Mysterious Case of the Blue Abdomen
Abstract
Methemoglobinemia is a disease process triggered by
various inciting factors. In this case, we looked at a patient presenting with
a blue abdomen. We theorized that this discoloration is caused by an atypical
presentation of methemoglobinemia precipitated by nitric oxide formation in a
patient with colorectal cancer.
Keywords: methemoglobinemia;
blue abdomen; colorectal cancer; carcinogenesis
Introduction
Colorectal
cancer (crc) is the 3rd leading cause of cancer death in the united states in
men and women1. Although 10% of crc
cases are due to hereditary conditions, the majority are sporadic. Colonic
carcinogenesis is believed to be multifactorial and many genetic and
environmental factors such as smoking, alcohol consumption, and western diet
may affect its occurrence2. Previous
studies3,4 have suggested an
association between h.pylori infection and an increased risk of colorectal
cancer, as well as an association between h.pylori and immune thrombocytopenia5 with improvement of thrombocytopenia after
treatment of h.pylori6.
Case presentation
A 20-year-old, ashkenazi-jweish-female with past medical history of
chronic immune thrombocytopenia (itp) partially resistant to treatment
presented to the emergency department (ed) complaining of 2 months abdominal
pain and significant weight loss (10 kg) over a period of 6 months. During the
preceding 2 months, she presented to the ed 3 times with complaints of
abdominal pain, nausea and vomiting. This included an admission where she
underwent gastroscopy which demonstrated severe gastritis with positive helicobacter
pylori (hp). Platelets at this time were 25 k/ul. Abdominal x-ray demonstrated
dilated small bowel loops with air fluid levels and was admitted for
hospitalization. The following day, after clinical improvement, treatment for
hp was initiated and she was released home, assuming the x-ray findings were
the result of gastroscopy air insufflations.
Upon the most recent visit, vitals
signs were normal. On physical exam, blue discoloration of the abdomen was noticed (figure 1).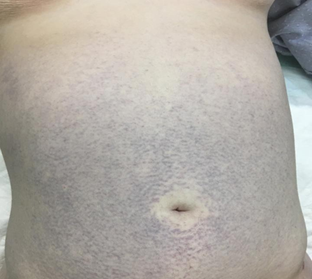
figure 1: blue discoloration of the abdomen
The abdomen was distended but soft,
with diffuse tenderness and no rebound or guarding. Laboratory workup was
unremarkable except for a platelet count
of 124 k/ul. An abdominal computed tomography (ct) scan
with oral and intravenous contrast demonstrated a dilated obstructed large
bowel with a suspicious concentric mass at the hepatic flexure (figure 2).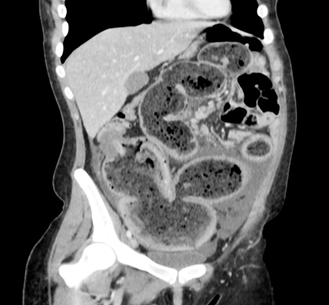
Figure
2: abdominal computed tomography (ct)
scan with oral and intravenous contrast demonstrated a dilated obstructed large
bowel with a suspicious concentric mass at the hepatic flexure
She was urgently taken to the
operating room for an exploratory laparotomy. The abdominal skin was bluish in color. Upon entering the abdomen, the small
and large bowels were markedly dilated. In addition, the ascending colon and
terminal ileum were ischemic. A small, indurated mass was defined at the
hepatic flexure as the cause of the complete bowel obstruction (figure 3). 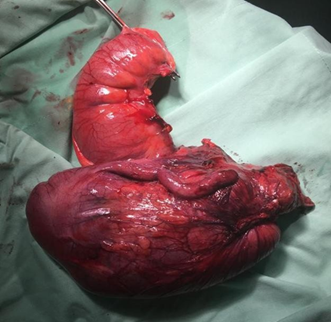
Figure
3: hepatic flexure of the complete
bowel obstruction
The colon distal to the obstruction
was pink and viable. There were several enlarged lymph nodes in the mesentery
of the ascending colon. No gross metastasis to the liver or peritoneum were
identified. A right colectomy with end-ileostomy was performed. During her
hospitalization, the platelet count was low. The patient clinically improved
over the following days. The ileostomy was pink and functional. The bluish
discoloration of her abdomen improved. On postoperative day 11, she was
discharged. The pathology report showed a signet cell mucus-producing carcinoma
of the ascending colon invading the muscularis propria into the peri-serosal
fat present at the radial resection margin with 3 affected lymph nodes out of a
total of 18 resected. The final pathologic stage was stage 3 adenocarcinoma.
Conclusion
Atypical presentations of complex disease processes will continue
to present to the emergency department. Early diagnosis of these cases,
especially cancer, will improve outcomes. In this specific case of the
20-year-old female with abdominal pain, there are several disease processes at
play and associations to be drawn. The first is between h.pylori and crc as
well as h.pylori and thrombocytopenia which has also been demonstrated in
previous studies. However, the more unique and perhaps novel presentation in
this case is attributed to the blue discoloration of the abdomen. Why was it
blue? - we have a theory. Although we were unable to find specific literature
linking nitric oxide production in crc, nitric oxide has been previously shown
to be a signaling molecule that can be elevated in tumors7. Increased levels of nitric oxide can also
precipitate methemoglobinemia. In this case, the methemoglobinemia presented as
truncal cyanosis causing the abdomen to be blue!
During surgery, the patient appeared to have abdominal obesity and
the intra-abdominal wall coloration was normal, therefore in addition to the
ischemic bowel appearing blue/black, the skin discoloration was localized to
the outer abdominal wall. The blue discoloration improved and resolved within
days after removal of the tumor. Methemoglobin level was measured at some point
during the first 24 hours of admission and was 1.8 (normal value less than
3.0). Pulse oximetry and blood coloration were normal.
We
have described an interesting case presentation of a patient with a blue abdomen. Further
studies involving the production of nitric oxide from crc and the resultant
localized methemoglobinemia are warranted to support our theory.
References
1. u.s. cancer statistics working group. U.s. cancer
statistics data visualizations tool, based on 2021 submission data (1999-2019):
u.s department of health and human services, centers for disease control and
prevention and national cancer institute. 2022.
2. wilmink ab. Overview of the epidemiology of
colorectal cancer. Dis colon rectum 1997;40(4):483-493.
3. vasilios p. Helicobacter pylori and colorectal
neoplasia: is there a causal link? World j gastroenterol. 2016;22(2):649-658.
4. zhang y, hoffmeister m, weck mn, chang-claude j,
brenner h. Helicobacter pylori infection and colorectal cancer risk: evidence
from a large population-based case-control study in germany. Am j epidemiol
2012;175(5):441-450.
5. kuwana m. Helicobacter pylori-associated immune
thrombocytopenia: clinical features and pathogenic mechanisms. World j
gastroenterol 2014;20(3):714-723.
6. asahi a, kuwana m, suzuki h, hibi t, kawakami y,
ikeda y. Effects of a helicobacter pylori eradication regimen on anti-platelet
autoantibody response in infected and uninfected patients with idiopathic
thrombocytopenic purpura. Haematologica 2006;91(10):1436-1437.
7. luanpitpong s, chanvorachole p. Nitric oxide and
aggressive behavior of lung cancer cells. Anticancer res 2015;35(9):4585-4592.