Psilocybin as a Tool in the Management of Palliative Care: An Historical, Pharmacological and Clinical Approach
Abstract
Psilocybin, a psychedelic compound in certain mushrooms, has been used for centuries in spiritual ceremonies and neuropsychiatric therapy. Despite its stigmatization as a Schedule I substance in 1970, research into psilocybin has resurged since the early 2000s, particularly in psychiatry and palliative care. This review examines psilocybinʹs potential to improve the quality of life in palliative care by reducing psychological distress and enhancing emotional well‐being. The discussion includes its historical context, pharmacokinetics, pharmacodynamics, legal status and future perspectives in palliative care.
Keywords: Psilocybin; Quality of life; Palliative care; New medical treatments; 5‐hydroxytryptamine; Psilocybe cubensis
Introduction
The term “psychedelic”, derived
from the Greek root’s “psyche” (mind or soul) and “delos” (to reveal), was
first coined in 1956 by psychiatrist Humphry Osmond1, who was researching lysergic acid diethylamide (LSD)2. By the late 1950s, Albert Hofmann from Sandoz
Laboratories identified and synthesized the psychoactive compounds psilocybin
and psilocin, found in Psilocybe mushrooms3.
Sandoz marketed psilocybin under the name Indocybin for basic
psychopharmacological research and clinical studies. Psilocybin saw a rapid
rise in popularity during the 1960s and was classified as a Schedule I drug in
19704.
Psychedelics are hallucinogenic substances that, once ingested, produce effects that alter the mind and distort reality, inducing hallucinations, delirium, emotional changes and feelings of detachment. They are classified into dissociative drugs-such as dextromethorphan (DXM), ketamine, Salvia divinorum and phencyclidine (PCP)-and serotoninergic and dopaminergic hallucinogens that interact with serotonin and dopamine receptors, respectively5,6
In 1963, Aldous Huxley introduced the idea that psychedelics could be useful for treating ʺexistential distressʺ at the end of life. On his deathbed, he requested two doses of LSD, passing away peacefully while his wife guided him “toward the light7”. Kast and Collins conducted a trial comparing the analgesic effects of LSD with opioids in terminal patients, demonstrating positive relief effects, although the research was discontinued in 19688,9.
Psilocybin is a secondary metabolite found in mushrooms. It has played a crucial role in Aztec and Maya ceremonies, referred to as “the flesh of the gods” by the Aztecs10. Ethnographic documents such as the Florentine Codex (1529‐1579) and records from botanist. Schultes in the 16th century confirm the use of psilocybin mushrooms11,10.
Psilocybin is now the subject of numerous investigations supporting its therapeutic potential in treating pain, headaches, mood disorders and anxiety, including major depression, post‐traumatic stress disorder and obsessive‐compulsive disorder2. Since psilocybin is a widely described natural compound, opportunities for patentability are limited. As a result, it does not easily fit into a conventional profit‐driven pharmaceutical development model. Consequently, non‐profit entities such as the Multidisciplinary Association for Psychedelic Studies (MAPS), the Usona Institute and the Heffter Research Institute have generously funded initial development efforts and covered funding gaps. A shared goal of these organizations has been to advance the understanding of psilocybinʹs therapeutic effects and other consciousness‐expanding substances while influencing policies to ultimately increase their accessibility4. Compass Pathways Ltd. and the Usona Institute have also received “breakthrough therapy” status from the FDA for psilocybin treatments, particularly for refractory depression and major depressive disorder12,13. The ability of psilocybin to promote the growth of cortical neurons has been hypothesized as responsible for the sustained therapeutic effects of psychedelics, highlighting their revolutionary potential in the neuropsychiatric field14.
Potential ecological role/occurrence of psilocybin
The ecological reasons for
psilocybin production are a topic of ongoing debate. However, there must be a
substantial ecological benefit to justify the energy cost of synthesizing and
accumulating this secondary metabolite, which generally constitutes between
0.5% and 2% of the mushroom’s dry weight. Two primary hypotheses attempt to
explain the occurrence of these metabolites15:
Monomer hypothesis: The monomer hypothesis suggests that psilocybin acts as a defensive agent against animals that might consume psilocybin‐producing mushrooms. In this context, crude methanolic extracts of Psilocybe (ranging from 2 to 23 μg/ml) have demonstrated lethal effects on brine shrimp (Arthropoda), though their effects on Caenorhabditis elegans (Nematoda) have been ambiguous. These data, along with the production of alkaloids triggered in the mushroom upon the formation of ephemeral fruiting bodies, suggest that nematodes may not be the primary target of psilocybin. Thus, the lack of solid evidence supporting this hypothesis indicates that its role as a defensive agent may be limited. Despite these considerations, some studies suggest that psilocybin‐producing fungi might use the compound to protect themselves and influence the neurochemical behavior of small animals16
Regarding the monomer hypothesis in mammals, such as humans, psilocybin exhibits negligible acute physiological toxicity, with an LD50/ED50 ratio > 1000 in mice. This limited interaction or lack of interaction with Psilocybe mushrooms in mammals raises questions about the evolutionary necessity of psilocybin as an ecological defense in these species. Moreover, it is argued that mammals evolved in an environment where fungi had already developed defense mechanisms against invertebrates over millions of years15.
Oligomer hypothesis: Mushrooms containing psilocybin instantly turn blue when the mycelium is damaged or as they age. For decades, this distinctive phenomenon has intrigued both chemists and amateur mycologists. Oligomerization converts the indole core derived from L‐tryptophan into a blue chromophore15.
Chemically, psilocybin can be viewed as the stabilized form of psilocin with a phosphate ester group as a protective moiety. This group is removed when the mycelium is damaged and psilocybin is exposed to intrinsic phosphatases or animals, followed by subsequent oligomerization of monomeric psilocin units. Based on this reaction, psilocybin may fulfill its true ecological role as a nascent or complete oligo‐/polymer. The complex and dynamically developed coupling products share a polyphenolic character and an aryl coupling with tannins and melanin’s. These are families of substances with exceptional ecological importance, making tissues resistant to microbial attack and damage, such as from light. Like tannins, blue oligomers precipitate proteins. Additionally, it is assumed that several tannins serve defensive purposes by generating reactive oxygen species15.
In contrast to conditions that favor psilocin oxidation, tannin‐based oxidations are also facilitated by a basic environment, such as the guts of arthropods feeding on living or decomposing plant matter. For example, studies with saprophagous larvae of Pentheria holosericea (which share an ecological niche with wood/dung‐dwelling fungi like Psilocybe) maintain extraordinarily high pH values (10-12) and strongly oxidative conditions within their midguts, which aligns well with the ʺpolymer hypothesisʺ. In this scenario, psilocybin would represent the deactivated precursor of a ʺdefense polymer upon consumption,ʺ with production potentially triggered instantaneously. Hypothetically, highly reactive psilocybin radicals might react directly with proteins and could have an immediate deleterious effect on insects feeding on the mushrooms15,17
Structural, Chemical and Pharmacology Characteristics of Psilocybin
Psilocybe mushrooms belong to the Basidiomycota phylum and are part of
the Strophariaceae family18. The
psychoactive effects of these mushrooms are due to two indole alkaloids:
psilocybin and psilocin (Figure 1). Psilocybin is a phosphorylated ester
of psilocin, which is found only in minimal quantities. These compounds were
initially identified by Hoffmann and his team, who analyzed a sample of P.
mexicana collected by Heim19.
Figure 1: Psilocybe mushrooms and Psilocybin. (A) Psilocybe cubensis (Photo donated by Edgar Gonzalez Rodriguez). (B) Chemical structure of psilocybin in 2D. (C) Chemical structure of psilocybin in 3D
Psilocybin (CAS Number: [520‐52‐5], 3‐[2‐(dimethylamino) ethyl] ‐1H‐indol‐4‐ol dihydrogen phosphate, C12H17N2O4P, MW = 248.2481 Da, mp = 224 ºC) is a natural psychedelic that degrades with heat and is water‐soluble. It belongs to the class of indole‐alkylamine hallucinogens and is considered to have minimal or no activity by itself, acting primarily as a prodrug for psilocin (CAS Number: [520‐53‐6], 3‐[2‐(dimethylamino) ethyl] ‐1H‐indol‐4‐ol, C12H16N2O, MW = 204.268 Da, mp = 174.5 ºC). Psilocybin has six hydrogen bond acceptors, three hydrogen bond donors and a logP of 0.03, which suggests it cannot easily cross the blood‐brain barrier. In contrast, psilocin, the active metabolite of psilocybin, has three hydrogen bond acceptors, two hydrogen bond donors and a logP of 1.32, making it significantly more lipophilic than its precursor. This increased lipophilicity allows psilocin to cross the blood‐brain barrier more readily, thereby exerting its psychoactive effects. Visually, psilocybin is typically purified as white needle‐like crystals, while psilocin appears as a dark brown to black oily liquid4.
The structure of psilocin (C) and other indole‐alkylamine psychedelics closely resembles that of the endogenous neurotransmitter serotonin (A), the hallucinogen N, N‐dimethyltryptamine (B) and the hallucinogenic compound bufotenine (D) derived from toads. These structures are illustrated in (Figure 2).
Figure 2:
Naturally Occurring Tryptamine Analogues of Psilocybin: (A)Serotonin, (B)
Dimethyltryptamine, (C)Psilocin, (D)bufotenine
Psychedelics are agonists of the 5‐hydroxytryptamine 2A receptor (serotonin, 5HT2AR) and have the capacity to induce profound changes in perception, cognition and mood. Recent studies suggest that these compounds may promote structural and functional neuroplasticity in the cerebral cortex through the activation of these receptors. However, the exact mechanism underlying this effect is not yet fully understood. It appears to be related to the activation of the tropomyosin receptor kinase B (TrkB), which regulates the mammalian target of rapamycin (mTOR) and α‐amino‐3hydroxy‐5‐methyl‐4‐isoxazolepropionic acid (AMPA) receptor signaling (Vargas et al., 2023)
It remains unclear why some 5‐HT2AR ligands promote neuroplasticity and produce lasting therapeutic responses while others do not. However, recent research suggests that the physicochemical properties of different ligands and the subcellular localization of receptors and proteins may provide a partial explanation for this phenomenon. For example, serotonin, due to its physicochemical properties, cannot passively diffuse through nonpolar membranes. In contrast, psychedelics from the tryptamine family, such as psilocybin, can traverse cellular membranes. This leads to the hypothesis that the activation of an intracellular population of 5‐HT2AR is necessary for 5‐HT2AR ligands to induce cortical structural plasticity and produce behavioral responses similar to antidepressants, which are more durable and stable over time14.
Although G protein‐coupled receptors (GPCRs), such as 5‐HT2A, are traditionally considered as initiators of signal transduction originating from the plasma membrane, there is growing evidence suggesting that GPCR signaling from intracellular compartments may play important roles in cellular responses to drugs. The concept of signaling bias has recently been proposed to explain signaling differences between endogenous peptide ligands that do not cross the membrane and membrane permeable ligands in opioid receptors. Additionally, distinct signaling induced by ligands has been observed between intracellular populations and those located at the plasma membrane, as seen with δ‐opioid receptors14.
In the case of 5‐HT2AR ligands, a significant proportion of these receptors in cortical neurons are located in the Golgi apparatus. Intracellular compartments, such as the Golgi apparatus, are slightly more acidic compared to the cytosol and extracellular space. Therefore, it is plausible that the protonation of psychedelics within the Golgi apparatus leads to their retention and sustained signaling, resulting in neuronal growth even after transient stimulation. This persistent growth, even after the drugs have been cleared from the extracellular space, is a distinguishing feature of serotonergic psychoplastogens14.
Although the mechanistic details linking intracellular activation of 5‐HT2AR with cortical neuronal growth are not yet fully understood, it is likely that they involve the signaling of AMPA receptors, TrkB and mTOR, as previously established14. Future research should focus on examining in greater detail the signaling interactions among these proteins
Heteromers of 5‐HT receptors
A receptor
heteromer is described as “a macromolecular complex consisting of at least two
functional receptor units with biochemical properties that are demonstrably
distinct from those of their individual components20”.
This definition is accompanied by a set of criteria that a proposed receptor
heteromer must meet to be considered a physiologically relevant heteromer.
These standards can be summarized into three key criteria (Table 1) that
are necessary to demonstrate that a heteromer observed in a transfected in
vitro environment has physiological relevance in an in vivo context. Firstly,
proving that the receptor protomers have the capability to closely interact in
vivo is a crucial initial step in validating the heteromeric complex.
Appropriate evidence for this includes confirming that the receptor protomers
are located within the same cell and sequestered within the same cellular
compartments in native or primary cells21,20.
This is most commonly achieved through immunoelectron microscopy,
co‐immunoprecipitation and proximity‐based biophysical techniques, such as
Förster Resonance Energy Transfer (FRET)22
and proximity ligation assays21,23.
These techniques can also be used to provide direct evidence of heteromer
formation by demonstrating the distance dependent association of receptor
protomers within physiologically relevant environments.
Table 1:
Summary of Characteristics of Heteromers with Physiological Relevance
|
1 Receptor protomers must have the
ability to closely interact in vivo. |
|
2 Exhibit biochemical properties
distinct from their individual monomers. |
|
3 Alteration of the heteromer’s
function should be observable after the loss of functionality of the heteromeric
receptor. |
Secondly, to establish the biochemical distinctiveness of a heteromer compared to its individual monomers, it must exhibit unique biochemical properties. These differences can arise from allosteric modulation among protomers, which leads to unique signaling, trafficking, ligand binding or other pharmacological characteristics. Verification can also involve identifying a specific ligand for the heteromer. Common techniques to assess these properties include ligand binding assays to observe changes in ligand affinity, assays to study how heteromers affect signaling cascades and methods to investigate their trafficking, particularly regarding internalization mediated by surface and agonists21,22.
Finally, it is essential that its functional disruption be evident when a functional receptor heteromer is lost. This can be achieved using various methods, including membrane‐permeable peptides targeting heteromer subunit interaction sites, transgenic animals expressing protomers that cannot form heteromers and knockout animal models lacking one of the necessary protomers. Once a model of heteromer disruption is developed, several techniques can demonstrate changes in heteromer assembly and function. These techniques include comparing functional characteristics between systems with and without the relevant receptor heteromers, using heteromer‐specific antibodies capable of detecting heteromers and employing bivalent ligands selective for heteromers to show alterations in heteromer assembly and function21,22,24.
To be recognized as a physiologically relevant receptor heteromer, it must meet at least two out of three criteria .outlined by NC‐IUPHAR guidelines. Historically, demonstrating these properties in receptor heteromers has been challenging due to the lack of suitable experimental tools. However, advancements in technology have made it more feasible to confirm the presence of receptor heteromers in native tissues. This progress has resulted in a greater number of identified and pharmacologically characterized receptor heteromers21.
Examples of the
physiological relevance of heteromers in pathophysiology
The diversity of
changes in traffic profiles resulting from heteromerization has implications
not only for heteromer formation, cellular localization and surface expression
but also for signaling. The formation of heteromers, such as D2R/5‐HT2AR, can
have significant implications for cellular signaling and the effects of
psychoactive substances25.
The activation of GPCRs can lead to G protein‐dependent signaling events as well as G protein independent signaling events, such as signaling pathways mediated by β‐arrestin
Heterodimerization can induce changes in the conformation of the binding site and alterations in the pharmacological profiles of ligands. Biased ligands selectively activate one pathway over another. Biased ligands towards heteromers are molecules that exert a different downstream effect and a different pharmacological effect when binding to a heterodimeric complex compared to when binding to a receptor monomer (Shahet al., 2020).
The serotonin receptor 5‐HT2A and the glutamate receptor mGluR2 send signals through Gαq and Gαi, respectively and both are targets of atypical antipsychotics. Antipsychotics targeting 5HT2A act as inverse agonists at the receptor to reduce Gαq signaling. Antipsychotics aimed at mGluR2 have an agonistic effect that increases signaling through Gαi. It has been shown that 5‐HT2A and mGluR2 form a heteromer in native tissue and in a non‐pathological state, this heteromer acts to modulate signal transduction through each receptor protomer to enhance Gαi signaling and decrease Gαq signaling. Untreated schizophrenia patients exhibit upregulation of 5‐HT2A alongside downregulation of mGluR2, which is thought to lead to a suboptimal ratio of receptors for heteromerization, resulting in reduced heteromer formation in schizophrenia (Gonzalez‐ Maeso et al.,2008)26,27. Based on these findings, it has been suggested that combined therapy with a 5‐HT2A inverse agonist like risperidone and an mGluR2 agonist like LY379268 could mimic the signaling balance provided by the heteromer, potentially leading to better outcomes for patients, supported by preclinical experiments. Despite this seemingly straightforward interaction between 5‐HT2A and mGluR2 mediated by heteromerization, further research has revealed complexities in the function of this heteromer that may inform future targeting strategies28,29.
Pharmacokinetics
and Pharmacodynamics and Behavioral Effects of Psilocybin/Psilocin When
ingested, psilocybin is absorbed and undergoes first‐pass metabolism in the
liver, where it is rapidly dephosphorylated into the psychoactive psilocin by
an unidentified enzyme (Figure 3). Psilocin then enters the systemic
circulation and crosses into the brain, where it can exert its psychoactive
effects. Psilocin undergoes both Phase I and Phase II metabolism. Although
predominantly Phase II (≥80%), a significant portion still experiences Phase I
reactions. Phase I metabolism first involves the oxidation of psilocin to
4‐hydroxyindole‐3‐aldehyde and its subsequent oxidation to
4‐hydroxyindole‐3‐acetic acid or reduction to 4‐hydroxytryptophol; the enzymes
involved in this process have not yet been identified. Phase II metabolism, via
UGT1A10 in the small intestine and UGT1A9 in the liver, results in the
formation of a psilocin glucuronide conjugate. These metabolites are then
excreted. A recent study found that the elimination half‐life of psilocin is
approximately 3 hours in healthy adults, depending on individual
characteristics and the route of administration. The complete metabolic pathway
of psilocybin has been minimally studied and much more information is needed to
determine the exact mechanisms involved in its metabolism. (Figure 3)
below outlines the series of reactions believed to be involved in the
metabolism of psilocybin through urinary metabolite analysis4.
Figure 3: Phase I and phase II metabolites of psilocybin
As previously mentioned, psilocybin is a prodrug that, when metabolized in the body, produces psilocin, which functions as an agonist of serotonin (5‐hydroxytryptamine) 2A receptors (5‐HT2AR) to produce a ʺmystical‐likeʺ hallucinogenic effect30 due to induced hyperfrontality13, which in turn mediates its antidepressant and anxiolytic effects31
A possible antidepressant mechanism of action of psilocybin is through the deactivation or normalization of hyperactivity in the medial prefrontal cortex (mPFC)32. During depression, the mPFC is typically hyperactive33. The antidepressant properties of psilocybin are mediated by the modulation of prefrontal and limbic brain regions, including the amygdala34. The amygdala plays an essential role in emotion perception and processing networks35. In cases of depression, an individual typically loses responsiveness to emotional stimuli36. It is also suggested that the hyperfrontal metabolic pattern produced after psilocybin administration and 5‐HT2A receptor activation is comparable to the metabolic patterns produced during acute psychotic episodes in chronic schizophrenics11
Psilocybin is also reported to bind with high affinity to the 5‐HT2A receptor subtype, but with low affinity to the 5‐HT1A receptor subtype. The interaction of psilocybin and psilocin with 5‐HT2A receptors to produce psychotomimetic effects has been confirmed in experiments with ketanserin, a 5‐HT2A antagonist that attenuates psilocybinʹs effects13. In addition to the interaction with 5‐HT2A receptors, it is also suggested that the psychopharmacological action of psilocybin may be mediated by receptors other than 5‐HT2A37,35. Psilocybin and psilocin also interact with 5‐HT1D and 5‐HT2C receptor subtypes13. Psilocybin is reported to produce significant changes in brain dynamics and functional connectivity (FC) between brain areas (Grimm et al., 2018). The psilocybin‐induced alteration in brain connectivity involves the disintegration of associative networks and the integration of sensory function networks11. This dissociation may mediate the subjective effects of psilocybin use and an unrestricted cognition state11. Similarly, a possible mechanism behind the psychotomimetic effects of psilocybin is the interactions with feedback circuits between the cortex and thalamus13. Psilocybin administration produces general cortical activation11
Upon oral
administration, psilocybin converts to psilocin, which binds primarily to
5‐HT2A receptors, inducing “mystical” hallucinatory effects through enhanced
glutamatergic activity. Psilocin also promotes neuroplasticity and neurogenesis
via BDNF and mTOR pathways, contributing to its pharmacological actions, these
actions are represented in (Figure 4)19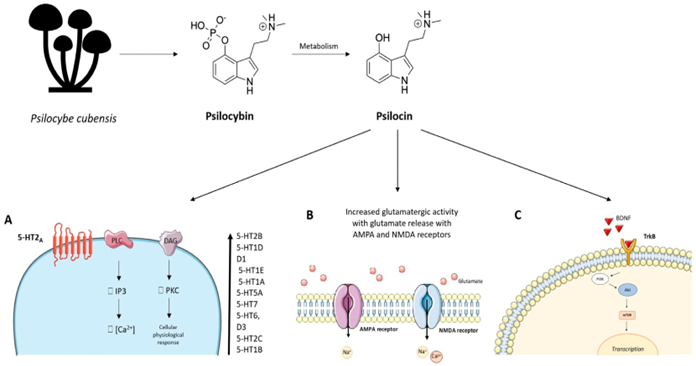
Figure 4: After oral administration, psilocybin loses its phosphate group and is fully converted into psilocin, which consequently represents the primary derivative responsible for its pharmacological activity. (A) Psilocin has a high affinity for the 5‐HT2A receptor and this binding is responsible for the ʺmysticalʺ hallucinatory effects induced by psilocin. In order of increasing affinity, psilocin can also bind to 5‐HT2B, 5‐HT1D, dopamine D1, 5HT1E, 5‐HT1A, 5‐HT5A, 5‐HT7, 5‐HT6, D3, 5‐HT2C and 5‐HT1B receptors. (B) Activation of the 5‐HT2A receptor in the prefrontal cortex by psilocin results in increased glutamatergic activity with the release of glutamate at AMPA and NMDA receptors on cortical pyramidal neurons. (C) Psilocin has been observed to exert its pharmacological action by enhancing neuroplasticity and neuritogenesis through the BDNF and mTOR pathways. Taken from Mastinu et al., 2023, The Bright Side of Psychedelics: Latest Advances and Challenges in Neuropharmacology. International journal of molecular sciences, 24(2), 1329. licensed under CC BY 4.
Effects of
Psilocybin Ingestion
The physical
effects of psilocybin ingestion depend on individual sensitivity to the
components and the doses administered. Tolerance to psilocybin varies widely,
so a safe practice is to start with low doses and assess specific reactions38. The effects of orally ingested psilocybin
begin within 20‐60 minutes, with a peak lasting 2 to 4 hours. A decrease in
effects is typically perceived between 3‐7 hours after consumption and
dose‐dependent late effects can be experienced up to 24 hours post‐ingestion4.
The usual dose in studies is 0.4 mg/kg. Psilocybinʹs effects are very similar to those of LSD. Symptoms reported include pupil dilation, changes in blood pressure, rhinorrhea, hypersalivation, slight increase in body temperature, mild sedation, dizziness and nausea. Muscle tension and tremors may occur and high doses can lead to increased heart rate39
Visual effects include color saturation, altered shape and color recognition, fusion of objects and colors, distorted perspective and heightened auditory sensitivity. Hallucinations often involve vivid, colorful shapes and varying figures seen with both closed and open eyes4.
Psychological effects associated with psilocybin use include euphoria, a sense of weightlessness, increased empathy, simultaneous emotional experiences, heightened musical appreciation, a feeling of “ego dissolution,” a need for “catharsis” and a sense of “rejuvenation”. Psilocybin use is also associated with altered perceptions of time and space, derealization and depersonalization. The effects can be perceived either positively or negatively depending on personal characteristics, knowledge of the objectives of psilocybin use and the individual’s particular life situation. Each psilocybin experience is expected to yield a distinct result and perception from the user. A summary of these effects is illustrated in (Figure 5)4,39
Figure 5:
Effects of Psilocybin in Humans. This figure summarizes the effects of
psilocybin in humans, which include pupil dilation, changes in blood pressure,
rhinorrhea, hypersalivation, a slight increase in body temperature, mild
sedation, dizziness and nausea. Muscle tension and tremors may occur, with high
doses potentially increasing heart rate39.
Visually, psilocybin causes color saturation, altered shape and color
recognition, fusion of objects and colors, distorted perspective and heightened
auditory sensitivity. Hallucinations often feature vivid, colorful shapes and
varying figures seen with both closed and open eyes
The theory of “ego dissolution” refers to a state where the sense of self is perceived as “suspended”. Psilocybin‐induced experiences allow individuals to become more open to their social environment, fostering a heightened sense of connection with their surroundings and the people around them. Research has shown that psilocybin significantly improves emotional facial recognition and reduces feelings of social exclusion, making it particularly beneficial in treating affective disorders such as depression and even suicidal risk38. The personal and social changes reported with psilocybin ingestion are also advantageous in treating anxiety by diminishing persistent negative cognitions and emotional responses to chronic illnesses, such as cancer40.
Adverse physiological effects of psychedelics are rare and generally transient, with cardiovascular responses linked to serotonergic and adrenergic actions being the most commonly reported. While precautions are advisable, severe adverse events are not typically observed. The primary risks arise from interactions with other medications, with psychological risks including adverse reactions in individuals predisposed to psychotic or manic episodes, trauma or depression and anxiety. These negative emotional responses generally resolve with proper preparation and post session psychological support, though more serious consequences such as existential crises, despair or self‐harm can occur41.
interactions with
other medications and other substances with psilocybin42 reveals various effects. Pretreatment with
anxiolytics such as buspirone significantly reduces psilocybin‐induced visual
distortion43. Antipsychotics like
chlorpromazine and haloperidol diminish psilocybinʹs effects on pupil dilation
and visual distortions, though haloperidol can increase anxiety without
affecting hallucinations44,45.
Risperidone also attenuates psilocybinʹs effects in a dose‐dependent manner45. SSRIs like escitalopram decrease negative
acute effects without altering the positive effects of psilocybin46. Ketanserin, a 5‐HT2A antagonist, blocks
various psilocybin effects, including those on the Altered State of
Consciousness Rating scale47,48.
Ergotamine does not significantly alter psilocybin experiences43 and alcohol consumption does not notably
affect psilocybin’s subjective effects, with most participants reporting
unchanged effect49.
Psilocybin‐Assisted
Therapy in the Palliative Care Setting
Psychedelic‐assisted
therapies leverage altered states of consciousness induced by agents such as
psilocybin, methylenedioxymethamphetamine (MDMA) or ketamine. Psilocybin has
been shown to reduce depression, anxiety and fear of death in patients with
cancer‐related depression and anxiety50,
with benefits persisting in 60% to 80% of patients at six months follow‐up51. Therefore, the goals of psilocybin treatment
in palliative care aim to enhance comfort, improve quality of life and
alleviate pain and stress52.
An important concept in palliative care is “existential distress”, which is linked to the anticipation of responses to physical and psychosocial changes occurring during chronic illnesses, aggressive treatments or conditions with high side effects, often seen in oncological diseases9. “Existential distress” is a relatively new concept and lacks a clear definition; however, it is reported to affect 9% to 29% of terminal cancer patients9. This distress is significantly related to the patient’s personality before the oncological diagnosis, the presence of psychiatric conditions, prior life traumas such as loss of family members due to similar diagnoses or clinical illness and the specific social and personal context at the time of disease onset. Anxiety, depression and existential distress are some of the possible clinical manifestations at the end of life. Psychedelic therapies, with their range of physical and emotional reactions approaching integrative personal experiences, hold significant spiritual relevance9
In the context of palliative care, it has become essential to explore new treatment approaches with an integrative focus that includes individual psychological interventions, psychiatric evaluation (for symptoms that may impact treatment and patient choices), psychosocial interventions involving family (to ensure clear understanding of diagnosis, treatment options and the best support methods tailored to each patient’s unique situation). Schimmel et al. review several important studies on the evidence of psilocybin in cancer patients. The prevalence of psychiatric symptoms in this population is estimated as follows: 14‐21% anxiety, 10‐25% mild depression, 15% major depression and 6‐19% adjustment disorders. A cross‐sectional study found that 12% of cancer patients (n=377) had a serious desire for death, with 52% of these meeting criteria for anxiety and/or depression9. An increasing number of oncology treatment units are expanding their therapeutic arsenal to address the mental health of patients with cancer diagnoses53.
Recent studies involving psilocybin, though limited in patient numbers, generally associate psilocybin with long‐term improvements in “existential distress”. Grob conducted an initial pilot study with advanced cancer patients (n=12) suffering from acute stress disorder, existential distress, generalized anxiety disorder and cancer‐related anxiety54, all according to DSM‐IV criteria. Patients received either psilocybin (0.2 mg/kg) or niacin (vitamin B3) as a placebo over two weeks. Improvements were observed in derealization and depersonalization dimensions associated with a positive mood (oceanic boundlessness) (p<0.001) and visionary restructuring (p<0.001). Ego dissolution results were marginally significant (p: 0.049). After one month, scores on the Beck Depression Inventory (BDI) decreased (p: 0.5), with more significant changes noted at six months (p: 0.03)54. The study did not show a reduction in analgesic use or cancer‐related pain intensity and no adverse effects from the intervention were reported9.
In 2016, Griffiths studied the effects of a high dose of psilocybin (22–30 mg/70 kg) versus a microdose considered as a placebo (1–3 mg/70 kg), building on previous studies (Griffiths et al.,2011; Griffiths et al., 2006) that established psychedelic and spiritual effects starting at 5 mg/70 kg. This study included patients with potentially terminal cancer and mood disorders (dysthymia, major depression or mixed anxiety and depression disorder). The research demonstrated that high doses of psilocybin significantly improved depression in the long term, with notable effects observed both after the second administration and at six months follow‐up. At six months, participants exhibited a clinical response rate of 78% to 83% in terms of reduced depression and anxiety symptoms. The psilocybin treatment also decreased demoralization and hopelessness, leading to improved spiritual well‐being and quality of life, both in the short term (after 2 weeks) and the long term (after 6 months). Additionally, the treatment positively impacted measures related to spirituality, transcendence, life satisfaction and acceptance of death, while reducing death‐related distress.
Earlier studies by Griffiths and colleagues initially focused on the effects of psychedelics in healthy subjects without psychiatric comorbidities55,56. These studies established the impact of psychedelics on mystical experiences, including feelings of unity and transcendence, fear of ego dissolution and visionary attitudes. They also assessed scales of mysticism, spirituality and transcendence related to death. The 2006 study compared high doses of psilocybin (30 mg/70 kg) with methylphenidate, while the 2011 study compared five doses of psilocybin (0, 5, 10, 20 and 30 mg/70 kg). It was found that subjective effects related to psychedelics began at 5 mg, with increased effects observed at higher doses of 20 to 30 mg. The higher doses were associated with mystical experiences, increased spirituality and a greater sense of transcendence related to death55.
In a randomized, crossover clinical trial with 29 patients suffering from cancer‐related anxiety and depression, participants received either 0.3 mg/kg of psilocybin or 250 mg of niacin, combined with psychotherapy. The study found that psilocybin significantly improved anxiety and depressive symptoms, reduced demoralization and hopelessness and enhanced spiritual well‐being and quality of life. Serious adverse events were not reported and no participants required psychiatric hospitalization. The most common adverse effects were increased blood pressure and heart rate (76%), headache (28%) and nausea (14%)57
Although it remains a debated topic, recent clinical trials involving oncology patients have shown significant reductions in death anxiety, as well as improved attitudes towards death and life57,56. Even notions of transcendence in death have been observed in subjects without psychiatric comorbidities, as demonstrated by Griffiths. Another dimension of psilocybin is its relationship to spiritual well‐being. A Phase II trial administered a fixed dose of 25 mg of psilocybin to 30 patients with cancer and major depressive disorder, evaluating spiritual and psychosocial healing using the NIH‐HEALS scale. The researchers found a significant improvement (p<0.001), although the study lacked a comparator and was not double‐blinded, which limits its methodological rigor58. Another pilot study, the HOPE trial, combined psilocybin with group psychotherapy for cancer patients with depression. This trial included three preparatory sessions, one high‐dose psilocybin session (25 mg) and three integration sessions with a total of 12 participants. The sessions resulted in events such as nausea, headache and hypertension, but no severe adverse events. The results showed significant improvement in depressive symptoms using the HAM‐D questionnaire (p<0.001), based on the FACIT scale. The emotional, functional, spiritual meaning, spiritual peace and spiritual faith domains showed statistically significant outcomes (p<0.05) and half of the group reported a mystical experience related to the transcendence of death scale (p: 0.014)59.
Demoralization, a form of existential suffering, was studied in older patients with a high‐impact chronic illness like HIV/AIDS. This study involved 16 patients who received both individual and group psychotherapy, the latter using the Short‐Term Supportive Group Therapy (SEGET) model, which is an existential psychotherapy centered on palliative care. The administration was divided into three groups: 0.3 mg/kg, 0.36 mg/kg and a control group. This study demonstrated a decrease in patient demoralization with a standardized effect size of 0.47 and a confidence interval of 0.21 to 0.6060.
In relation to palliative care there are few studies of psilocybin. It could be stated that the palliative conditions identified were oncology patients57,56,59 and one study on patients with high impact chronic diseases such as HIV/AID60. In most studies these palliative conditions were related to psychiatric problems of anxiety and depression, but also to such important issues as demoralization, well‐being and acceptance of death. To conclude the studies that addressed the psychiatric problems of depression and anxiety in patients with oncological conditions, evaluating the use of psilocybin were those of Ross, Griffths concluding that there was a significant decrease in the psilocybin intervention groups57,56, versus placebo. In addition, there was improvement in spiritual, emotional aspects as well as in well‐being and acceptance of death. In relation to high impact chronic pathologies, the use of psilocybin decreased demoralization60. Other studies explored the acute effects of psilocybin in healthy people without psychiatric comorbidities56,55 that preceded the studies in palliative patients and that allowed exploring the experiences and emotional dimensions of psilocybin use
Regarding the doses administered, all the studies handled different doses and administration schedules, although low doses could be differentiated from high doses. From this it was concluded that in the long term, high doses allow sustained effects in the improvement of depression, positive attitudes towards life, self‐perception, spirituality and increased acceptance and positive attitudes towards death. While these positive effects are of great importance for palliative patients, this must be weighed against the adverse effects, even more so when dealing with patients with high morbid burden. Consequently, adverse events were associated with high doses, with increased blood pressure and heart rate, headache, nausea and emesis, as well as physical discomfort being the most prevalent56,60,58 (Table 2).
Table 2:
Summary of clinical studies with psilocybin in healthy and palliative patients
|
Research |
Methodology |
Research instruments and measurements |
Conclusions |
|
Palliative patients | |||
|
Lewis, et al59 HOPE trial (A Pilot Study of Psilocybin
Enhanced Group Psychotherapy in Patients with Cancer) |
Open‐label, singlearm, pilot study with
psilocybinassisted group therapy in cancer patients with DSM‐5 depressive
disorder. High dose of 25 mg psilocybin. Included 12 participants. |
It included 3 preparatory sessions, a
high‐dose psilocybin intake session of 25 mg and 3 integration sessions with
12 participants, follow‐up at 2 weeks and 26 weeks. The primary outcome was
the measurement of depressive symptoms, measured by the 17‐ItemHAMD scale at
day 0 and at 2 weeks and 26 weeks after the intervention. Secondary outcomes
included the Functional Assessment of Chronic Illness TherapySpiritual
Well‐Being Scale version 4 (FACIT‐sp) and The Death Transcendence Scale.
MED‐30 (Mystical experience questionnaire) was administered at the end of the
day of dose administration. |
Nausea and headache were reported as adverse
effects. The primary outcome was measured with the HAM‐D scale, 3/12 patients
showed a clinically significant change (4‐6 points) and 7‐12 patients a
clinically substantial change (7‐12 points). 6 of 12 patients showed
remission criteria (<7 points), these outcomes had a statistically
significant p value of <0.001. In relation to the FACIT‐sp. Outcomes with
statistical significance were in the emotional, functional, spiritual
meaning, spiritual peace and spiritual faith dimensions with p <0.05. |
|
Schneider, et al58 |
This is Phase II, single‐center, open label
trial, 30 cancer patients with major depressive disorder received a fixed
dose of 25 mg of psilocybin. |
The NIH‐HEALS, a selfadministered, 35‐item,
measure of psycho‐social spiritual healing was completed at baseline and
post‐treatment at day 1, week 1, week 3, and week 8 following psilocybin
therapy. |
Reported adverse eventsincluded headache
(80%), nausea (40%), tearfulness (27%), anxiety (23%), euphoria (23%),
fatigue (23%), and mild impairment of psychomotor functioning (10%). All
three factors of NIH‐HEALS: connection, reflection & Introspection, and
Trust & Acceptance demonstrated positive change with a p value less than
0,05. |
|
Anderson, et al60 |
Single‐arm, openlabel, single‐arm pilot study
of psilocybin‐assisted group therapy in a group of 18 cisgender men with
HIV/AIDS aged ≥ 50 years and with moderate to severe demoralization assessed
with the Demoralization Scale‐II (DS‐II) of ≥8/32. A dose of 0.3 mg/kg was
administered for cohort 1, 0.36 mg/kg for cohort 2 and 3. Overall the average
dose was 27.1 mg/dose. |
Seven psychotherapy sessions were
conducted. The scale used to evaluate
benefits and harms was a 7point Likert scale. The Schedule of Attitudes
towards Hastened Death (SAHD) was administered at baseline, end‐oftreatment
and 3‐month follow‐up. The Montreal Cognitive Assessment (MoCA) was performed
at enrollment and end‐oftreatment. The primary clinical outcome was
administered at least at baseline, end‐of‐treatment, and 3‐month follow‐up on
the 16‐item self‐report DS‐II. |
Moderate to severe adverse effects reported
were severe anxiety reaction, paranoia, hypotension, and hypertension, but
none were serious.A clinically significant change in demoralization was
detected from baseline to 3‐month follow‐up, with a standardized effect size
of 0.47, and a confidence interval of 0.21 to 0.60. |
|
Ross, et al57 |
Randomized, double‐blind, crossover clinical
trial in which a single dose of psilocybin (0.3 mg/kg) versus a control of
niacin (250 mg) is administered to 29 oncology patients suffering from
anxiety or depression. |
Hospital Anxiety and Depression Scales (HADS)
in its measures of Anxiety, Depression and Total; STAI in its measures of
Trait and State Anxiety; BDI with a baseline measurement, at one week and
then at 26 weeks after the second dose. and at 6.5 months followup. |
The percentages of symptom remission and
anxiolytic/antidepressant response were significantly higher in the
psilocybin group compared to the control. No significant differences were
observed between groups after the administration of psilocybin in the second
session to the group that previously received placebo. At 6.5 months
follow‐up, clinical response rates for measures of anxiety and depression
ranged from 60 to 80% for all participants as a whole and when compared to
baseline. |
|
Griffiths, et al56 |
Randomized, double‐blind, crossover, clinical
trial comparing low to high doses of psilocybin 1 to 3 mg/70 kg vs 22 to 30
mg/70 kg, in 56 patients with a diagnosis of lifethreatening cancer and a DSM
IV diagnosis of anxiety or mood disorder (dysthymia, depression, major, or
mixed). |
Meetings with monitors: before consumption
(mean of 3 sessions), the day after psilocybin consumption (mean of 1.2
hours), meetings between the first and second psilocybin session (mean of 2.7
sessions), and after the second session and followup at 6 months (mean of 2.5
sessions). Monitoring of cardiovascular measures. Questionnaire carried out
by the monitors. HRS (Hallucinogen Rating Scale), 5D‐ASC (Dimension Altered
State of Consciousness), MEQ30 (Mystical Experience Questionnaire, Four
factor scores (Mystical, Positive mood, Transcendence of time and space, and
Ineffability). For depression GRID‐HAM‐D‐17, and anxiety HAM‐A with SIGH‐A.
LAP‐R Death Acceptance, Death Transcendence Scale. |
Adverse effects reported: increased blood
pressure more in high than in low doses (34 vs 17%), nausea and emesis in
high doses (15%), physical discomfort of any type more in high than in low
doses (21 vs 8%). High doses cause cardiovascular changes, as well as result
in visual effects and increased happiness. At the level of depression
questionnaires, in the first session there was no difference between high and
low doses, but in the second session and at 6 months, high doses were more
effective. Measures of spirituality showed similar increases. High doses
produced higher ratings on persistent positive effects on attitudes about
life, self, mood change, behavior change, and spirituality. There were
increases in positive attitudes toward death and acceptance of death, greater
with higher doses. |
|
Grobs, et al52 |
Randomized, single‐blind, crossover,
placebocontrolled clinical trial in patients with advanced cancer and
anxiety. use of moderate doses of psilocybin (0.2mg/kg). 12 adults with
advanced cancer and anxiety (according to DSM‐IV). |
The following scales were used: BID (Beck
Depression Inventory), Profile of Mood States, State‐Trait Anxiety Inventory,
5D‐ASC (5‐Dimension Altered States of Consciousness) y Brief Psychiatric
Rating Scale, with follow‐up at 6 months after treatment. |
Psilocybin administration increased blood
pressure and heart rate. In relation to 5D‐ASC psilocybin had effects on
anxious ego‐dissolution and auditory disturbances. It also led to positive
depersonalization, positive mood, manic experiences, elementary hallucinations,
change of meaning of perceptions, facilitated recall and facilitated
imagination. Psilocybin helped to decrease anxious and depressive symptoms.
Mood improved for 2 weeks, and the sustained improvement in BID was sustained
for 6 months. |
|
Healthy adults without psychiatric disorders | |||
|
Griffiths, et al55 |
Double‐blind, cross‐over, semirandomized
clinical trial. Use of psilocybin doses of 0, 5, 10, 10, 20 and 30 mg/70 kg.
Each participant went through the different doses including the 0 dose which
would be the equivalent to placebo. Eighteen healthy adults with no history
of psychiatric disorders or dependence on alcohol or psychoactive substances
participated. |
Five sessions were carried out, with follow‐up
at 1 month and 14 months. Cardiovascular parameters were measured and
questionnaires were applied by monitors. Use of Hallucinogen Rating Scale,
APZ, which includes three scales: OSE (oceanic immensity: mystical experience
that includes feelings of unity and transcendence), AIA (fear of ego
dissolution), and VUS (visionary). ARCI (Addiction Research Center
Inventory), States of Consciousness Questionnaire, Mysticism Scale, and Death
Transcendence Scale. |
At the level of cardiovascular parameters,
elevated blood pressure was related to higher doses. The subjective effects
related to hallucinogens were experienced with doses starting at 5 mg, but
increased with higher doses, and the mystical experience increased with the
dose. However, with the highest dose 30 mg/70 kg, fear or anxiety was
experienced (39% of participants). Persistent effects were dose‐related. 61%
considered the experience as the most spiritually significant in their lives,
and 83% among their top five experiences.
|
|
Griffiths, et al56 |
Randomized, double‐blind, crossover clinical
trial to evaluate the acute (7h) and long‐term (2 months) effects of
psilocybin (30mg/70kg compared to methylphenidate (40mg/70kg), in 36 healthy
volunteers without psychiatric comorbidities, without previous experience
with hallucinogens. 30 patients had 2 sessions and 6 patients 3 (2 with
methylphenidate, 1 with unblinded psilocybin), the latter 6 were excluded
from the analyses. |
The following scales were used: HRA
(Hallucinogen Rating Scale), APZ, ARCI (Addiction research center inventor),
States of consciousness questionnaire, Mysticism Scale. |
Methylphenidate and psilocybin presented
alterations in heart rate and blood pressure. Psilocybin obtained higher
scores in HRS, APZ, and ARCI. The respective scales of the mythical
experience obtained higher scores for psilocybin. At two months, psilocybin
produced higher scores on positive attitudes, mood, social effects, and
behavior, and at two months the mysticism scales were higher for psilocybin. |
End of life
One of the central
goals of palliative care is to support individuals at the end of life by
addressing suffering and managing symptoms, with pain and anxiety management
being crucial for both patients and their families. Additionally, addressing
existential and spiritual concerns is essential61.
Pharmacotherapy options include opioids, serotoninergic antidepressants,
sedative‐hypnotics and neuroleptics. However, these do not always provide
immediate relief; for example, selective serotonin reuptake inhibitors (SSRIs)
for depression do not have an immediate effect. In fact, in the first wave of
psychedelic studies, these substances were administered to terminal patients at
the end of life.
For the administration of psilocybin, it is recommended to discontinue SSRIs for at least 4 to 5 half‐lives to prevent serotonin syndrome, often referred to as “serotonin washout”. Although the concurrent use with other psychotropic drugs has not been extensively studied, some research suggests that combining psilocybin with haloperidol may lead to experiences of derealization accompanied by agitation and anxiety, while lithium has been associated with seizures. Caution is advised when using steroids concurrently due to the risk of inducing mania and when using opioids due to sedation. Nevertheless, psilocybinʹs potential analgesic effects have also been investigated, with studies indicating pain reduction at low doses61.
Letheby reflects on how these effects are achieved, proposing that reduced fear of death may result from inducing experiences that diminish such fears. She connects this to acute psychedelic experiences, drawing parallels with non‐drug‐induced experiences such as mystical‐religious experiences, meditation and near‐death experiences, which share phenomenological and psychological benefits. Letheby concludes that psychedelic experiences involve relaxation and a reevaluation of abstract, high‐level beliefs about the self and the world. This process can dissolve and reconstruct mental models, potentially leading to decreased fear of death, improved depressive and anxiety symptoms and acceptance of lifeʹs processes62.
Legal Status of
Psilocybin
Psychedelic mushrooms have a long history intertwined with human rituals, religious practices, medical uses and recreational activities. In Mexico, the genus Psilocybe includes 53 known psychedelic species, historically used by indigenous cultures such as the Nahuatl, who referred to them as “teonanácatl”, meaning “flesh of the gods”. This term was first documented by the Franciscan friar Bernardino de Sahagún between 1569 and 1582. However, during the Spanish colonization, the ceremonial use of these mushrooms was suppressed and omitted from literature63
It was not until the 20th century that these mushrooms were rediscovered academically. Austrian physician Blas Pablo Reko first described the religious practices of the Mazatec indigenous people in northern Oaxaca using mushrooms. In 1938, botanist Richard Evans Schultes visited Huautla with Reko to observe ceremonies, collect samples and identify the mushrooms, which were Psilocybe caerulescens, Panacolus campanulatus and Stropharia cubensis. The identity of the Mazatec sacred mushrooms was further elucidated by writer and ethnobotanist R. Gordon Wasson and his wife, mycologist Valentina Pavlovna, who, guided by María Sabina, became the first Westerners to experience and describe the religious and mystical experiences induced by these mushrooms. Mycologist Roger Heim identified the sacred mushroom as Psilocybe and chemist Albert Hofmann isolated the compound, naming it psilocybin63
In the 1950s and 1960s, psilocybin and other psychedelics garnered significant scientific and psychiatric interest. However, political and cultural factors, particularly during the 1970s, led to increased regulation and a halt in research. Recently, there has been renewed interest in the potential of psychedelics for treating psychiatric conditions and psychological effects related to end‐of‐life processes. Psilocybin and MDMA are being studied in conjunction with psychotherapy for their therapeutic potential. Psilocybin shows promise in treating psychological alterations associated with oncological diseases64, depression, anxiety9 and substance addiction.
Psilocybin has been designated as a ʺbreakthrough therapyʺ by the FDA and is currently in Phase II clinical trials. This research involves teams from the US, UK and Switzerland, with initial studies conducted at Johns Hopkins University, New York University and Harbor UCLA. In the UK, the Medical Research Council supported psilocybin studies for treatment‐resistant depression in 2015. COMPASS Pathways is conducting the first large‐scale clinical trial of psilocybin therapy for treatment‐resistant depression in Europe and North America13
Although emerging evidence supports the use of psilocybin for treating PTSD (Gill et al., 2020; Goldberg et al., 2020; Illingworth et al., 2021; Nutt et al., 2020)65,66,14, its efficacy for anxiety disorders, substance abuse and end‐of‐life disturbances is also being explored67.
In Australia, no psychedelic substances have been approved for clinical use. In February 2020, Australiaʹs Therapeutic Goods Administration (TGA) rejected a proposal to reclassify MDMA and psilocybin from Schedule 9 (Prohibited Substances) to Schedule 8 (Controlled Drugs), which are substances with potential for abuse or dependence. Controlled drugs have legitimate therapeutic uses and can be prescribed under strict legislative controls, whereas prohibited substances lack established therapeutic use and are only available for medical or scientific research, analytical, educational or training purposes67.
Countries where psilocybin is decriminalized or legally used include Jamaica, the Netherlands, the Bahamas, Brazil, Nepal, Mexico, Peru and Portugal. In the United States and Canada, its use is restricted to approved clinical studies or religious groups that sanction its use52
Recent systematic reviews have highlighted that the number of studies remains limited, with few participants60,65,67. Additionally, reviews have generally focused on specific diagnoses and it is not always clear if there were language restrictions in bibliographic searches. Only one review assessed the overall credibility of results using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework recommended by Cochrane Collaboration, but this was limited to PTSD and how the guidelines were applied remains unclear [Varker et al., 2021]. For drugs with a rapid onset of action, it is important to consider studies with both inactive and active controls in separate comparisons. This is because if participants are told there will be a brief interval before the onset of any effects, it becomes very apparent that they are receiving an inactive placebo. Thus, there is a risk that any response in the intervention group may be amplified by expectancy effects, while in control groups, it may be diminished by disappointment from receiving a placebo (Muthukumaraswamy et al., 2021). Such contrasting reactions can artificially inflate the treatment effect67.
Perspectives
The future of
psilocybin in palliative care is contingent on several key developments:
·
*Regulatory Changes*: Legislative
shifts toward decriminalization and medicalization of psilocybin are crucial.
Efforts to reclassify psilocybin from a Schedule I substance to a controlled
but medically accessible drug are underway in various regions. Success in these
areas will facilitate broader clinical application and acceptance.
·
*Clinical Research*:
Expanding the body of clinical evidence through large‐scale, randomized
controlled trials will solidify psilocybin’s efficacy and safety profile.
Research should focus on optimal dosing, administration protocols and long‐term
outcomes to establish standardized guidelines for use in palliative care.
·
*Interdisciplinary Integration*:
Incorporating psilocybin therapy into existing palliative care frameworks
requires collaboration across medical, psychological and spiritual
disciplines.
Training healthcare providers in psychedelic‐assisted therapy and integrating
these practices into holistic care models will be vital for seamless adoption.
·
*Public Perception and Education*: Addressing
public misconceptions and educating both the general public and healthcare
professionals about the benefits and risks of
psilocybin is essential.
Initiatives to destigmatize psychedelic therapy and highlight its scientific
backing can foster a more supportive environment for its integration.
·
*Ethical and Cultural Considerations*: Ethical
considerations, including informed consent, patient autonomy and cultural
sensitivity, must guide the use of psilocybin in palliative care. Respecting
diverse cultural perspectives on psychedelics and ensuring equitable access to
therapy will be important as the field progresses
While significant challenges remain, the potential of psilocybin to transform palliative care is immense. Through continued research, advocacy and interdisciplinary collaboration, psilocybin can become a valuable tool in the holistic management of terminal illness, providing relief and comfort to patients during their most vulnerable times
Conclusions
Psilocybin emerges
as a promising therapeutic option in palliative care, demonstrating significant
potential to enhance the quality of life for terminally ill patients. Its
ability to alleviate psychological distress, improve emotional well‐being and
facilitate profound existential experiences highlights its utility beyond
traditional psychiatric applications. Current research supports the notion that
psilocybin can effectively reduce symptoms of anxiety, depression and
existential distress in patients with advanced illness, offering a holistic
approach that addresses both physical and psychological suffering.
The investigation of psilocybin as a therapeutic agent is increasingly complex when considering the potential ʺentourage effectʺ of whole mushroom extracts versus isolated psilocybin. While psilocybin, a naturally occurring tryptamine alkaloid, has shown promise in treating various psychiatric disorders, recent preclinical studies highlight that psilocybin‐containing mushroom extracts (PME) may produce different biological effects compared to chemically synthesized psilocybin (PSIL). Comparative analyses reveal that PME exhibits more pronounced and sustained impacts on synaptic plasticity markers and metabolomic profiles in animal models. Specifically, PME has been observed to significantly increase neuroplasticity‐related proteins such as GAP43 and synaptophysin in several brain regions, effects not as robustly seen with PSIL alone. Additionally, metabolomic data suggest a gradient of metabolic effects between vehicle, PSIL and PME, indicating that the full spectrum of compounds in PME may contribute synergistically to its therapeutic efficacy. This suggests that the complex mixture of bioactive compounds in PME could be crucial for its enhanced effects (ref). Further research is essential to elucidate the specific molecules responsible for these differential effects and to clarify how the entourage effect may influence the therapeutic outcomes of psilocybin‐based treatments.
The integration of psilocybin into palliative care could represent a significant shift in end‐of‐life care paradigms, underscoring the importance of addressing mental and emotional health alongside physical needs. Despite these promising findings, the advancement of psilocybin therapy faces challenges due to existing stigmatization and legal restrictions. Addressing these barriers through continued advocacy, rigorous clinical trials and comprehensive policy reforms is crucial for unlocking the full therapeutic potential of psilocybin.
In the realm of Psychedelic‐Assisted Therapies (PAT), the patient‐doctor relationship plays a pivotal role. The success of such therapies is closely tied to the patients trust in their physician, the thoroughness of the information provided about the medication and the clarity of communication regarding expected physical and psychological effects. Patients must be well‐informed about their diagnosis, treatment alternatives and the specific goals of the prescribed therapy. Ensuring that patients are comfortable with the treatment plan and have the option to discontinue if desired aligns with ethical standards and bioethical principles.
·
To mitigate the risk of adverse reactions,
it is essential to have a comprehensive understanding of the patient’s medical
history, including any psychiatric conditions, recreational substance use and
diagnostic health issues. Rigorous clinical assessments should be conducted to
monitor vital signs and other variables potentially impacted by psilocybin. An
open dialogue regarding the patient’s expectations and concerns about
psilocybin, along with a clear explanation of its potential benefits and risks,
is fundamental.
· Oncological diseases often disrupt patientsʹ lives by affecting their physical capabilities, causing chronic exhaustion and limiting daily activities, while also impacting work and economic situations and eliciting reactions from their support network. In such contexts, a lack of personal relationships or adequate social support can exacerbate existential stress. Addressing these multifaceted challenges with an integrated approach that includes innovative therapies like psilocybin could significantly improve the overall well‐being of patients in palliative care.
Author contributions: Andres David Turizo‐Smith: Methodology, Conceptualization, Validation, Investigation, Resources, Writing ‐ review & editing. Natalia Botero Jaramillo: Conceptualization, Investigation, Writing ‐ original draft. Diana Milena Berrio Cuartas: Writing ‐ review & editing, Investigation, Conceptualization.
Funding: No funding was received for the publication of this work
Acknowledgements: We would like to express our sincere gratitude to Edgar Gonzalez Rodriguez, a cultivator of mushrooms and fungi, for providing us with the photograph of Psilocybe. We also extend our heartfelt thanks to Dr. Viviana Peskin, psychiatrist, for her valuable feedback and thorough review of the draft.
Conflict of
Interest:
The authors declare that there is no conflict of interest regarding the
publication of this article. The research was conducted in the absence of any
commercial or financial relationships that could be construed as a potential
conflict of interest.
Ethics Statement: This
study did not involve human participants or clinical interventions requiring
registration. Therefore, a clinical trial number is not applicable.
References
7.
Huxley J. Transhumanism. J Humanistic
Psycho 1968;8(1):73‐76.
11.
Nichols DE. Psilocybin: from ancient magic
to modern medicine. J antibiotics 2020;73(10):679-686.
44.
Keeler MH. Chlorpromazine
antagonism of psilocybin effect. Int J Neuropsychiatry 1967;3:66-71.
62.
Letheby C. Cómo reducen los psicodélicos el miedo a la muerte? Neuroethics 2024;17:27.
63.
Guzmán G. Hallucinogenic Mushrooms in
Mexico: An Overview. Econ Bot 2008;62:404-412.