Marine-Lenhart Syndrome about a Case
Introduction
The most common cause of hyperthyroidism is autoimmune (graves basedow disease). It usually presents with diffuse goiter, but between 25-30% have thyroid’s nodules. Most of them are cold nodules on scintigraphy, therefore, hypo functioning and between 0.8 - 2.7% are hyper functioning1-5. The association of thyrotoxicosis due to graves' disease (gbd) and toxic nodules is what is called marine-lenhart syndrome (mls). It is rare and the prevalence ranges between 2.7 - 4.1%3,6.
It’s characterized by the presentation of graves' disease accompanied by hyper functioning’s nodules with the following characteristics: a) thyroid scintigraphy performed with tc-99m pertechnetiate, which shows graves' disease with one or more hyper functioning nodules; (b) hyper uptake in nodules after increase in tsh level due to radio-ablation; (c) return to nodal function after stimulation with endogenous or exogenous tsh; (d) benign histopathology. They present thyroid-stimulating antibodies. Although these characteristics are discussed7. This entity is more common in women, between 40 - 50 years old, with hyperthyroidism of several years of evolution and failed treatments with ats.
Keywords: hyperthyroidism; graves basedow disease; thyroid scintigraphy
Case report
A
73-year-old woman, with no notable family history, a light smoker, who came for
control of her thyroid disease (hyperthyroidism) after abandoning treatment for
two years. From his history, diagnosis of gbd of 10 years of evolution, with
grade iii goiter, 3cm left thyroid nodule, clear limits, smooth surface, not
painful. Bright eyes, conjunctival redness and chemosis in the eyes. On
ultrasound, right thyroid lobe measuring 12 x 14 x 46 mm. We don't see nodules.
Left lobe measuring 24 isthmus without particularities. Scintigraphy with hyper
enhancement nodule in the left lobe, with the rest of the parenchyma inhibited.
He receives synthetic ant thyroid drugs (ptu for two years and methimazole for
4 more years). He refuses to receive iodine 131 at that time.
Two
years after starting ant thyroid treatment, a repeat ultrasound showed a single
nodule in the left lobe measuring 22 x 26 x 40mm. When eufunction is achieved,
a fine-needle aspiration puncture of the left thyroid nodule is requested and a
bethesda ii cytogram is obtained.
At
the time of the consultation, he presented insomnia, palpitations, and had lost
10 kg of weight in recent months. The examination revealed grade iii goiter, an
irregular surface with a nodule in the left lobe of approximately 4 cm. Regular
rhythm of 120 cycles/minute. Discrete exophthalmos, lacrimal gland hypertrophy,
bilateral chemosis. Positive moebius sign. From the laboratory tsh: 0.09 uu/l
(0.27-4.7), t4l 1.9 ng/dl, t3l: 3.9 ng/dl (0.93-1.7). Thyroperoxidase
antibodies (tpo) and thyrotropin-stimulating antibodies (tsi): positive.
Thyroid
ultrasound: total volume of 26 cc, right lobe 18 x 19 x 42 mm and left lobe 24
x 27 x 48. Nodule 22 x 25 x 39 mm, peripheral vascularization with little
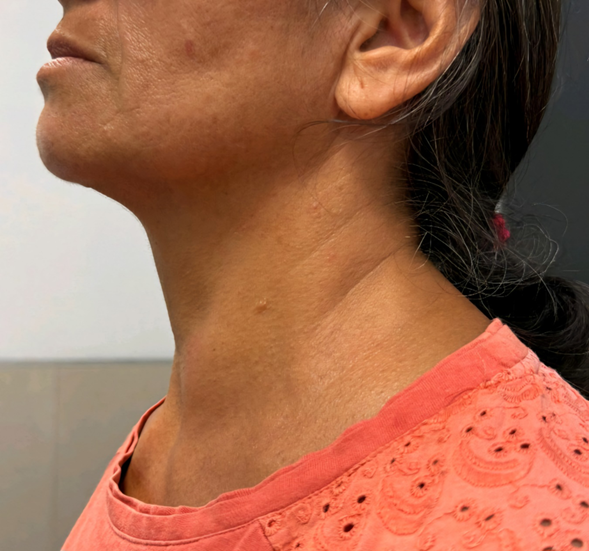
central. No lymph nodes (figure 1).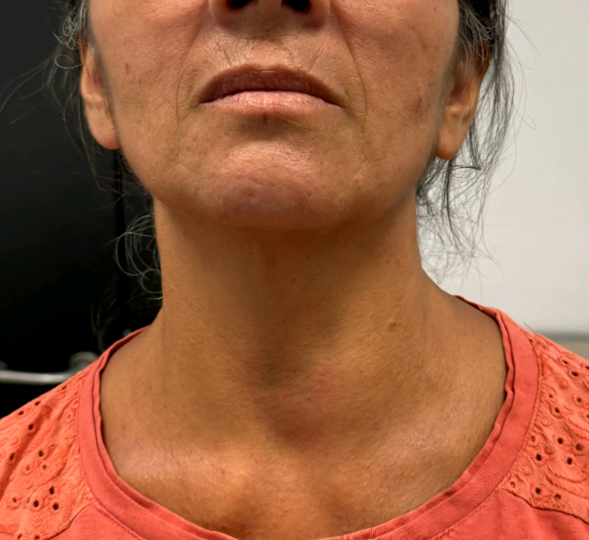
Figure
1.
Front and profile photo showing the major thyroid nodule in the left thyroid
lobe
Thyroid
scintigraphy with ct-99: irregular hyper uptake, highlighting a cap-shaped area
of hyper uptake, with a thyroid nodule in the left lobe with characteristics
of multi-nodularity and functional blockage of the rest of the glandular
parenchyma (figure 2).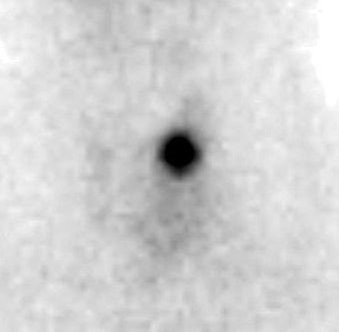
Figure 2. Thyroid scan compatible with
marine lenhart’ syndrome.
Orbital
tomography: bilateral exophthalmos. Optic nerves and intraorbital, intra and
extraconal fat without alterations. Increased thickness of both internal rectus
muscles. Methimazole 10 mg/day and propranolol 20 mg every 12 hours were
started. After 2 months he was euthyroid, prednisone 0.5 mg/kg/day was started
for a month and a therapeutic dose of iodine 131, 25 mci was administered
without complications. Two months later he developed hypothyroidism and was
started to be replaced with levothyroxine.
Discussion
Sml
is a rare pathology that associates graves' disease with hyper functioning
nodules. Diagnostic criteria are not well established. It can be done in a
classic way in which the nodules on the scintigraphy present the “cold”
appearance described by charkes, and in a variant in which cases are
characterized by nodules with a “hot” appearance. This last subtype includes
cases of functioning nodules with thyroid carcinomas and cases in which graves'
disease and functioning nodules can appear at different times in the evolution,
such as after treatment with synthetic antithyroid drugs or radioactive iodine.
The last presentation is the one present in our patient due to hyperuptake in
multinodularity.
The
uptake in the characteristic scintigraphy is given by the expression of nis in
the thyroid follicles, which increases, stimulated by tsh-stimulating
antibodies in graves' disease (independent of endogenous tsh), and
cold-hypofunctioning nodules have expression of nis tsh dependent. And in the
physical examination, exophthalmos and acropathies have been reported. Series
of sml have been reported in which exophthalmos was present in up to 50% of
patients7.
It
is not clear if the hyperfunctioning nodules represent a form of localized -
autoimmune graves' disease or if they could be an acquired and localized
mutation in the tsh receptor gene, producing a constitutive activation of the
tsh receptor, which leads to the development of a toxic adenoma8.
The
activity of toxic thyroid nodules can be enhanced by stimulators such as tsh or
antibodies against the tsh receptor. In summary, our case presents a variant of
sml characterized by ocular disease and hyperuptake in the thyroid
scintigraphy. Treatment can be with radioiodine, surgery, and synthetic
antithyroid drugs.
Surgery
is preferred in suspected malignancy, moderate to severe orbitopathy,
compressive symptoms, symptomatic goiter and patient preference. Synthetic
antithyroids are preferred in severe hyperthyroidism, advanced age, to achieve
euthyroidism before definitive treatment. Radioactive iodine is a safe,
definitive therapy that requires that the patient does not have active thyroid
ophthalmopathy, in which patients are generally left with thyroid hypofunction
- hypothyroidism and therefore must be replaced with exogenous thyroid hormone9 as is the case presented.
There
are reports in which radiotherapy in sml induces hypothyroidism more frequently
than in multinodular goiter, reported by danno, d. Et al, (42.9 vs. 9.0%, p =
0.005)10. Remember that this
pathology should be suspected if, before stopping treatment with synthetic
antithyroid drugs, the patient presents a relapse, accompanied by autonomous
functioning nodules by scintigraphy. We must remember that this pathology is
not malignant and can be accompanied by thyroid carcinoma, but it is the
minority of cases, so follow-up is essential in these patients.
References
4. Marine d, lenhart ch. Pathological anatomy
of exophthalmic goiter: the anatomical and physiological relation soft he
thyroid gland to the disease; the treatment jama internal medicine 1894;8(3):265-316.
10. Oka
m. A marked goiter involved in marine-lenhart syndrome. J gen fam med
2019;20(1):37-38