Oropharyngeal Schwannoma: A Rare Case Report and Literature Review
ABSTRACT
Schwannomas are tumors arising from the nerve
sheath usually benign, solitary and usually slow growing. In head and neck
region, they are mostly located in tongue, floor of mouth but rarely in
oropharynx and tonsils. They are usually asymptomatic but may cause dysphagia.
They rarely undergo malignant degeneration. We present a case of
dysphagia in a 17 year old female caused by giant schwannoma of the oropharynx.
An MRI was performed showing a pharyngeal process affecting the posterior
pharyngeal wall and compressing the oropharyngeal airway. The treatment was
based on surgical excision with no sign of recurrence in the follow up.
Keywords: Schwannoma; Transoral; Oropharynx; histological; Treatment;
Dysphagia
INTRODUCTION
Schwannomas (also called schwannomas), first described by Verocay1, are benign tumors
of the peripheral nerves that typically appear as slow-growing, solitary
lesions. It originates from the proliferation of Schwann cells2; these are
individual, encapsulated benign tumors arising from tumors Schwann cells of
peripheral nerves, cranial nerves and autonomic nerves3. While they can
develop in any part of the body, the head and neck region is the most common
location (25-48%), with intraoral origins being rare occurrences (1%)4,5. We present a case of dysphagia in a 17 year old female
caused by giant schwannoma of the oropharynx.
CASE REPORT
We present the case of a 17-year-old girl with no
prior medical conditions, who sought evaluation at our department due to a
four-month history of increasing throat swelling, which caused swallowing
difficulties, changes in her voice and nocturnal snoring. Clinical examination
using a tongue depressor revealed a large bulging mass rising from the
posterior wall of the pharynx, posterior and inferior to the left posterior
tonsillar pillar. The overlaying mucosa was unevenly covered with a white
membrane. Indirect laryngoscopy showed a sizable mass towering above the
bent-forward epiglottis. (Figure 1)
No signs of lymphadenopathy were detected upon physical examination. The rest
of the otorhinolaryngologic examination showed no abnormalities.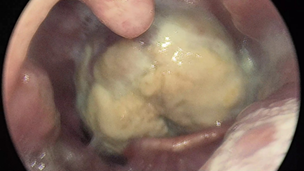
Figure
1. Clinical examination showing a
bulging mass rising from the posterior wall of the pharynx
An
MRI was performed showing a pharyngeal process affecting the posterior
pharyngeal wall and compressing the oropharyngeal airway.
The mass appears well-defined, roughly oval in shape, with irregular contours.
It exhibits isosignal on T1 and heterogeneous hypersignal on T2 imaging. Post
Gadolinium injection, it shows intense and heterogeneous enhancement. Its was
measuring approximately 40x30.5 mm, extending to 48 mm. It displaces the
epiglottis and extends to the supraglottic level, filling the left piriform
sinus completely and the right piriform sinus partially. It comes into intimate
contact, with loss of separation line in places, with the left palatine tonsil.
Posteriorly, It comes into contact with the vertebral bodies of C2-C3 and C4
without osseous lysis, as well as the intervertebral discs of C2-C3 and C3-C4. (Figure 2).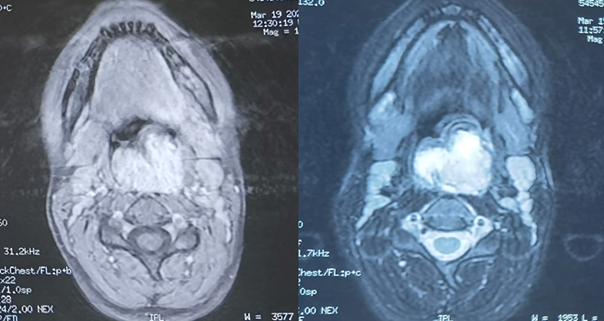
Figure
2: Facial MRI performed showing a
pharyngeal process affecting the posterior pharyngeal wall
An initial biopsy under local anesthesia was performed
revealing a remodeled and ulcerated Schwannoma. Given the benign nature of the
tumor, it was decided to perform a transoral excision under general anesthesia.
The tumor was friable and bled upon contact. The excision was thorough,
reaching deep into the tissue and extending to the superior pharyngeal
constrictor muscle. The surgical planes were closed using 2-0 Vicryl sutures. (Figure 3) The post-operative course
was uneventful with no complications noted. the patient was discharged after
24-hour observation period.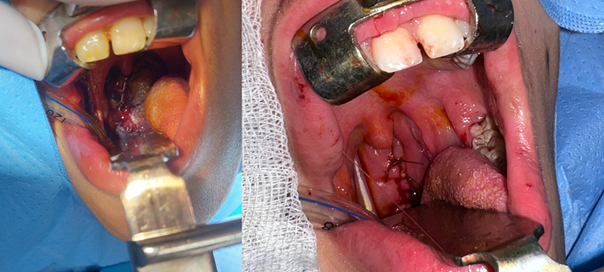
Figure
3: Post-operative image after the
excision of the mass and surgical suture
The histological examination reveals a spindle-shaped tumor
proliferation arranged in bundles. It consists of spindle cells without visible
cytoplasmic boundaries, arranged in short or intertwined bundles, with nuclear
palisades and cellular whorls. The cells have elongated nuclei, which are
non-atypical. The blood vessels have thin walls. Immunohistochemical analysis
demonstrates diffuse expression of pS100 in tumor cells. They did not express
CD34, EMA, or AML. (Figure 4)
Definitive diagnostic of schwannoma was favored. After a one-year follow-up,
the patient showed no signs of tumor recurrence.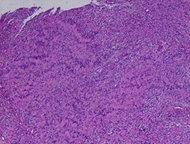
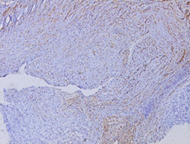
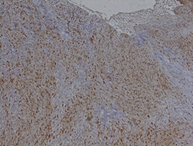
Figure 4: Histological aspect
DISCUSSION
The schwannoma, also known as neurilemmoma, neurinoma, perineural
fibroblastoma, is a tumor that grows slowly and typically remains encapsulated.
It is generally asymptomatic and does not cause any symptoms. Malignant
transformation of schwannomas is extremely rare. Pharyngeal presentations of
extracranial schwannomas, although rare, account for a quarter of all cases
that occur in the head and neck6. However, it appears to be more prevalent
during the ages spanning the second and third decades of life7. Research conducted
by William et al. revealed that 83% of the cases studied was male population,
whereas Lucas showed a stronger inclination towards females. Hatziotis and
Asprides, along with Enzinger and Weiss, observed an equal distribution between
both genders8,9.
In the oral cavity, lesions are most common in the soft tissues, more
commonly the tongue, followed by the palate and buccal mucosa, and may have
clinical manifestations similar to other benign lesions, lesions such as
mucoceles, fibromas, lipomas, and benign salivary gland tumors10,11. The most common
location is in the parapharyngeal space of the neck. Clinical signs and
symptoms vary depending on the size and location of the tumor and the nerve of
origin. They are usually
asymptomatic and appear as painless swelling. Oropharyngeal
schwannoma rarely occurs and causes dysphagia, odynophagia, radiating pain12.
The patient was initially asymptomatic, dysphagia
gradually develops due to growth. There, tumor spread to surrounding areas is
well sealed parapharyngeal spaces are rare but can cause compression vascular
structure12 However, isolated supraglottic
oropharyngeal schwannomas are rare. Holinger and Johnston13
found among 1197 cases of supraglottic oropharyngeal benign lesions only one
case of schwannoma. New and Erich14
found only one case of schwannoma among 722 cases of benign supraglottic
oropharyngeal tumors.
The process of diagnosing involves the use of imaging studies, such as
CT scans, and histology12. CT scans are important, but so are Magnetic
Resonance Imaging (MRI). Schwannomas, when scanned, appear to have lower
density and exhibit peripheral enhancement when contrast is applied. This is
observed in MRI scans of schwannomas. On T1-weighted images, these tumors
exhibit a relatively low signal intensity, while on T2-weighted images, they
display a high signal intensity. Typically, the enhancement of these tumors is
uniform throughout12. The MRI sequence exhibits a distinct contrast in the
'salt-pepper' characteristics representing the low signal intensity of vascular
flow12. Schwannomas are
macroscopically usually bordered and encapsulated. Histopathologically, five
schwannoma variants have been described: common schwannomas, plexiform
schwannomas, cellular schwannomas, epithelioid schwannomas, and ancient
schwannoma15. There are two
different histological patterns of common schwannoma: known as Antoni type A
and type B. Antoni type A tissue is characterized by dense Schwann cells with
nuclear palisades, whereas Antoni type B tissue has loosely arranged cellular
pleomorphism. Vascularity is not a prominent feature, and necrosis and mitotic
activity are rare. In type A, cells are sometimes arranged in a palisade-like
arrangement with nuclei next to each other in strips and cytoplasm in adjacent
strips, a pattern known as "Verocay bodies."16.
CONCLUSION
Schwannoma located in the oropharynx is extremely rare causing a progressive
difficulty of swallowing. The diagnosis is based on MRI and histological
studies. The treatment of the choice is surgical excision with a less
frequently recurrence after complete excision.
REFERENCES
2. Sayed SI, Rane P, Deshmukh A, et al. Ancient schwannoma of the parapharynx causing
dysphagia: A rare entity. Ann R Coll Surg Engl 2012;94(7):e217-e220.
3. Wang B, Yuan J, Chen X, Xu H, Zhou Y, Dong P.
Extracranial non-vestibular head and neck schwannomas. Saudi Med J 2015;36(11):1363-1366.
4. Williams HK, Cannell H, Silvestor K, Williams DM.
Neurilemmoma of head and neck. Br J Oral Maxillofac Surg 1993;31(1):32-35.
5. Pfeifle R, Baur DA, Paulino A, Helman J. Schwannoma of
the tongue. Report of 2 cases. J Oral Maxillofac Surg 2001;59(7):802-804.
6. de Bree R, Westerveld GJ, Smeele LE. Submandibular
approach for excision of a large schwannoma in the base of the tongue. European
Arch Oto-rhino-laryngology 2000;257(5):283- 286.
7. Kao G. Neurilemoma. eMedicine.com
2012.
8. Williams HK, Cannell H, Silvester K, Williams DM.
Neurilemmoma of the head and neck. British J oral Maxillofac Surg
1993;31(1):32-35.
9. Enzinger FM, Weiss SW. Soft tissue tumours. St Louis:
MO Mosby 1995;821-50.
10. Leu YS, Chang KC. Extracranial head and neck
schwannomas: A review of 8 years’ experience. Acta Otolaryngol 2002;122:435-437.
11. Lambade PN, Palve D, Lambade D. Schwannoma of the
cheek: Clinical case and literature review. J Maxillofac Oral Surg 2015;14(2):327-31.
12. Nemade HO, Jaiswal SA, Borade VR. Schwannoma of Oropharynx:
A Rare Presentation. Otorhinolaryngol Clin 2013;5(2):104-106.
13. Kanth TVR, Nanayya R, Satyaprabhakar Y, Blandeena K, Murthy
PSN. Giant supraglottic schwannoma. Indian J Otolaryngol Head Neck Surg 2006;58(10):397-398.
14. New GB, Erich JB. Benign tumours of the larynx; A
study of 722 cases. Arch Otolaryngol 1983;28:841-910.
15. Weiss SW. Enzinger
and Weiss's soft tissue tumors. Malignant soft tissue tumora of uncertain type.
2001:1538-1545.
16. López-Jornet P, Bermejo-Fenoll A. Neurilemmoma of the
tongue. Oral Oncology Extra 2005;41(7):154-157.