Sarcomatoid Renal Cell Carcinoma with Rhabdoid Features Presenting as Xanthogranulomatous Pyelonephritis
Abstract
Renal cell carcinoma (rcc) encompasses a diverse range of kidney malignancies, with clear cell rcc being the most common subtype. This article discusses a rare case of a 61-year-old male initially suspected of having xanthogranulomatous pyelonephritis, ultimately diagnosed with sarcomatoid rcc featuring focal rhabdoid differentiation. The aggressive nature of this case highlights the importance of thorough diagnostic procedures. Rcc, representing 90% of kidney cancers, has various histological subtypes with distinct grading systems. Sarcomatoid and rhabdoid rcc, though rare, can occur in any subtype and are associated with poorer prognosis. Genetic alterations contribute to their aggressiveness. Current guidelines recommend imaging and biopsy for rcc diagnosis, with surgical management and surveillance for high-risk features. Emerging treatments like pembrolizumab and combination therapies signify a shifting paradigm in rcc management, necessitating tailored approaches based on histology and risk factors.
Keywords: carcinoma; renal cell; clear cell; neoplasm; sarcomatoid; histology; immunohistochemistry
Introduction
Renal cell
carcinoma (rcc) comprises a diverse range of cancers originating from renal
tubular epithelial cells1, with more than 10 distinct histological subtypes
identified1,2. Clear cell rcc (ccrcc) is the predominant subtype, followed
by papillary and chromophobe, respectively3. Sarcomatoid dedifferentiation, can occur in up to 15% of
rcc patients4, it is characterized by a transformative growth pattern of
the epithelial neoplasm into malignant spindle-shaped cells, exhibiting
aggressive behavior5, this tumor manifests as sheets of malignant spindle
cells with immunohistochemical and ultrastructural features resembling both
stromal and epithelial cells5, initially considered a distinct histological subtype,
sarcomatoid transformation is now recognized in current classification systems
as a characteristic with the potential to arise in any subtype of rcc6,7. Rhabdoid
differentiation is also observed in rcc, most frequently in clear cell rcc8. Pure rhabdoid rcc
is an uncommon and highly aggressive malignancy of the pediatric population. In adults, pure rhabdoid rcc is extremely
rare, while rcc with rhabdoid features are more commonly found alongside
clear-cell carcinoma. Very few studies report rhabdoid features alongside
sarcomatoid rcc. We present one such patient in whom
renal mass was found to have sarcomatoid rcc with focal rhabdoid features which
mimicked xanthogranulomatous pyelonephritis.
Case presentation
A 61-year-old male with a
history of atrial fibrillation and heart failure. Admitted to the hospital due
to progressive bilateral lower limb swelling secondary to worsening acute
kidney injury. On work up, renal ultrasonography showed enlarged right kidney
measuring 20*13 cm with multiple complex hypoechoic lesions largest 10 cm, with
suspected etiology of xanthogranulomatous pyelonephritis (xgp). On history the
patient wasn’t complaining from any right sided abdominal/flank pain,
hematuria, weight loss or any urinary symptoms.
History of anemia hb 7.5,
been evaluated previously by hematologist, with bone marrow biopsy and flow
cytometry, which ruled out mds and myeloproliferative disorder. Further imaging
was done, computed tomography (ct) of the abdomen and pelvis without
intravenous contrast due to the impaired renal function, showed abnormal
appearing right kidney, with multiple cystic and dense appearing regions
measuring 17 cm in length, with retroperitoneal, enlarged lymph nodes adjacent
to the ivc up to 3 cm.
In addition, ct chest showed
suspicious pulmonary nodules for metastasis, a trial of ir guided drainage of
the suspected abscess vs mass was done, which was inconclusive for any
malignancy. Laboratory results are shown in (table 1).
Table 1. Laboratory results
|
|
|
Normal range |
|
Hb |
7.5 g/dl |
13.5-17 g/dl |
|
Mcv |
87 fl |
80-100 fl |
|
Serum iron |
16 mcg/dl |
65-175 mcg/dl |
|
Ferritin |
996 ng/ ml |
24-336 ng/ ml |
|
Iron
saturation |
12.7 % |
20-50% |
|
Tibc |
126 mcg/dl |
250-450 mcg/dl |
|
Ldh |
99 u/l |
125-220 u/l |
|
Esr |
98 mm/hr |
0-20 mm/hr |
|
Crp |
19.8 mg/l |
<10mg/l |
|
Creatinine
clearance |
30 ml/min |
>90 ml/min |
Renal nuclear scan (99 m mag3 scan) showed negligible right kidney function, which supported the decision to proceed with nephrectomy. Pathological exam of the right radical nephrectomy showed poorly differentiated sarcomatoid renal cell carcinoma with focal rhabdoid features, involving the right entire kidney with extension into the renal sinus and perirenal fat with extensive necrosis, focal tumor invasion into the renal vein, but not lumen, negative extension into the adrenal gland and ureteral resection margin with staghorn calculus of the renal pelvis with abscess formation.
Positive right
retroperitoneal lymph node for poorly differentiated sarcomatoid, and rhabdoid
rcc, infiltrating, fibroadipose tissue and regional blood vessels. Tumor
markers with positive for, ck7, vimentin, ae1/ae3, pax8, cd10, negative for
ck20, rcc, p40, gata-3. Pathological
stage pt3a n1, mx (figures 1 and 2)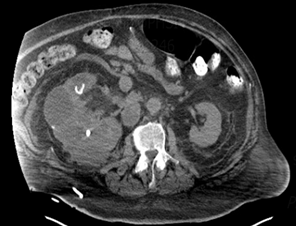
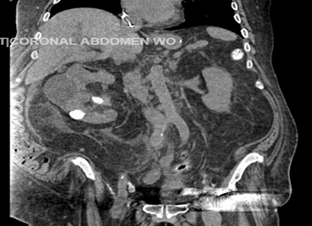
Figure 1. Computed tomography of the abdomen and pelvis with oral contrast.
Showing multiple cystic and solid appearing areas, dense
calcifications in the inferior aspect.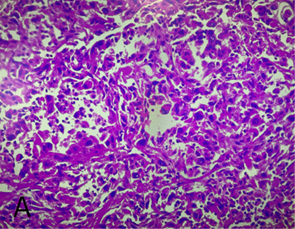
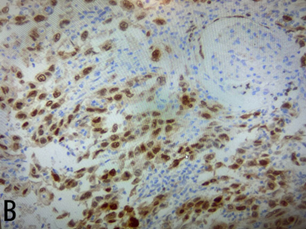
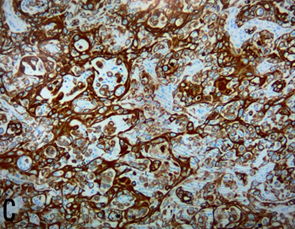
Figure 2. Histological findings
A: h/e stain, tumor shows moderate atypical spindle cells
forming fascicles and rhabdoid features with no epithelial component, b: pax8
slide, c: positive for vimentin
The decision was made to refer the patient to a highly
specialized cancer center due to the poor prognosis and requirement of an
experienced center to tackle such an aggressive tumor, to our best current
knowledge the patient passed away after 2 months of diagnosis and didn’t
receive any treatment.
Discussion
Rcc is the
most common kidney cancer, accounts for ~ 90% of all kidney cancer cases. In
2023, the estimated incidence of rcc is 81,800, resulting in nearly 14,890 of
annual deaths9
according to the classical histopathological classification, rcc can be
classified into three main groups: clear cell carcinoma (ccrcc ) which
accounting for 75% of cases, papillary renal cell carcinoma (prcc) making up
15–20% of cases, and chromophobe cell renal carcinoma (chrcc) representing 5%
of cases10. Multiple grading systems have
been used to stratify rcc [11] the who/isup grading system, introduced as a
replacement for the fuhrman grading system for rcc, relies on nucleolar
prominence alone to identify grade 1 to 3 tumors. Conversely, extreme nuclear
pleomorphism, sarcomatoid morphology, rhabdoid morphology are utilized to
identify grade 4 tumors12,13. Tumor grade has been considered an independent prognostic
factor for rcc, where higher grades carry worse prognosis14.
Both
sarcomatoid and rhabdoid rcc can arise in any type of rcc, more commonly in
ccrcc, sarcomatoid dedifferentiation accounts up to 15% of rcc cases and the
incidence of rhabdoid transformation within rcc is 4%15,13 with a mean age of early 6015
Multiple
genetic alterations that are alterations independent of those fundamental to
original rcc tumor formation have been identified in sarcomatoid and rhabdoid
differentiation these genetic alterations
include chromosomal rearrangements such as loss of chromosomes 9q, 15q, 18p/q and 22q[16], gains of 1q and
8q have been associated with metastatic disease17,18 in
sarcomatoid case, in rhabdoid tumors loss of bap1 or pbrm1 on chromosome
3p has been also noticed19-21 and loss
of chromosome 9, loss of chromosome 11q and loss of chromosome 17p22. These genetic alterations have been associated with poor outcome in renal cell carcinoma with
rhabdoid and sarcomatoid features22-24, it tends to
have an aggressive behavior with high tendency for early metastasis, causing
a rapidly fatal outcome with a median survival rate of 8 in rhabdoid rcc25 and 4-12 months
in sarcomatoid rcc.
Management of stage i
disease is primarily surgical with either partial or radical nephrectomy, more
frequent surveillance imaging studies are recommended post-surgery in srcc
patients26. Radical nephrectomy
is preferred in locoregional rcc with sarcomatoid features, though even with
early surgical management in localized srcc; patients faced a 72% recurrence
rate at a median time of recurrence of 26.2 months27,
for patients with advanced disease; cytoreductive nephrectomy is recommended,
as it showed improved survival with a median of 10.2 months in comparison to
5.5 months in patients who did not undergo surgery28.
No benefit of radiotherapy on overall survival was demonstrated in comparison
to surgical management alone29.
Systemic cytotoxic chemotherapy failed to demonstrate improved survival for
patients with srccs, while targeted therapy with vegf inhibitors (i.e
sunitinib, sorafenib, axitinib, pazopanib, tivozanib or bevacizumab) only
showed limited overall response rates of 11-19%, even with combination of
cytotoxic and targeted therapy response rates remained modest at best30. This resulted in increasing interest in
studying the role of immune checkpoint inhibitors. Expression of pd1/pdl1 was
shown to be higher in srccs (54%) in comparison to non-sarcomatoid rcc (17%),
moreover retrospective subgroup analysis from checkmate 214,keynote-426 studies
demonstrated higher rates of pdl1 positivity 51%, 74.5–79.6% respectively30,31; therefore multiple trials have been
done showing promising results for immune checkpoint inhibitors as a treatment
modality for advanced srcc, with 44% mortality risk reduction, and overall
response rate of (52.6%) over
sunitinib’s (20.7%) which was considered
the standard of care then32, systemic therapy with axitinib +
pembrolizumab or ipilimumab + nivolumab
or axitinib + avelumab or atezolizumab+bevacizumab was studied in the keynote-42633 checkmate 21434 ,javelin renal 10135
immotion15136 trials
respectively, those therapies persistently showed improved outcomes as
demonstrated in subsequent meta analyses30,32.
Disclosures
Ethics approval and consent to participate: not applicable
Consent for publication: not applicable
Availability of data and materials: not applicable
Conflicts of interest: no conflict of
interest
Funding: not applicable
Acknowledgements: not applicable
Authors contributions
Writing, review and editing: shatha
elemian, nooredeen isbeih, bader al omour, sawjanya kalluri, amr ramahi,
Supervision and critical review: gunwant
guron, hamid shaaban
References
1. Eble jn, sauter g, epstein j, sesterhenn i. World health organization
classification of tumours. Pathology and genetics of tumours of the urinary
system and male genital organs. 2004;68-69.
23.
Humphrey pa. Renal cell carcinoma with rhabdoid features. J urology 2011;186(2):675-676.
26. National comprehensive cancer network (nccn). Kidney cancer
version 2.2024-january