Revolutionizing Esophageal Squamous Cell Carcinoma Treatment: Immunotherapy Breakthroughs
ABSTRACT
The
advent of immunotherapy has significantly transformed the treatment paradigm
for esophageal carcinoma. The adjuvant therapy study CheckMate-577 showcased
the PD-1 inhibitor nivolumab's improved disease-free survival (DFS). In first-line treatment, combining PD-1 inhibitor pembrolizumab with chemotherapy
(KEYNOTE-590) yielded heightened overall survival (OS) in PD-L1
positive squamous cell carcinoma patients
with a combined positive
score (CPS ≥ 10). Furthermore, nivolumab, whether with ipilimumab or chemotherapy
(CheckMate 648), exhibited superior OS compared to chemotherapy alone. Two
impactful clinical trials have notably positioned immunotherapy as the
forefront for early-stage treatment in advanced esophageal cancer. These trials,
delving into immune checkpoint inhibitors, disclosed prolonged progression-free
survival compared to conventional treatments, with one trial revealing an
overall survival improvement. While ongoing, these findings prompt experts in
esophageal cancer to envision potential establishment of new standard treatment
protocols for this historically challenging cancer type. On March 22, 2021, FDA
approval was granted for pembrolizumab (Keytruda) combined with chemotherapy
for specific esophageal or gastroesophageal cancer patients ineligible for
surgery, traditional chemotherapy, or radiation. This regulatory decision
rested upon insights from the KEYNOTE-590 trial, detailed in the Cancer
Currents report. This review seeks to synthesize these trials, integrating them into prevailing treatment strategies, possibly
introducing early
immunotherapy for advanced esophageal cancer.
Keywords:
Immunotherapy; Pembrolizumab; Esophageal cancer; Adenocarcinoma
INTRODUCTION
Esophageal cancer is one of the deadliest disease
with the universal
health burden over 60,0000 new
cases which gets reported annually1. Esophageal cancer is divided in two
subtypes squamous cell cancer (ESCC) mainly in the proximal part of esophagus
and Adenocarcinoma mostly in the distal part. Squamous subtype of esophageal
cancer comprises 90% of all carcinomas and remaining 10% is adenocarcinoma. There
is also one third subtype small cell carcinoma which is very rare and have not much studied
in depth. In this particular literature review we are going to emphasize
on esophageal squamous cell cancer.
Current
clinical trials have shown very promising results of immunotherapy in the early
treatment of advanced ESCC. Two large trials have demonstrated that immune
checkpoint inhibitors have extended the duration, that individuals with advanced
esophageal cancer lived
without their cancer
progressing when compared to
standard treatments2. In one of these studies, patients who received a checkpoint
inhibitor not only experienced extended progression-free survival but
also lived longer overall. These promising results indicate that immunotherapy may indeed become a crucial
component of early treatment for specific individuals
with advanced esophageal cancer.
Before
the advent of checkpoint inhibition in ESCC treatment, the standard approaches
were neoadjuvant radiochemotherapy along with surgery for localized cases. Definitive
chemoradiation for those cases where surgery was not an option but systemic
chemotherapy for advanced metastatic disease3. As per CROSS trial ,which is referenced in3,4 patients with locally advanced resectable ESCC underwent
preoperative treatment which involves five weekly cycles of carboplatin and paclitaxel along
with concurrent radiotherapy (totaling 41.4 Gy in 23
fractions) followed by surgery. The study revealed that the neoadjuvant
chemoradiotherapy approach let to a substantial 10- year overall survival
benefit, demonstrating a notable 13% increase in survival. This likely suggests
that the treatment approach had a significant positive impact on long-term
survival in patients with ESCC3,4.
Following
the failure of a palliative first-line treatment for squamous cell carcinoma,
there is a lack of randomized trials that definitively establish the benefits
of chemotherapy compared to best supportive care (BSC). In such cases, when
patients have a good performance status, common chemotherapy options
often include irinotecan, paclitaxel or docetaxel. These treatments are frequently utilized in the absence of
concrete evidence from randomized trials5,6.
The introduction of immune checkpoint inhibitors (ICIs) has had a huge impact on the treatment approach for ESCC.
These inhibitors have significantly altered the treatment regimen. Key
components in this immunotherapeutic approach involve the programmed death
receptor 1 (PD-1) and its ligand (PD-L1), which play an important role in
regulating the immune response against cancer cells. This mechanism has led to
a transition in the way ESCC has been managed. When cytotoxic T-cells bind to
cancer cells through their PD-1 receptor interacting with its ligand,
PD-L1, on the surface of cancer cells, it enables those cancer cells to
evade the anti-tumor immune response. This interaction acts as a mechanism by
which cancer cells can suppress the immune system’s ability to target and
destroy them, contributing to the progression of the tumor. Immune checkpoint
inhibitors work by blocking this interaction, thereby restoring the immune
response and enhancing the body’s ability to combat cancer cells7.
Certainly,
based on the background this narrative review’s
main objectives are overview of immunotherapy in ESCC
which aims to present the current landscape of immunotherapy, role of ICIs and there utilization in treatment and how to include them at different
stages of cancers and also significance of promising biomarkers in ESCC
immunotherapy which can be essential for patient selection and treatment
response prediction.
OVERVIEW OF IMMUNE CHECKPOINT INHIBITORS
The
immune system has built-in safeguards to prevent it from attacking healthy cells.
Checkpoint proteins act as switches
that regulate immune responses. Cancer cells can
manipulate these switches to evade immune attacks. Immune checkpoint inhibitor
drugs target these proteins, potentially restoring the immune system's
ability to target
and combat esophageal cancer cells.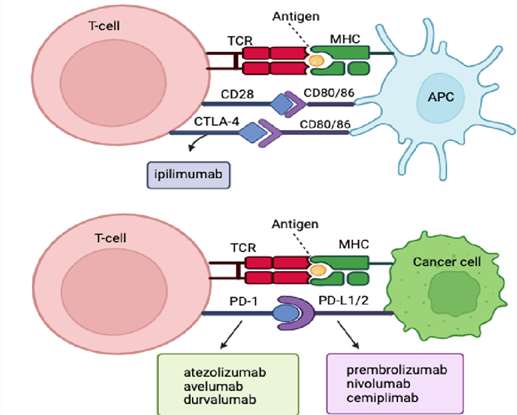
Figure 1. The Function of Immune Checkpoint
Inhibitors in Cancer Treatment
PD-1 Inhibitors
Pembrolizumab
(Keytruda) and nivolumab (Opdivo) are medications that specifically target
PD-1, a protein found on T cells, a component of the immune system.
PD-1 usually serves to prevent
T cells from attacking healthy cells. By inhibiting PD-1, these
drugs enhance the immune system's ability to target and combat cancer cells. This mechanism can lead to tumor shrinkage or a reduction in their growth
rate7,8.
Pembrolizumab is employed in the treatment
of certain advanced
esophageal or gastroesophageal junction (GEJ) cancers, particularly when
standard treatments like surgery
and chemoradiation are not viable
options. Depending on the
specific circumstances, Pembrolizumab may be administered as a standalone
treatment or in combination with chemotherapy. Nivolumab has several
applications in the treatment of esophageal and gastroesophageal junction (GEJ)
cancers:
· For individuals who underwent chemotherapy and radiation (chemoradiation) before surgery and have
residual cancer following surgery.
· As a standalone treatment for advanced
squamous cell cancer of the esophagus, often after attempting
chemotherapy.
· In combination with chemotherapy as the initial treatment for
advanced squamous cell cancer
of the esophagus, potentially extending survival for some
patients.
· In combination with chemotherapy for those with advanced
adenocarcinoma of the esophagus or advanced cancer
of the gastroesophageal junction (GEJ).
CTLA-4 Inhibitor
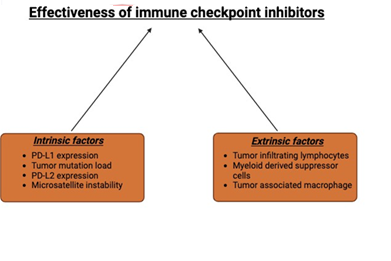
Figure 2. Effectiveness of immune checkpoint inhibitors
GENETIC SIGNATURES FOR IMMUNOTHERAPY
In cases of metastatic ESCC where the prognosis is often poor due to its aggressive nature of the disease, selecting the best treatment approach is of utmost importance. This decision involves creating a well-defined and evidence-based plan for therapy to ensure that patients receive the most effective and individualized treatment options. So the outcomes of significant clinical trials indicate that there exists a subset of patients referred to as "responders" who experience greater benefits from immunotherapy. These individuals demonstrate a more favorable response to immunotherapeutic treatments compared to others. Conversely, these trials have also revealed that there is a group of patients for whom immunotherapy does not provide any discernible benefits. Indeed, in these patients, the application of ICIs may potentially exacerbate their medical condition. Several factors contribute to this, including the extended time it takes for some patients to respond to immunotherapy and the possibility of hyper progression, a phenomenon where the disease progresses even more rapidly during treatment9,10.
Hence, research has been focused on exploring predictive and prognostic molecular biomarkers in recent years. These markers are essential for identifying suitable candidates for immunotherapy11,12.
In the context of immunotherapy, the expression of PD-L1 is a significant factor. Nevertheless, landmark trials in this area have demonstrated substantial variation in how PD-L1 is assessed, scored, and the threshold values used. This lack of standardization has resulted in inconsistencies in its evaluation. In this context, a systematic review and meta-analysis by Leone AG et al. has been published. This review encompassed data from 5,257 patients who participated in ten randomized controlled trials involving immunotherapy for advanced Esophageal Squamous Cell Carcinoma (ESCC)13. In this analysis, the use of immunotherapy demonstrated improved survival outcomes (Hazard Ratio: 0.71), and this effect was consistent across different regions (Asian versus non-Asian) and treatment lines (first-line versus subsequent lines). Additionally, immunotherapy led to enhancements in Progression-Free Survival (PFS) and Objective Response Rate (ORR) with Hazard Ratio (HR) of 0.78 and an odds ratio of 1.5, respectively.
ESCC Represents a Distinct Medical Condition.
The KEYNOTE-590 trial, which compared pembrolizumab plus chemotherapy to chemotherapy alone, significantly influenced clinical practice. However, a challenge emerged from including both (ESCC) and esophageal or Siewert type I Gastroesophageal Junction (GEJ) adenocarcinomas in the same trial, with ESCC accounting for 73% of the patients. Given its strong association with DNA damage from smoking and alcohol, ESCC displays heightened sensitivity to immunotherapy compared to gastroesophageal adenocarcinomas, highlighting the need to consider ESCC as a distinct and separate disease14.
The initial results show improved median Overall Survival (OS) in various groups. Patients with Esophageal Squamous Cell Carcinoma (ESCC) and a Programmed Death-Ligand 1 (PD-L1) Combined Positive Score (CPS) of 10% or higher exhibited notably increased OS by 13.9 months as compared to 8.8 months.
Similarly, the overall ESCC patient group also experienced enhanced OS by 12.6 months as compared to 9.8 months. This positive trend extended to all patients with a PD-L1 CPS of 10% or higher which is 13.5 months as compared to 9.4 months and the entire study population is 12.4 months as compared to 9.8 months. However, in an exploratory analysis, patients with a PD-L1 CPS less than 10% did not experience a significant improvement in median OS when pembrolizumab was added to chemotherapy showed 10.5 months as compared to 10.6 months14.
Data from the CheckMate 648 trial, presented at the 2021 ASCO Annual Meeting, suggests that Nivolumab and Ipilimumab are potential first-line treatments for advanced (ESCC). The trial included three arms: chemotherapy alone, Nivolumab with chemotherapy, and Nivolumab with Ipilimumab. Both Nivolumab and chemotherapy and Nivolumab and Ipilimumab showed improved Overall Survival (OS) compared to chemotherapy, especially in patients with a Programmed Death-Ligand 1 Combined Positive Score (PD-L1 CPS) of 1% or higher and in all patients in the study. These innovative combinations were generally well-tolerated, with Nivolumab and chemotherapy showing slightly more toxicity than chemotherapy alone. Thus the drawback of this study was its lack of blinding15.
Adjuvant Immunotherapy Enhances Disease-Free Survival
The standard treatment for esophageal and gastroesophageal junction (GEJ) cancer has been trimodality therapy, which involves neoadjuvant chemoradiation followed by surgery. Despite this approach, recurrence rates have been notably high, especially for individuals with remaining pathologic disease. The lack of validated adjuvant therapies led to ongoing surveillance as the standard of care.
CheckMate-577, a phase III trial, was the first to demonstrate a significant survival benefit in the adjuvant setting. Updated findings presented at the ESMO 2021 Congress and subsequently published in The New England Journal of Medicine confirm that adjuvant nivolumab continues to enhance disease-free survival compared to a placebo (median, 22.4 vs. 10.4 months)16. As a result, the FDA approved nivolumab in May 2021 for patients with fully removed ESCC or (GEJ) cancer who still have some remaining disease after neoadjuvant chemoradiotherapy. Nivolumab was safe and, importantly, didn't negatively affect the patients' quality of life8. It's very much uncertain whether these findings can apply to patients who have received perioperative chemotherapy, like the FLOT regimen, commonly used in the treatment of (GEJ) cancer in certain centers. For those patients who faces disease recurrence shortly after adjuvant nivolumab, the optimal treatment approach remains uncertain. However, a reasonable strategy involves providing chemoimmunotherapy to patients who experiences relapse of more than 6 months after completing adjuvant therapy and using chemotherapy alone for those with disease recurrence during or within 6 months of time.
FUTURE DIRECTIONS
In the past couple of years, significant progress has been made in the field of gastroesophageal cancers, yet there remains a substantial demand for further advancements. Likely, there is a pressing need for improved biomarkers to accurately identify the patients who truly gain benefits from immunotherapy. Recent understanding suggests that gastroesophageal cancers differ from melanoma, indicating the existence of potentially more effective immune checkpoints that should be the focus of targeted therapies. There is a promising lineup of agents in development that target immune checkpoints like CD40, CD73, and many more. It is hoped that one of these will lead to the next significant breakthrough in the field. While we wait for a major breakthrough, it's important to recognize the value of adjuvant therapy in treating gastroesophageal cancer and the accomplishment of surpassing a 1-year survival rate in metastatic cases.
REFERENCES
1. Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN Estimates of incidence and mortality worldwide for 36 cancers in 185 Countries. CA Cancer J Clin 2021;71(3):209-249.
2. Lordick F, Mariette C, Haustermans K, Obermannová R, Arnold D. Oesophageal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 2016;27:50-57.
3. yck BM, van Lanschot JJB, Hulshof M, et al. Ten-Year outcome of neoadjuvant chemoradiotherapy plus surgery for esophageal cancer: The randomized controlled cross trial. J Clin Oncol 2021;39:1995-2004.
4. Van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 2012;366(22):2074-2084.
5. Moehler M, Maderer A, Thuss-Patience PC, et al. Cisplatin and 5-fluorouracil with or without epidermal growth factor receptor inhibition panitumumab for patients with non-resectable, advanced or metastatic oesophageal squamous cell cancer: A prospective, open-label, randomised phase III AIO/EORTC trial (POWER). Ann Oncol 2020;31:228-235.
6. Luo H, Lu J, Bai Y, et al. Effect of Camrelizumab vs. Placebo added to chemotherapy on survival and progression-free survival in patients with advanced or metastatic esophageal squamous cell carcinoma: The ESCORT-1st randomized clinical trial. JAMA 2021;326(10):916-925.
7. Bonotto M, Garattini SK, Basile D, et al. Immunotherapy for gastric cancers: Emerging role and future perspectives. Expert Rev Clin Pharmacol 2017;10:609-619.
8. https://www.cancer.org/cancer/types/esophagus-cancer/treating/immunotherapy.html
9. Shah MA. Bennouna J, Doi T, et al. KEYNOTE-975 study design: A Phase III study of definitive chemoradiotherapy plus pembrolizumab in patients with esophageal carcinoma. Futur Oncol 2021; 17(10):1143-1153.
10. Sun JM, Shen L, Shah MA, et al. KEYNOTE-590 Investigators. Pembrolizumab plus chemotherapy versus chemotherapy alone for first-line treatment of advanced oesophageal cancer (KEYNOTE-590): A randomised, placebo-controlled, phase 3 study. Lancet 2021;398:759-771.
11. Metges, JP, Kato K, Sun JM, et al. First-line pembrolizumab plus chemotherapy versus chemotherapy in advanced esophageal cancer: Longer-term efficacy, safety, and quality-of-life results from the phase 3 KEYNOTE-590 study. J Clin Oncol 2022;40:241.
12. Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Efficacy and safety in key patient subgroups of nivolumab (NIVO) alone or combined with ipilimumab (IPI) versus IPI alone in treatment-naıve patients with advanced melanoma (MEL) (CheckMate 067) Eur J Cancer 2015;51:664-665.
13. Leone AG, Petrelli F, Ghidini A, Raimondi A, Smyth EC, Pietrantonio F. Efficacy and activity of PD-1 blockade in patients with advanced esophageal squamous cell carcinoma: A systematic review and meta-analysis with focus on the value of PD-L1 combined positive score. ESMO Open 2022;7(1):100380.
14. Wakelee HA, Altorki NK, Zhou C, et al. IMpower010: Primary results of a phase III global study of atezolizumab versus best supportive care after adjuvant chemotherapy in resected stage IB-IIIA non-small cell lung cancer (NSCLC). J Clin Oncol 2021;39:8500.
15. Doki Y, Ajani JA, Kato K, et al. Nivolumab combination therapy in advanced esophageal squamous-cell carcinoma. N Eng J Med 2022;386:449-462.
16. Gyawali B, Hey SP, Kesselheim AS. Evaluating the evidence behind the surrogate measures included in the FDA's table of surrogate endpoints as supporting approval of cancer drugs. E Clinical Medicine 2020;21:100332.