Necrotizing Pancreatitis After Gastric Bypass: A Unique Case Highlighting the Need for Guideline Adherence or Strategic Deviation for Better Outcomes
Abstract
Introduction
Bariatric surgery, including Sleeve Gastrectomy (SG) and Roux-en-Y Gastric Bypass (RYGB), is an effective procedure for weight loss but carries postoperative risks such as acute pancreatitis (AP). We present a case of AP post-RYGB leading to necrotizing pancreatitis, sepsis and respiratory failure, which required complex care.
Case presentation
A female in her early 50s presented to the emergency room with acute
abdominal pain two and half months post-Roux-en-Y Gastric Bypass (RYGB).
Initial tests revealed acute pancreatitis on CT, but biliary dilation and
gallstones were not detected. Despite standard of care her condition worsened
leading to transfer to intensive care unit. Repeat CT showed necrotizing
pancreatitis, ascites and pleural effusions. At a tertiary care center, an EUS
showed altered anatomy due to RYGB. Initial drainage procedures were deferred
until the pancreatic collection matured. Sixteen days later, an EUS-guided
cyst-gastrostomy failed due to solid necrosis. She developed sepsis, requiring
vasopressors and broad-spectrum antibiotics lead to IR-guided drainage of newly
developed abscess. A follow-up CT indicated extensive necrosis, prompting an
endoscopic ultrasound directed trans-gastric ERCP (EDGE) procedure with lumen
apposing metal stent (LAMS) placement. The patient later developed a pleural
effusion requiring chest tube insertion. Persistent necrosis led to an
endoscopic necrosectomy and a double pigtail stent placement. She was
discharged with oral antibiotics and follow-up recommendations after a
prolonged hospital course involving multidisciplinary management and complex
procedures.
Conclusion
Bariatric surgery-associated pancreatitis may require earlier,
non-standard interventions due to altered anatomy; research is needed for
guideline updates.
Keywords: Acute necrotizing
pancreatitis; Gastric Bypass; Guidelines; EDGE procedure; LAMS; OTS clips
Introduction
Bariatric surgery is a widely recognized treatment for individuals with a
high BMI. Sleeve Gastrectomy (SG) and Roux-en-Y Gastric Bypass (RYGB) are being
the two most commonly performed procedures. Both surgeries are effective in
promoting long-term weight loss and improving obesity-related health
conditions, while also having comparable low perioperative risk profiles1. Biter LU et al. illustrated overall
outcomes of the RYGB, with higher total weight loss following RYGB compared to
SG and lower rates of de novo GERD and dyslipidemia2.
While RYGB is generally safe and effective, there are still potential
postoperative complications, such as early anastomotic leaks, which have an
incidence rate of approximately 0.6% to 4.4%3.
Other complications include gastrointestinal bleeding, venous thromboembolism
and kinking or stenosis of the gastrointestinal tract, gallstones, small bowel
obstruction, marginal ulceration and internal hernia3. Acute pancreatitis (AP) is a less common
complication following bariatric surgery, with single-center studies reporting
rates between 0.2% and 1.04% for both SG and RYGB3,4.
The Mean time frame of developing AP after bariatric surgery is 3.5 years5. There was an increased risk of developing
AP in patients who had undergone RYGB especially in the case of prior
cholecystectomy, whereas the presence of only gallstones had a lower rate of
incidence6. AP regardless of the
type of bariatric surgery however tends to be mild and does not require
escalations of care such as an ICU admission3,4.
Kroner, et al. compared the mortality of AP in between patients with a history
of bariatric surgery and without a history of bariatric surgery, resulting in
patients with a history of bariatric surgery tend to have lower mortality,
morbidity and resource utilization7.
Nonetheless, it can be fatal and necessitates early diagnosis and treatment5. We present a case report of acute
pancreatitis following RYGB, which got complicated with necrotizing
pancreatitis, ascites, sepsis and acute hypoxic respiratory failure, requiring
a prolonged hospital stay and necessitating uncommon procedures due to altered
anatomy.
Case Presentation
A female in her early 50s presented to the emergency room with acute onset of abdominal pain, sharp in nature, located at periumbilical region without any radiation. Abdominal pain was progressively worsening without any aggravating or relieving factors and it was associated with nausea, non-bloody non-bilious vomiting and loss of appetite. She had a significant past medical history of Class 3 Severe Obesity status post Laparoscopic Roux-en-Y Gastric Bypass Surgery two and a half months before the current presentation. Other past medical history was urinary incontinence managed with urethral suspension and retropubic sling placement, asthma, obstructive sleep apnea, gastroesophageal reflux disease, anxiety, hyperlipidemia, prediabetes, breast augmentation and a tubal ligation.
Upon presentation, she was
afebrile with a maximum temperature of 98.2°F, vitals were as follows: BP 170-180s/80-100s
mm Hg, HR 80s-90s bpm, RR18 - 20s/min. Laboratory workup was noticeable for
Amylase 1162 U/L, Lipase 2787 U/L, Bicarb 21 mmol/L, ALT 57 U/L, AST 67 U/L,
glucose 170 mg/dL, BUN 24 mg/dL, creatinine 1.5 mg/dL, lactate 2.6 mmol/L and
an elevated white blood cell count 14.9 x10^3/uL. CXR did not show any evidence
of acute cardiopulmonary disease. CT scan of the abdomen and pelvis with IV
contrast showed acute pancreatitis with extensive fluid and peripancreatic
inflammatory changes throughout abdomen and possible developing ileus (Figure
1A and 1B). However, no evidence of gallstone or biliary dilatation or
organized fluid collection or necrosis were noticed. In ED, she received a
total of 4396 ml of ringer lactate (LR), IV antibiotics
(Piperacillin-tazobactam), IV morphine 4 mg x2, IV hydromorphone 1.2 mg and IV
antiemetics. The Patient was admitted for the management of acute pancreatitis
and she was started on ringer lactate (LR) at 200 cc/hr, PRN antiemetics and
pain medication and IV Piperacillin-tazobactam. Patient was kept NPO due to
nausea and vomiting and IV pantoprazole was started.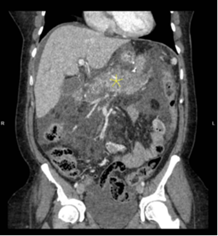
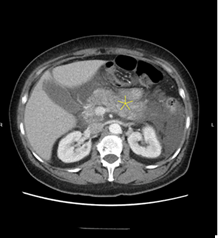
Figure 1A and 1B: CT scan image of showing
inflammation of pancreas (Yellow Star)
The patient remained hemodynamically stable overnight
but continued to experience severe abdominal pain for which she was started on
hydromorphone PCA pump. Next morning, she developed abdominal distension,
obstipation, increased guarding, hypoactive bowel sounds and tympany,
accompanied by persistent tachycardia and BP in 140s-150s/90s-100s mm Hg,
despite her pain being well controlled with the PCA pump. She was saturating
well on room air without any abnormal beathing sounds on examination. Her
morning labs showed results of BUN and creatinine getting better (20 mg/dL and
1.00 mg/dL respectively), however, increasing of hematocrit (HCT) to 48.8 was
concerning, hence, ICU evaluation was obtained for clinically worsening of
pancreatitis and general surgery team was consulted for concern regarding
abdominal compartment syndrome. Due to worsening of clinical condition
including tachycardia, persistent hypertension, worsening abdominal distension,
guarding and rigidity and persistent obstipation as well as newly developed
oliguria, dyspnea and worsening HCT she was transferred to the ICU for closer
monitoring and higher level of care. Subsequently, US abdomen revealed
cholelithiasis and distended gallbladder. Due to progressively deteriorating
clinical condition and worsening lactic acidosis, repeat STAT CT abdomen and
pelvis was performed, which showed necrotizing pancreatitis (Figure 2),
large loculated ascites and bilateral pleural effusion with lower lobe
atelectasis.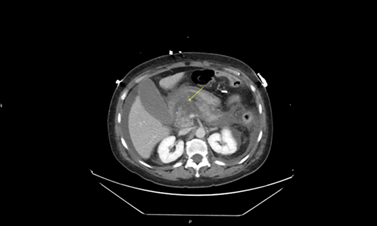
Figure 2: CT scan showing pancreatic
necrosis (Yellow arrow)
On the next day, she LR rate
was lowered at 75 ml/hr and eventually stopped and IV furosemide 20 mg was
given, which yielded only 125 cc urine output. Due to progressively worsening
dyspnea and a failed trial of NIV (non-invasive ventilation), the patient was
intubated. Bladder pressure was elevated at 13 mmHg. Later, she was transferred
to the tertiary center for higher level of care for possible endoscopic
ultrasound (EUS) cysto-gastrostomy and necrosis drainage.
At the tertiary care center,
she underwent Endoscopic Ultrasound (EUS) with a planned gastro-gastrostomy and
stent placement. The EUS procedure revealed altered surgical anatomy consistent
with a Roux-en-Y Gastric Bypass (RNYGB), with the esophagus, gastroesophageal
(GE) junction and gastrojejunostomy (GJO anastomosis being normal. During the
EUS, the excluded stomach was identified and distended using a 19-gauge needle
under fluoroscopic guidance. However, it showed that most of the neck and body
of the pancreas were best visualized from the pouch near the intended stent
placement site. Due to concerns about interfering with pancreatic drainage, the
decision was made to defer the EDGE (Endoscopic ultrasound directed
trans-gastric ERCP) procedure for future peripancreatic fluid drainage when the
collection becomes mature. Following the procedure, the patient was
hemodynamically stable and was downgraded from the ICU and she was managed
conservatively.
After 16 days, once the collection matured, she
underwent endoscopic ultrasound (EUS) guided gastro-cystostomy but was unable
to have the stent open properly due to the solid nature of collection, hence it
was closed with over the scope clip (OTSC). Following this procedure, overnight
she became febrile at 100.7-degree F, tachycardic at 110s-120s beats per
minute, hypotensive 60s-70s/40s mm Hg, blood work-up was notable for WBCs
37,000/uL (from 7000/uL) and lactate 2 mmol/L, creatinine 1.5 mg/dl.
Subsequently, she was transferred to the ICU for the requirement of the
vasopressors, she was given one dose of meropenem, vancomycin and tobramycin
each and was continued on meropenem. STAT CT abdomen and pelvis was performed
which showed enhancing pancreatitis with slightly larger necrotic pancreatic
wall, now containing multiple pockets of air concerning the superimposed
infection, abscess or the fistulation to adjacent bowel. IR guided drainage and
Jackson-Pratt (JP) drain placement showed purulent collection (Figure 3).
She was continued on meropenem for antibiotic and started improving transiently
with coming off pressors next day of drain placement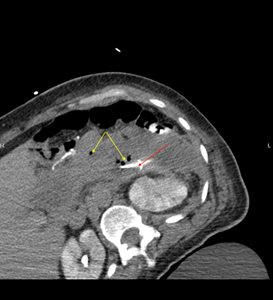
Figure 3: CT image showing multiple air
pockets (Yellow arrow) and JP drain placement (Red arrow)
Five days later, a follow-up
CT scan of the abdomen and pelvis revealed extensive pancreatic and
peripancreatic necrosis extending bilaterally to the pelvis, with some
improvement in the previously noted walled-off necrosis in the body and neck,
where a pigtail catheter was already in place. After careful consideration by a
multidisciplinary team, it was decided to proceed with an EDGE procedure,
involving the placement of a lumen-apposing metal stent (LAMS) at a
cysto-gastrostomy through a gastro-gastric (GG) fistula at the remnant stomach,
to drain necrotic pancreatic and peripancreatic material (Figure 4). Two
weeks later, the JP drain was removed. However, shortly after removal, the
patient developed a large left-sided pleural effusion (Figure 5),
causing tension physiology, for which a chest tube was placed. Meanwhile,
repeat CT imaging showed no change from the prior scan, indicating a failed
cysto-gastrostomy stent in draining the collection. As a result, the patient
underwent an endoscopic necrosectomy with the placement of a double pigtail
stent to aid drainage. The patient was eventually discharged with oral
antibiotics and advised to follow up with outpatient gastroenterology. The
stent was removed 40 days after the initial placement and plan was made to do
cholecystectomy.
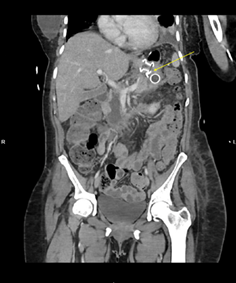
Figure 4: CT image with necrotizing
pancreatitis and lumen-apposing metal stent (LAMS) at a cysto-gastrostomy
(Yellow arrow)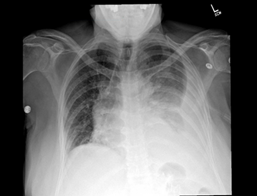
Figure 5: Chest Xray showing large left
sided pleural effusion
Discussion
Acute pancreatitis is not an
uncommon complication of bariatric surgeries like sleeve gastrectomy or
Roux-En-Y gastric bypass. This case highlights a rare situation where a patient
admitted for acute pancreatitis quickly progressed to necrotizing pancreatitis
within 48 hours, gallstones noted on ultrasound could be a contributing factor,
however, gallstones can be a byproduct of rapid weight loss because of
bariatric surgery. This case also highlights an altered anatomy after gastric
bypass which might have contributed to such a prolonged and complicated
hospital course and forced us to explore some unconventional routes in making
interventions.
Although post-gastric bypass
pancreatitis is typically mild, severe cases have been reported. Baran et al.
and Wang et al. described instances of severe pancreatitis due to blood clots
in the biliary tree after laparoscopic Roux-en-Y, likely caused by the reflux
of pancreatic and biliary contents through the ampulla of Vater5,8. Daster, et al. suggested that the
mechanical effects of the surgery could lead to pancreatic inflammation from
biliary leakage9. However, unlike
these cases where pancreatitis occurred during or shortly after surgery, our
patient developed acute pancreatitis months later. Weight loss after bariatric
surgery improves cardiovascular health, potentially reducing the severity of
pancreatitis7. Kerbage, et al.
found that mortality and outcomes in patients with post-surgery pancreatitis
are similar to those without weight loss surgery5.
Furthermore, gallstones are
the most common risk factor for developing necrotizing pancreatitis10. Rapid weight loss after bariatric
surgery, particularly in patients with a BMI over 40 kg/m², is a known cause of
cholelithiasis11, as is the case
with our patient. Bariatric surgery mobilizes cholesterol and triglycerides
from adipose tissue into the blood, leading to increased liver uptake. The
liver then secretes these lipids into bile, causing supersaturation and
gallstone formation. In addition to above, reduced gastrointestinal and
gallbladder motility, due to lower cholecystokinin levels, contributes to
cholestasis and promotes gallstone formation12.
The incidence of gallstones after bariatric surgery, in fact, have been so common
that physicians have considered prophylactic concurrent cholecystectomies. This
has also been thought to mitigate the risk of acute pancreatitis caused by
gallstone formation11,13. A
conservative approach to prevent gallstones and reduce pancreatitis risk
post-surgery is a 6-month course of 500-600 mg daily ursodeoxycholic acid
(UDCA) as explained by Miller et al. and Sugerman, et al.14,15. Interestingly enough, afferent loop
syndrome which is the distention of the biliopancreatic-jejunal anastomosis after
a Roux-en-Y procedure has been shown to cause pancreatitis, be it an early or
late complication16,17. While it
not being the primary cause, afferent loop distention may be contributory in
the outcomes of the case above and undoubtedly puts the patient at a higher
risk of developing pancreatitis than the general population.
In severe cases of acute
pancreatitis, the leakage of pancreatic enzymes can result in damage to the
pancreatic tissue, leading to complications such as fluid accumulation in
spaces, including ascites and pleural effusions. Additionally, a compromised blood
supply to the pancreatic tissue can result in necrosis. Necrotic tissue is at
risk of an infection, which can progress to sepsis and multi-organ failure.
Infected necrotizing pancreatitis carries a mortality rate of nearly 100%
without intervention and up to 30% even with surgical treatment18,19. Current guidelines recommend
moderately aggressive fluid resuscitation during the initial 24-48 hours to
mitigate inflammatory leakage and prevent necrosis20.
Evidence also supports early enteral nutrition as soon as it is tolerated, as
it reduces the risk of infectious complications by preventing gut bacterial
translocation20. However, while
early feeding helps lower the risk of infection, it does not necessarily
prevent necrosis, making frequent monitoring essential during the first 48
hours, ideally every 6-8 hours20,21.
This includes tracking BUN and hematocrit (HCT) levels, along with bedside
assessments of vital signs, urine output and overall fluid status. Ideally,
decreasing BUN and HCT levels indicate adequate hydration and renal perfusion.
Conversely, persistent or rising levels are associated with a poor prognosis
and may necessitate escalation of care. In our case, the observation of an
elevated heart rate, blood pressure and rising HCT levels suggested clinical
deterioration, prompting notification of intensive care for higher-level
management. When a patient does not respond to optimal treatment,
investigations for complications are warranted. In our case, repeat CT imaging
revealed necrotizing pancreatitis that rapidly progressed to infected necrosis,
despite adherence to guideline-based management.
In this case, the altered
anatomy following Roux-en-Y gastric bypass presented significant challenges for
performing advanced endoscopic procedures. According to the American College of
Gastroenterology (ACG) guidelines, in cases of acute necrotizing pancreatitis
involving stable patients, interventions should ideally be delayed until the
necrotic collection matures (ideally 4 weeks)20.
Our management approach attempted to align with these recommendations by
initially deferring the EDGE (endoscopic ultrasound-directed trans-gastric
ERCP) procedure to a later stage. However, due to clinical deterioration, we
proceeded with the intervention around the three-week mark, which ultimately
failed and necessitated a necrosectomy with drain placement.
Kroner et al. proposed that
patients with a history of bariatric surgery may experience reduced mortality,
morbidity and resource utilization in cases of acute pancreatitis7. This is likely attributed to
post-surgical changes affecting pancreatic and gastrointestinal functions,
including both hormonal and anatomical modifications. In retrospect, evaluating
whether this patient truly aligned with the typical profile of a stable acute
necrotizing pancreatitis case is essential. The altered anatomy from the Roux-en-Y
gastric bypass, specifically the gastric and peripancreatic surgical changes,
likely played a significant role in compromised peripancreatic drainage. This
could have contributed to inadequate fluid evacuation and increased the risk of
local and systemic complications. The consideration arises as to whether
performing an EDGE procedure earlier might have provided any benefit by
addressing the drainage issue earlier and preventing complications from
progressing.
Furthermore, the patient's
unique anatomical configuration could have predisposed her to a higher risk of
infection and sepsis due to the impaired drainage. The necessity for multiple
interventions may have exacerbated this risk, potentially compounded the
inflammatory response and facilitated septic progression. Ultimately, the
combination of altered anatomy and delayed but necessary procedures extended
the clinical course and may have necessitated prolonged management and
monitoring. These insights underscore the importance of individualized
decision-making in cases involving complex postoperative anatomy, where early
intervention strategies might need to be adapted to optimize patient outcomes
and prevent severe complications.
Conclusion
Bariatric surgery may be
directly or indirectly associated with acute pancreatitis (AP) by promoting
gallstone formation because of rapid weight loss. This case involves acute
pancreatitis that progressed to necrotizing pancreatitis, which subsequently became
infected and leaded to sepsis, despite adherence to initial guideline-based
management. Here, we emphasize the importance of monitoring critical parameters
to assess the prognosis and initiate earlier escalation of care. A significant
point of discussion is about the potential benefits of earlier intervention
that deviates from standard guidelines, particularly in patients with altered
functional and anatomical conditions. Based on our hypothesis and observations,
we advocate for further investigation into whether early endoscopic
intervention results in improved outcomes for patients with a history of an
altered anatomy who develop necrotizing pancreatitis.
Declaration
Authors declare no conflict of
interest regarding the publication of this article.
Disclosure
Authors declare no
financial/funding source(s) regarding the publication of this article.
Previous Publication/presentation/submission
This manuscript has never been
considered for publication/presentation/submission as a full manuscript or part
of it.
Consent
Written informed consent was
obtained from the patient for publication of this case report and accompanying
images.
Author Contribution
KJY: Conceptualization, Data
Collection, Writing-Review and Editing, Project Administration, Visualization,
Validation.
SA: Writing-Review & Editing,
Resources, Data Collection.
PC: Writing Original Draft, Data
Collection.
MP: Writing Original Draft.
JU: Writing Original Draft.
NN: Writing Original Draft.
AK: Writing Original Draft.
AK: Writing Original Draft
DK: Review & Editing, Project
Administration, Supervision.
AVP: Review & Editing, Project
Administration, Supervision.
SS: Review & Editing,
Supervision.
WG: Review & Editing, Project
Administration, Supervision, Validation.
Data Availability Statement
All data used to support the
findings of the study are included within the article, no additional data
required.
References
5.
Baran KC, de Brauw M. Pancreatitis following bariatric
surgery. BMC Surg 2019;19:77.