Perivalvular Abscess in Prosthetic Aortic Valve Endocarditis: Case Report and Short View
ABSTRACT
The perivalvular
cardiac abscess is a severe condition associated with infective endocarditis,
leading to significant morbidity and mortality if not diagnosed and managed
adequately. The incidence of perivalvular abscess among patients with infective
endocarditis is between 30% to 40%, with the aortic valve having a higher
predisposition than the mitral valve and annulus. It appears to occur more
often in prosthetic valve endocarditis than in native valve endocarditis, and
the most common pathogen isolated is Staphylococcus aureus. Surgical
treatment is usually the final treatment but the time of intervention is of
high importance to get the best results. Perivalvular abscess often
necessitates complex surgical techniques that remain always a challenge related
to anatomical surprises that can be found in situ.
We describe a case with
prosthetic valve endocarditis (PVE) of mechanical aortic valve with non
coronary sinus perivalvular abscess and vegetations around 1.2cm, complicated
with severe regurgitation, due to paravalvular leakage in a patient who
underwent aortic valve replacement 25 years ago. The bacterial strain isolated
is S.Aureus non MRSA .A small subaortic ventricular septal defect was found
during examination of the outflow tract of left ventricle. Furthermore ,we are also taking a
short view of literature related to the choice of surgical strategy and
management of this entity of patients.
Conclusion:
Aortic
valve endocarditis with perivalvular abscess formation remains a therapeutic
challenge and surgical treatment is a gold corner. The surgical technique
chosen is always challenging because of the tissue destruction and the necessity
of anatomic aortic root reconstruction. The time of intervention should be
evaluated carefully according to every patient.
Keywords: Infective endocarditis
(IE); Prosthetic valve endocarditis (PVE); Perivalvular abscess
INTRODUCTION
Infective endocarditis
involving the left side valves of the heart remains a serious medical problem,
with substantial morbidity and mortality. Its incidence has been reported from
1.7 cases1 to 6.2 cases2 per
100 000 person-years, the risk increasing significantly with advancing age.
Prosthetic valve
endocarditis (PVE) is a microbial infection that occurs on parts of a
prosthetic valve and accounts for 20% of infective endocarditis. It occurs in
up to 6% of patients who have valve prosthesis. The incidence of perivalvular abscess among
patients with infective endocarditis is between 30% to 40%. It is the most severe form of infective
endocarditis and is associated with high morbidity and mortality. The early
diagnosis and initiation of treatment improves outcome and decreases
complications and mortality.
The perivalvular
cardiac abscess is a severe complication of infective endocarditis, with a
significant morbidity and mortality.
We present a case with perivalvular
abscess and vegetations around 1.2cm in PVE of mechanical aortic valve complicated also with central
regurgitation and important paravalvular leak, in a patient who underwent aortic
valve replacement 25 years ago. The management of this patient was a challenge
due to comorbidities and severe clinical presentation that precluded urgent
surgical intervention.
CASE PRESENTATION
A 50-year-old man B.M
presented to emergency department, because of more than 10 days history of
recurrent fever, chest pain, productive cough, headache, myalgias, arthralgias.
The patient reported
aortic valve replacement 25 years ago and significant medical history of
Chronic Kidney Disease (CKD), history of Mellitus Diabetes type II, Arterial
Hypertension st II, Heart Failure NYHA II, Secondary Anemia.
On
presentation to the emergency department, he went through a physical
examination and were detected Osler nodes, Janeway lesions and subungual
hemorrhages, characteristic findings in infective endocarditis.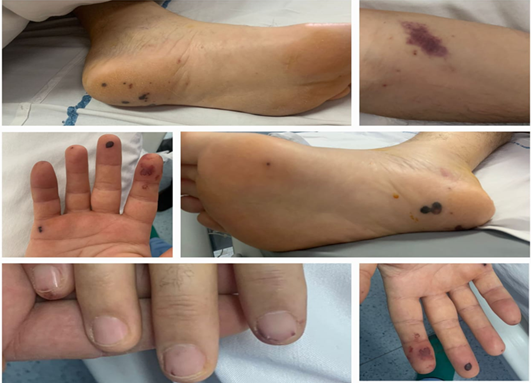
Figure 1. Red
arrows-Osler
Nodes; Yellow arrows-Janeway lesions; Green arrows-subungual hemorrhages
His vital signs on admission were as follows: Heart rate (HR): 100-115 beats/min blood pressure (BP): 160-60 mmHg; pulse oximetry (SPO2): 90% in 5L O2; temperature:39,5°C, bilateral bronchial rales; peripheral edema.
Laboratory
investigations show elevated blood cell count with leukocytosis (26’700/mm3)
and deviation to the left; neutrophils 77%; Hemoglobin 9.8 g/L; high C reactive
protein (27.2 mg/L-<0.5mg/dl. The COVID-19 throat swabs PCR result was
negative.
Computed tomography scan
of thorax, abdomen, and pelvis did not show any source of infection. A
trans-thoracic echocardiogram showed mild aortic regurgitation and mitral
regurgitation with no clear vegetation, however, trans-esophageal
echocardiogram (TEE) showed reduced left ventricular function with moderate
central aortic regurgitation. Hypoechogenic mass along the aortic side of the
mechanical aortic valve was noted. Important paravalvular leak and non-coronary
sinus abscess was suspected (Figure 2). 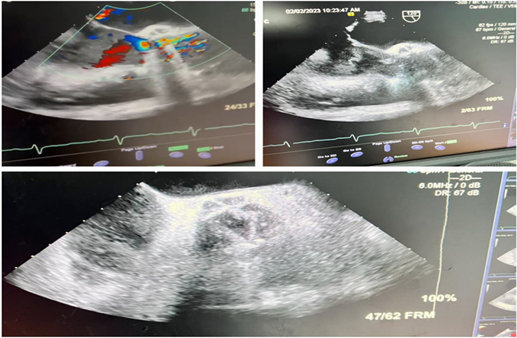
Figure 2. Transesophageal echocardiography findings Red
arrows-abscess
Blood cultures on
admission grew Staphylococcus aureus non-MRSA (VITEK –MS and VITEK-2 Compact.
Therefore, the patient was started intravenous antibiotics: Ceftriaxone 2.0g
12hourly, Levofloxacine 500mg 24 hourly , Vancomycin 1.0g 12 hourly , Flagyl
500mg 12 hourly according the antibiogram.
The patient was
transferred to Cardiac Surgery Department after 5 days of and underwent the
intervention: Mechanical aortic valve replacement with SJM Regent valve nr.21.
The
intervention was performed under general anesthesia, through the median
sternotomy, under cardiopulmonary by-pass through aorto-bicaval canulation. Locally
we found aortic annular abscess that involved more than half of annulus
circumference starting from middle of noncoronary annulus, involving all left
annulus and through the left-right commissure ended near the middle of right
coronary annulus. Demonstrated in the following picture (Figure 3).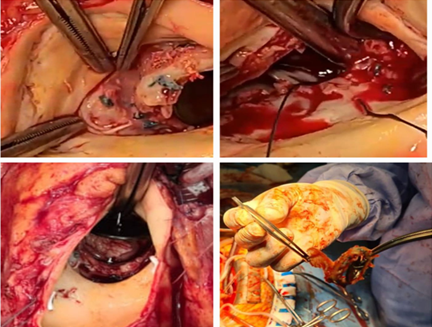
Figure 3. Periannular abscess,
prosthetic dehiscence and vegetations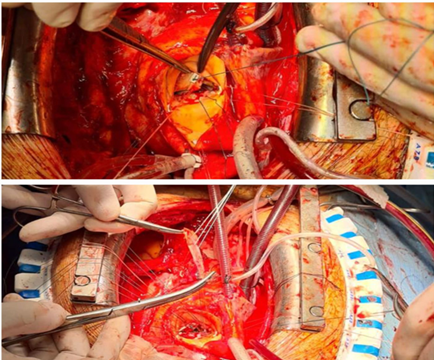
Figure 4. Surgical Technique
Reconstruction
of aortic annulus with pericardial and synthetic patch was necessary together
with aorto-mitral continuity. We found casually a small subaortic VSD which was
closed with two ticron 2/0 sutures with pledged (Figure 4). Postoperative trans-thoracic echocardiogram showed normo-functional
mechanical aortic valve with mean gradient of 9mmHg.
The patient did good post-operative course and
followed the protocol under antibiotic regimen.
DISCUSSION
Perivalvular abscess is the second most common finding
in IE, and along with its complications such as pseudoaneurysm and fistula
formation, is urgent indication of surgical intervention because of increased
mortality rate3.
Murdoch
et al. looked at the presentation, etiology and estimated the patient incidence
of infective endocarditis at about 15%. In regards to prosthetic tissue or
prosthetic valves, the infection tends to be entirely peri-annular, extends to
the myocardium, and results in paravalvular abscesses from dehiscence of the
valve. The incidence of perivalvular abscess among
patients with infective endocarditis is between 30% to 40%, with the
aortic valve having a higher predisposition than the mitral valve and annulus. Native
aortic valve endocarditis, usually located in a weak part of the annulus
near the atrioventricular node (AV), clearly demonstrates the anatomic
predisposition and exemplifies why abscesses and heart block presents as
frequent sequelae. Additionally, the severe extension of perivalvular infection
can also result in extrinsic coronary compression, or disruption, leading to an
acute coronary syndrome4.
Echocardiography
is pivotal in diagnosing cardiac abscess or IE by enabling vegetation
detection, assessment of valvular damage, evaluation of resulting hemodynamic
abnormalities, and observation of associated complications. Statistical
analyses indicate that transthoracic echocardiography has a sensitivity of 60%
to 75% for vegetation detection, whereas transesophageal echocardiography has a
sensitivity exceeding 95%5. In
echocardiography, abscess typically presents as hypoechoic area in perivalvular
zone without detectable blood flow inside6.
CT finding of a markedly thickened area around the aortic root is indicative of
an aortic root abscess, and may show good relation to TEE and pathologic
findings7.
The most common cardiac complication of PVE is heart
failure. This can result from prosthetic valve dehiscence, which leads to
valvular insufficiency, or myocardial infarction from emboli. Other cardiac
complications are perivalvular abscess formation, intracardiac fistula, and
pericarditis. Non-cardiac complications are usually a result of an embolic
event, metastatic abscess formation, or a mycotic aneurysm. The incidence of
embolic events ranges from approximately 15% to 35% and can occur one to two
years after the abscess resolution8.
Although
the biggest discussion in management of perivalvular abscess stays in when and
what to do to reach the best outcomes?!
What?
In treating intracardiac abscesses, it is
vital to provide appropriate antibiotic
therapy as soon as possible. Blood cultures should be acquired to
identify the pathogenic bacteria and assist antibiotic selection. Until culture
results are available, empirical antibiotic medication should be started to
address a wide range of potential infections. The surgical method chosen is
determined by factors such as the size and location of the abscess. The high
inpatient mortality rates (12% to 24%) for peri-annular abscesses, irrespective
of the surgical technique4.
It
has been shown that endocarditis caused by Staphylococcus aureus and other
virulent microorganisms on valves in the left side of the heart are best
treated with early surgery.
The surgical principle of radical resection of all
infected or even suspiciously infected edematous tissues.
The complexity of the operations ranged from resection of part of the valve
annulus and surrounding tissues with reconstruction with a patch to radical
removal of the base of the heart including the entire aortic root, the
intervalvular fibrous body, the posterior mitral annulus and part of the
interventricular septum and atrial walls.
Various
surgical techniques are used to treat complicated aortic valve endocarditis:
patch, prosthesis, homograft. It is difficult to compare clinical outcomes of
such complex operations such as surgical treatment of endocarditis with
paravalvular abscess from different institutions. However, based on the reports
by9,10 is likely that the risk of
recurrent endocarditis is reduced by the use of aortic valve homograft in these
patients, but the long-term survival is influenced by numerous factors and the type of
valve is certainly not the most important one. Guidelines support the
use of both homografts and stentless bioprostheses in aortic valve endocarditis
with paravalvular abscess formation. The choice between these approaches is
currently based on the extent of infection, surgeon or institutional preference
and demographic factors.
When?
The operative mortality rate for the surgical
treatment of aortic root abscess varies from 3.9% to 25%11. Early intervention, not emergent, for IE is
known to have better outcomes and this reason leads surgeons to early operation
if life-threatening sequelae do not develop. Emergent surgery has a higher
mortality rate than does early and elective surgery (14.3% vs 9.3%)11. Studies have shown that early
(delayed-urgent) surgery has better outcomes than emergent surgery. Our
strategy is to undertake aortic root surgery after stabilizing the patient's
infection, hemodynamics and general status. However, some patients with
periannular extension of infection or myocardial abscess could potentially be
treated without surgical intervention11.
These patients include: Patients with small (less than 1 cm) abscesses,
patients who do not have complications of heart block, an echocardiographic
progression of abscess during antibiotic therapy, patients who do not have
valvular dehiscence or insufficiency. We should also mention that the outcomes
of prosthetic valve endocarditis are worse than that those of native valve
endocarditis. The operative mortality is higher than in native valve
endocarditis and the long-term survival is not as good. David and colleagues
reported that the early and late outcomes of PVE were worse than those of NVE,
because PVE's frequent association with paravalvular abscess made the surgical
reconstruction more complex. It is important for cardiovascular surgeons to be
aware that these complicated reconstructive procedures have higher mortality
and morbidity rates than does simple valve replacement for active IE11.
CONCLUSION
Perivalvular abscess can be manifested with severe complications and requires
early complex reconstructive surgery. Surgical intervention should be
tailored to the patient's specific situation.
Conflicts of interest: The authors declare that there is no conflict
of interest regarding the publication of this article.
References
1. Berlin
JA, Abrutyn E, Strom BL, et al. Incidence of infective endocarditis in the
Delaware Valley, 1988-1990. Am J Cardiol 1995;76(12):933-936.
2. Hogevik
H, Olaison L, Andersson R, Lindberg J, Alestig K. Epidemiologic aspects of
infective endocarditis in an urban population. Medicine 1995;74(6):324-339.
3. Bruun NE, Habib G, Thuny
F, Sogaard P. Cardiac imaging in infectious endocarditis. Eur Heart J
2014;35(10):624-632.
4. Ting M, Wang CH, Chi NH, Hsu RB, Chen YS, Yu HY. Outcome
for surgical treatment of infective endocarditis with periannular abscess. J Formos Med Assoc 2020;119(1):113-124.
5. Yuan XC, Liu M, Hu J, Zeng X, Zhou AY, Chen L. Diagnosis
of infective endocarditis using echocardiography. Medicine 2019;98(38):17141.
6. Habib
G, Badano L, Tribouilloy C, et al. Recommendations for the practice of
echocardiography in infective endocarditis. Eur J Echocardiogr 2010;11:202-219.
7. Feuchtner
GM, Stolzmann P, Dichtl W, et al. Multislice computed tomography in infective
endocarditis: comparison with transesophageal echocardiography and
intraoperative findings. J Am Coll Cardiol 2009;53(5):436-444.
8. Desaint
P, Chauvat A, Garaud T, et al. Perivalvular mitral abscess fistulised to
the pericardial cavity revealing staphylococcal endocarditis. Heart Lung Circ 2018;27:e34-e37.
9. Yankah
AC, Pasic M, Klose H, Siniawski H, Weng Y, Hetzer R. Homograft reconstruction
of the aortic root for endocarditis with periannular abscess: a 17-year study.
Eur J Cardiothorac Surg 2005; 28(1):69-75.
10. Sabik JF, Lytle BW, Blackstone EH, Marullo AG, Pettersson GB,
Cosgrove DM. Aortic root replacement with cryopreserved allograft for
prosthetic valve endocarditis. Ann Thorac Surg 2002;74(3):650-659.
11. Tuarez
JFR, Yelamanchili VS, Law MA. Cardiac abscess. StatPearls 2023.