sinus that extends from the apex of the piriform sinus.
However, instead of passing superiorly to reach the anterior left upper thyroid
lobe, they travel inferiorly. Cysts can occur anywhere in the neck, all the way
down to the mediastinum, but they are typically found next to the thyroid gland.
It is challenging to differentiate radiologically between anomalies involving
the third and fourth branchial clefts due to their close closeness. The link
between the sinus tract and the superior laryngeal nerve needs to be surgically
identified in order to provide an appropriate diagnosis.
Due to their uncommon incidence, there are conflicting
data on recurrence and complication rates and there are no standard
recommendations for diagnosis and treatment. In an effort to supplement the few
information available, we aimed to compile a summary of the clinical
characteristics of all reported cases of third branchial abnormalities and to
determine the most effective courses of action for both diagnosis and therapy.
Our analysis examines the characteristics of all published cases of fourth branchial
arch abnormalities, building on the framework of a prior review that we just
published6.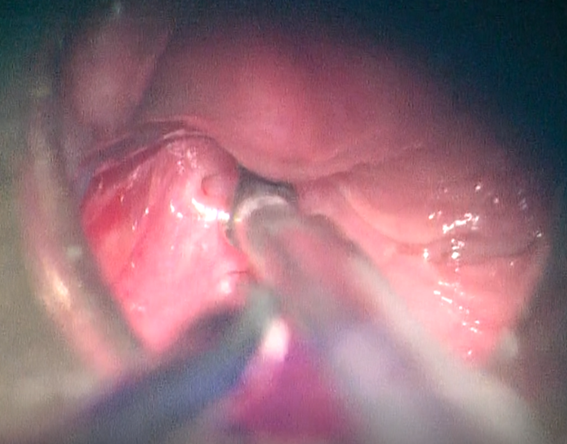
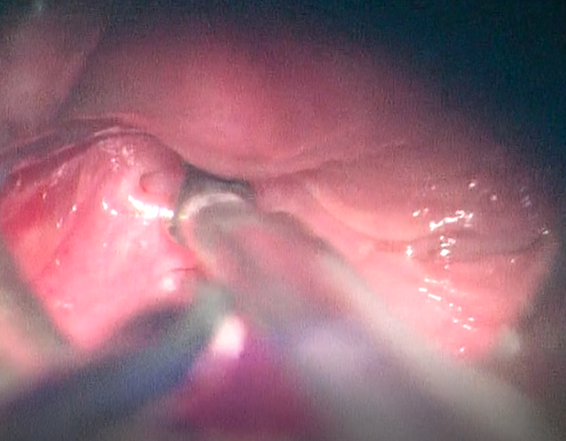
Figure 1: Orifice of the
fistula in the left piriform fossa in endoscopic view
The second clinical case is a 6
years old child admitted with a left latero-cervical tumefaction presenting a
history of recurrent superinfection. A hypopharyngoscopy was performed on the
suspicion of a 3rd or 4th cleft fistula, revealing the presence of a fistulous
orifice at the bottom of the left sinus prirforme. The both patients underwent
cauterization with very good post-operative improvement, nine months after surgery, there were no signs of
neck infection or purulent episodes.
Discussion
Anatomically speaking, anomalies of the third
branchial arch are remnants of a tract that emerges from the base of the
pyriform sinus. After passing over the thyrohyoid membrane, it ascends to loop
around the glossopharyngeal nerve below the hypoglossal nerve. From there, it
descends posterior to the internal or common carotid artery and ascends
anterior to the vagus nerve. The anterior border of the point where the middle
and lower thirds of the sternocleidomastoid muscle converge is where the
external aperture, if it exists, is usually located 86. The anatomy of vestiges
of the sinus tract from the fourth branchial arch shows that there is no
difference between the right and left side. Nonetheless, third and fourth
branchial arch defects appear similarly clinically, to the point where some
writers have proposed combining them into a single entity7. However, these two types of anomalies are
anatomically different: the tract of fourth branchial arch anomalies originates
from the apex (caudal end) of the pyriform sinus and passes through the
cricothyroid membrane beneath the superior laryngeal nerve, whereas third
branchial arch anomalies are thought to originate from the base (cranial end)
of the pyriform sinus and pass above the superior laryngeal nerve.2.
The lesions associated with the complex third and
fourth branchial pouch sinuses might arise at any point along the fistula's
path. Third and fourth branchial pouch sinus lesions are classified into three
forms based on the existence of internal or external fistulas: sinus, fistula
and cyst types. These types have been previously documented in the literature8-13. The sinus kind is the most prevalent
among them. The type of fistula is typically brought on by recurrent iatrogenic
incision and drainage, abscess ulceration or secondary infection. The
percentage of the fistula type (37.3%) was higher in our study because patients
who had multiple recurrence events following surgery or recurrent drainage
incisions were referred to our hospital. For patients with third or fourth
branchial pouch sinus lesions, we recommend clinically refined subtypes based
on observation of a large number of cases, taking into account medical history,
physical signs, imaging examinations and in-office laryngoscopy. This could lead
to a more successful treatment plan.
Third arch anomalies were discovered to be strongly
left-sided (89% vs. 11% on the right); this occurrence may be connected to the
trajectory of the nearby fourth branchial arch, which similarly produces
anomalies that are primarily left-sided.4 we discovered that the main
techniques for diagnosing third branchial arch anomalies were direct
laryngoscopy and barium swallow; MRI was mainly utilized when cystic anomalies
were present. The cases under investigation included a variety of reported treatment
modalities. Although incisions with drainage were routinely made, most of the
time the first course of treatment was unsuccessful. The most prevalent
procedure for neck abscesses that did not involve thyroid involvement was
open-neck surgery that involved fistula tract excision. In the majority of
cases of acute suppurative thyroiditis, a partial thyroidectomy was performed19-24.
Rare congenital branchial arch malformations in
children are PSF and PSC20. PSC
frequently manifests as thyroiditis, respiratory failure or a neck abscess in
newborns20,25,30. The
pharyngobranchial duct, which connects the third and fourth pharyngeal pouches
to the pharynx, is the source of the problem20,26,25.
PSC is uncommon in new borns, thus the diagnosis and course of treatment are
still unclear and difficult to understand. Previously, the most usual and
comprehensive course of therapy involved the complete excision of the cyst and
fistula, with or without damaged thyroid tissue20,25,30.
However, research using internal opening ablation to
treat PSF has recently been published. Numerous ablation methods, such as TCA26,27, electrocauterization28,31,32, radiofrequency ablation33, laser cauterization29 and others, have been documented34. The usefulness of endoscopic
radiofrequency ablation in comparison to endoscopic-assisted surgery was
documented by Chen et al33.
According to their findings, patients who underwent radiofrequency ablation
stayed in the hospital for far less time than those who underwent
endoscopically assisted surgery. A retrospective investigation comparing TCA
with excision was published by Hwang et al26.
According to their report31,
patients had TCA chemocauterization, with a 46.1% recurrence rate.
Chemocauterization or re-excision was an effective treatment for all
recurrence-prone individuals. The first report of a large endoscopic
electrocauterization cohort came from Chen et al31.
The sinus tract was ablateted by inserting the flexible cautery electrode from
Bugbee into the sinus tract. They promoted this process in addition to incision
and drainage as a first-line therapy32.
Authors believed that cauterization was an appropriate first-line treatment for
PSF in a different paper that highlighted the benefits of electrocauterization.
Cha et al12
alternatively used trichloroacetic acid chemocauterization in 44 patients with
an initial success rate of 77%. Compared with other forms of cautery,
electrocauterization may be superior in permanently closing the sinus tract.
Electrocauterization also seems to be comparable in efficacy to the traditional
approach of open excision, which has a reported success rate of 85% overall and
92% when performed with partial thyroidectomy. Although some authors have
suggested that the combined approach should be used initially as the standard
for treatment, we believe it best reserved for cases that persist after
endoscopic management alone14,15.
In a case series by Pahlavan et al16,
patient experienced treatment failure with both cauterization and excision with
hemithyroidectomy. This patient was eventually treated by means of pharyngotomy
with obliteration of the left pyriform fossa 5% to 6% of patients have been
documented to have surgical site infections, salivary fistulas and vocal cord
paralysis as consequences following surgical and cauterization operations. Due
to inflammation and edema that may eventually compress these nerves during
electrocautery, paralysis of the superior and recurrent laryngeal nerves may
result. Despite the paucity of evidence, the majority of research come to the
conclusion that cauterization which is less invasive, has a lower risk of
complications and can be done concurrently with other operations like incision
and drainage in the event of an abscess should be the main course of treatment16,17. Moreover, reports of sclerosing
agent-treated cases that were successful have been published18.
The outcomes of endoscopic surgery are comparable to
those of open surgery, despite the fact that the less invasive endoscopic
approach appears to be favoured these days due to the decreased chance of
laryngeal nerve injury. In their description of a novel endoscopic procedure,
Huang et al. Used the KTP laser in conjunction with fibrin glue on five
children without reporting any complications or recurrences8.
Conclusion
Pyriform Fossa Sinus Tracts (PFST) have been described
more frequently in recent years, regardless of their embryologic origin. This
highlights how crucial it is that clinicians taking care of patients with
recurring neck infections especially those on the left side of the neck take
them into account. There are several PFST treatment options, most broadly
classified as open or endoscopic. This endoscopic treatment of piriform fossa
sinus tracts is reasonably easy to recommend due to its ease of use and little
related morbidity. Recurrence is possible, just like with open excision.
References
1. Daoud
FS. Branchial cyst: an often forgotten diagnosis. Asian J Surg 2005;28(3):174-178.
2. Fang
T, Yue L, Fasahat KM, et al. A fatal case of severe neck abscess due to a third
branchial cleft fistula: morphologic and immunohistochemical analyses. Diagn
Pathol 2016;11(1):87.
3. Filippo
C, Sara S, Luigi M, et al. Fourth branchial cleft anomaly: management strategy
in acute presentation. Int J Pediatr Otorhinolaryngol 2014; 78(9):1480-1484.
4. Aneeza
WH, Mazita A, Marina MB, et al. Complete congenital third branchial fistula:
does the theoretical course apply. Singap Med J 2010;51(7):122-125.
5. Watson
GJ, Nichani JR, Rothera MP, et al. Case series: endoscopic management of fourth
branchial arch anomalies. Int J Pediatr Otorhinolaryngol 2013;77(5):766-769.
6. Abe
K, Fujita H, Matsuura N, et al. A fistula from pyriform sinus in recurrent
acute suppurative thyroiditis. Am J Dis Child 1981;135(2):178.
7. Residual
fistula of fourth branchial arch anomalies and recurrent left-side cervical
abscess: clinical case and review of the literature. Hallak B, Bouayed S,
Leishman C, Sandu K. Case Rep Otolaryngol 2014;2014:931279.
8. Kenealy
JF, Torsiglieri AJ, Tom LW. Branchial cleft anomalies: a five year
retrospective review. Trans Pa Acad Ophtalmol Otolaryngol 1990;42:1022-1025.
9. Choi
SS, Zalzal GH. Branchial anomalies: a review of 52 cases. Laryngoscope 1995;105(9):909-913.
10. Ford
GR, Balakrishnan A, Evan JNG, et al. Branchial cleft and pouch anomalies. J
Laryngol Otol 1992;106(2):137-143.
11. Boyd
J, Templer J, Havey A, et al. Persistent thymopharyngeal duct cyst. Otolaryngol
Head Neck Surg 1993;109(1):135-139.
12. Gan
YU, Lam SL. Imaging findings in acute neck infection due to pyriform sinus
fistula. Ann Acad Med Singapore 2004;33(5):636-640.
13. Gibbs
CM, Nichols FC, Kasperbauer JL, et al. Meal-induced dysphagia and otalgia
secondary to a pyriform sinus fistula. Dig Dis Sci 2004;49:1560-1562.
14. Ahmed
J, De S, Hore IDB, Bailey CM, Hartley BEJ. Treatment of piriform fossa sinuses
with monopolar diathermy. J Laryngol Otol 2008;122(8):840-844.
15. Shrime
M, Kacker A, Bent J, Ward RF. Fourth branchial complex anomalies: a case
series. Int J Pediatr Otorhinolaryngol. 2003;67(11):1227-1233.
16. Derks LS, Veenstra HJ, Oomen KP, Speleman L, Stegeman
I. Surgery
versus endoscopic cauterization in patients with third or fourth branchial
pouch sinuses: a systematic review. Laryngoscope 2016;126(1):212-217.
17. Saadoun A. Endoscopy for
a fourth branchial cleft cyst. Am J Otolaryngol 2020;41:102623.
18. Kim MG, Lee NH, Ban JH, Lee KC, Jin SM, Lee S. Sclerotherapy
of branchial cleft cysts using OK-432. Otolaryngol Head Neck Surg 2009;141(3):329-334.
19. Huang
Yun-Chen, Peng Steve Shinn-Forng, et Hsu Wei-Chung. KTP laser assisted
endoscopic tissue fibrin glue biocauterization for congenital pyriform sinus
fistula in children. Int J Pediatr Otorhinolaryngol 2016;85:115-119.
20. Nicoucar
Keyvan, Giger Roland, Pope JR, Harrison G, et al. Management of congenital
fourth branchial arch anomalies: a review and analysis of published cases. J
Pediatr Surg 2009;44(7):1432-1439.
21. Sayadi
SJ, Gassab I, Dellai M, et al. Laser coagulation in the endoscopic management
of fourth branchial pouch sinus. In: Annales d’oto-laryngologie et de chirurgie
cervico faciale: bulletin de la Societe d’oto-laryngologie des hopitaux de
Paris 2006;123(3):138-142.
22. Derks LS, Veenstra HJ, Oomen KP, Speleman L, Stegeman
I. Surgery
versus endoscopic cauterization in patients with third or fourth branchial pouch
sinuses: a systematic review. Laryngoscope 2016;126(1):212-217.
23. Saadoun A. Endoscopy for a
fourth branchial cleft cyst. Am J Otolaryngol 2020;41(6):102623.
24. Kim MG, Lee NH, Ban JH, Lee KC, Jin SM, Lee SH Sclerotherapy
of branchial cleft cysts using OK-432. Otolaryngol Head Neck Surg 2009;141(3):329-334.
25. Zhu
H, Xiao X, Zheng S, Shen C. Diagnosis and management of pyriform sinus cyst in
neonates: 16‐years experience at a single center. J Pediatr Surg 2017;52:1989-1993.
26. Hwang
J, Kim SC, Kim DY, Namgoong JM, Nam SY, Roh JL. Excision versus trichloroacetic
acid (TCA) chemocauterization for branchial sinus of the pyriform fossa. J
Pediatr Surg 2015;50(11):1949-1953.
27. Yanagisawa
S, Oshio T, Kano M, Kano M, Tsuji Y, Morikawa Y. Endoscopic chemocauterization
for pyriform sinus fistula in children. Pediatr Int 2017;59(7):807-811.
28. Jordan
JA, Graves JE, Manning S, Mcclay JE, Biavati MJ. Endoscopic cauterization for
treatment of fourth branchial cleft sinuses. Arch Otolaryngol Head Neck Surg
1998;124(9):1021-1024.
29. Wang
S, He Y, Zhang J et al. CO2 laser cauterization approach to congenital pyriform
sinus fistula. J Pediatr Surg 2018;53(7):1313-1317.
30. Li
WX, Dong Y, Zhang A, et al. Surgical treatment of fourth branchial apparatus
anomalies: A case series study. J Otolaryngol Head Neck Surg 2020;49(1):79.
31. Chen
EY, Inglis AF, Ou H, et al. Endoscopic electrocauterization of pyriform fossa
sinus tracts as definitive treatment. Int J Pediatr Otorhinolaryngol 2009;73(8):1151-1156.
32. Ishinaga
H, Kobayashi M, Otsu K, et al. Endoscopic electrocauterization of pyriform
sinus fistula. Eur Arch Otorhinolaryngol. 2017;274(11):3927-3931.
33. Chen
T, Chen J, Sheng Q, et al. Pyriform sinus fistula in children: A comparison of
endoscopic‐assisted surgery and endoscopic radiofrequency ablation. J Pediatr
Surg 2021;56(4):800-804.
34. Xia L, Lin Z, Lin X, et al. The treatment
of congenital pyriform sinus fistula: A single‐center experience. Pediatr Surg
Int 2020;36(7):779-788.