Sildenafil for Worsening Group 3 Pulmonary Hypertension in CPFE: A Case of Rapid Recovery from Acute Hypoxic Respiratory Failure: Is RV-PA Uncoupling the Key
Abstract
We report the case of
an 81-year-old male with combined pulmonary fibrosis and emphysema (CPFE),
idiopathic pulmonary fibrosis (IPF) and WHO Group 3 pulmonary hypertension
(PH), who presented with acute on chronic hypoxic respiratory failure. Imaging
revealed sub-segmental pulmonary embolism (PE) and right heart catheterization
confirmed worsening PH with poor right ventricular-pulmonary artery (RV-PA)
coupling. Despite the severity of his condition, the patient demonstrated rapid
and sustained improvement in oxygenation following the initiation and titration
of sildenafil. This case highlights the diagnostic and therapeutic challenges
in managing CPFE with superimposed PE and underscores the potential role of
pulmonary vasodilator therapy in select patients with advanced Group 3 PH.
Keywords: Combined pulmonary fibrosis and emphysema, pulmonary
hypertension, sildenafil, pulmonary embolism, RV-PA coupling, case report
Introduction
Combined pulmonary fibrosis and emphysema (CPFE) is a
distinct clinical entity characterized by upper-lobe emphysema and lower-lobe
fibrosis, often associated with severe pulmonary hypertension (PH) and poor gas
exchange. PH in CPFE is typically classified as WHO Group 3 and is associated
with increased morbidity and mortality1. Acute decompensation in
CPFE may result from infection, thromboembolic events or disease progression.
The role of pulmonary vasodilators in Group 3 PH remains controversial due to
concerns about ventilation-perfusion mismatch, but emerging evidence suggests
potential benefit in select patients with right ventricular (RV) dysfunction
and poor RV-PA coupling2-4.
Case Presentation
An 81-year-old male with chronic hypoxic
respiratory failure secondary to CPFE, IPF and WHO Group 3 PH presented to the
emergency department with worsening dyspnea and non-massive hemoptysis. His
baseline oxygen requirement was 8-10 L/min. On arrival, he was hypoxic with
saturations in the 70s despite 10 L/min of oxygen. He was immediately placed on
heated high flow oxygen at 60% FiO2 and 40 l/min. He denied any recent travel,
sick contacts.
His medical history included dyslipidemia,
benign prostatic hyperplasia, depression, hypothyroidism, transient ischemic
attack, non-obstructive coronary artery disease, peripheral vascular disease,
glaucoma and meralgia paresthetica. He had a 60–80 pack-year smoking history,
quit in 2021 and had occupational exposure to fumes from car sales. He denied
bird exposure and had served in the National Guard. He has been following with
pulmonology clinic for the last 6 months. His ambulatory medications include Nintedanib
150 mg BID for pulmonary fibrosis, Inhaled Treprostinil 64mcg QID for his
pulmonary hypertension. It could not be titrated up due to adverse effects.
On examination, he was in mild to moderate
respiratory distress, requiring heated high-flow oxygen at 40 L/min and 60%
FiO₂. He was hemodynamically stable with blood pressure of 114/70, respiratory
rate of 26/min and afebrile. Physical exam revealed bilateral crepitations, a
systolic murmur and digital clubbing. Neurologically, he was alert and
oriented.
CT pulmonary angiography revealed acute
sub-segmental PE in the right lower lobe and possible embolism in the left
lower lobe. Background findings included a UIP-pattern of fibrosis, upper-lobe
emphysema, bilateral traction bronchiectasis and mildly enlarged mediastinal
and hilar lymphadenopathy (Figures 1-4).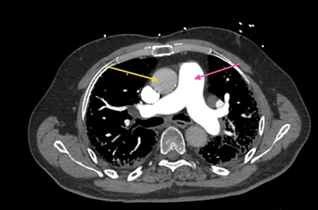
Figure 1: CT Chest with IV contrast, axial view, soft tissue
window, at the level of pulmonary artery demonstrating enlarged pulmonary
artery compared to aorta, which is highly suggestive of pulmonary arterial
hypertension

Figure 2: CT Chest with IV contrast, axial view, soft tissue
window, at the level of ventricles demonstrating enlarged right ventricle (pink
arrow) compared to left ventricle (yellow arrow), which is highly suggestive of
pulmonary arterial hypertension or right ventricular overload
Figure 3: CT Chest with IV contrast, coronal view, lung window,
demonstrating severe emphysema in the upper lobes (yellow arrow) and honey
combing in the lower lobe’s indicative of fibrosis (pink arrow). This pattern
is consistent with Combined Pulmonary fibrosis with emphysema 
Figure 4: CT Chest with IV contrast, coronal view,
lung window, demonstrating severe emphysema in the upper lobes (orange arrow)
and severe honey combing in the lower lobe’s indicative of fibrosis (blue
arrow). This pattern is consistent with Combined Pulmonary fibrosis with
emphysema
He was started on
heparin infusion, empiric antibiotics and corticosteroids and admitted to a
medical progressive unit. Antibiotics and steroids were discontinued after
negative infectious workup (negative procalcitonin, negative CRP) and stable
radiographic findings when compared to his old Computer tomography scans. His
ambulatory medications were continued which included Nintedanib, Inhaled
Treprostinil and other inhaled therapies-Budesonide, Arformoterol and
Revefenacin. Heparin was later transitioned to Apixaban after hemoptysis
subsided.
His Pulmonary
function tests obtained 3 months prior showed preserved FVC (110%) and FEV₁
(113%) with reduced TLC (75%) and severely decreased DLCO (20%). A six-minute
walk test revealed a distance of 259 meters with a nadir SpO₂ of 91% on 10
L/min oxygen.
Echocardiography on
the day of admission revealed normal left ventricular function (LVEF 55–60%),
RV enlargement with mildly reduced systolic function and estimated pulmonary
artery systolic pressure of 75 mm Hg with diastolic septal flattening
suggestive of increased right ventricular filling pressures, enlarged right
atrium and moderate tricuspid regurgitation. The TAPSE/SPAP ratio was 0.272 (Figures
5-9).
Figure 5: 2D Echocardiogram, Apical 4 chamber view demonstrating enlarged right
ventricle (pink arrow) compared to left ventricle (yellow arrow). As can be
seen here, the majority of the apex in this view is dominated by right
ventricle suggesting right ventricular overload
Figure 6: 2D Echocardiogram, Parasternal
short axis view demonstrating enlarged right ventricle (pink arrow) compared to
left ventricle (yellow arrow) confirming right ventricular overload
Figure 7: 2D Echocardiogram, Color doppler
demonstrating tricuspid regurgitation (green arrow), an indirect marker of
severity of pulmonary hypertension
Figure 8: 2D Echocardiogram, Continuous
wave doppler estimated tricuspid velocity of 3.88m/sec highly suggestive of
pulmonary hypertension
Figure
9: 2D
Echocardiogram, Tissue doppler imaging of the right ventricle
Right heart catheterization
confirmed severe PH with mean PAP (Pulmonary artery pressure) of 31 mmHg, PVR
(pulmonary vascular resistance) of 10 WU, Right ventricle DP/DT of 528 and Fick
cardiac index of 1.49 L/min/m². Left ventricular stroke work index was noted to
be 36.71. The right heart catherization findings were significantly worse than
the one that was obtained 3 months ago which revealed a PVR of 3.5 WU and
cardiac index of 2.5 L/min/m2. His prior coronary angiogram obtained 3 months
ago revealed 30-40% stenosis of left main coronary arteries, otherwise only
mild irregularities were noted in the rest of the coronary arteries.
Due to persistent hypoxia despite
anticoagulation for 72 hours needing heated high flow at 60% FiO2 and 40 l/min,
sildenafil 20 mg TID was initiated, as a therapeutic trial after a shared
decision process involving risks versus benefit. Within six hours of sildenafil
initiation, FiO₂ requirements decreased from 60% to 40%. The dose was further
increased after 48 hours to 40 mg TID without adverse effects. Oxygen needs
returned to baseline within 24 hours needing 8- 10 l/min in the next 36 hours.
The patient declined referral to
a pulmonary hypertension center due to travel concerns. He was discharged on
sildenafil 40 mg TID and inhaled Treprostinil. At three-month follow-up, he
remained stable on baseline oxygen and medications. A perfusion scan showed low
probability of pulmonary embolism.
Discussion
Combined pulmonary fibrosis and
emphysema (CPFE) is a complex and under recognized syndrome characterized by
the coexistence of upper-lobe emphysema and lower-lobe fibrosis, typically in
older male smokers. It is associated with relatively preserved spirometry but
severely reduced diffusing capacity (DLCO), profound hypoxemia and a high
prevalence of pulmonary hypertension (PH), particularly WHO Group 3 PH due to
chronic lung disease and hypoxia1,2. The pathophysiology of PH in CPFE is multifactorial, involving hypoxic
vasoconstriction, vascular remodeling and destruction of the pulmonary
capillary bed3. The
presence of PH in CPFE is associated with significantly worse outcomes,
including reduced exercise capacity and survival4. The management of CPFE remains
largely supportive, with no disease-specific therapies. Treatment strategies
include supplemental oxygen, pulmonary rehabilitation and management of
comorbidities. Lung transplantation is considered in advanced cases, although
many patients are ineligible due to age or comorbidities3,4.
While the use of pulmonary
vasodilators in WHO Group 3 PH is not routinely recommended due to concerns
about worsening ventilation-perfusion mismatch, emerging evidence suggests that
a subset of patients with severe PH and right ventricular (RV) dysfunction may
benefit from targeted therapy4,5.
Our patient had severe combined
pulmonary fibrosis and emphysema with relatively preserved spirometry and
decreased DLCO. He had evidence of pulmonary hypertension and tolerated Inhaled
Treprostinil that was initiated in the outpatient setting.
Our patient’s acute
decompensation was precipitated by a sub-segmental PE. Although often
considered clinically insignificant, sub-segmental emboli can be
hemodynamically destabilizing in patients with pre-existing PH and limited
cardiopulmonary reserve. The PE likely exacerbated RV dysfunction, contributing
to worsening hypoxia and increased oxygen requirements.
The evidence regarding the use of
systemic pulmonary vasodilators in Group III pulmonary hypertension is not
clear with conflicting findings. The guidelines recommend initiating inhaled
Treprostinil for Group III pulmonary hypertension associated with pulmonary
fibrosis, but not emphysema.
Inhaled Treprostinil, a
prostacyclin analog, has recently emerged as a promising therapy for Group 3
PH. The INCREASE trial demonstrated that inhaled Treprostinil significantly
improved 6-minute walk distance, reduced NT-proBNP levels and delayed clinical
worsening in patients with PH due to ILD, including CPFE6. Unlike systemic vasodilators,
inhaled agents offer the advantage of targeted pulmonary vasodilation with
minimal systemic effects, potentially reducing the risk of
ventilation-perfusion mismatch. Our patient was already on inhaled Treprostinil
at baseline and continued therapy during hospitalization and follow-up, likely
contributing to his clinical stability.
In patients with Group III
pulmonary hypertension, it is generally recommended to avoid endothelial
antagonists, however phosphodiesterase inhibitors may be considered in select
patients, however it is still unclear who would benefit the most3,4. It is likely that RV-PA (Right
Ventricle-Pulmonary artery) uncoupling may play a role to determine a response
to systemic pulmonary vasodilator therapy in patients with CPFE associated with
severe pulmonary hypertension.
RV-PA coupling describes the
relationship between RV contractility and pulmonary arterial afterload. It is a
critical determinant of RV efficiency and adaptation in PH. The gold standard
for assessing RV-PA coupling is the ratio of end-systolic elastance (Ees) to
arterial elastance (Ea), but surrogate markers such as TAPSE/SPAP and stroke
volume/end-systolic volume ratios are increasingly used in clinical practice.
Studies have shown that impaired RV-PA coupling is associated with worse
exercise capacity, clinical deterioration and mortality in PH patients7,8.
Despite anti-coagulation, his
oxygen requirements persisted beyond 72 hours. On the right heart
catheterization, the presence of a markedly elevated pulmonary vascular
resistance (10 WU), reduced cardiac index (1.49 L/min/m²) and a low TAPSE/SPAP
ratio (0.272) indicated poor RV-pulmonary artery (RV-PA) coupling, which
prompted us to consider Sildenafil therapy.
Sildenafil, a phosphodiesterase-5
inhibitor, is FDA-approved for WHO Group 1 pulmonary arterial hypertension
(PAH) and has demonstrated benefits in improving exercise capacity, pulmonary
hemodynamics and quality of life5. Although its use in Group 3 PH is off-label, small studies and case
reports have shown that sildenafil may improve oxygenation and RV function in
select patients with interstitial lung disease (ILD)-associated PH,
particularly those with evidence of RV-PA uncoupling9,10. In our case, the patient has
evidence of impaired RV-PA coupling and experienced a rapid and sustained
improvement in oxygenation following sildenafil initiation, supporting its
potential utility in this context. The temporal improvement strongly suggests
the role of sildenafil. Although, other possibilities remain that either the
complete effect of anticoagulation was delayed beyond 72 hours or the
possibility of improvement in right ventricular function.
Interestingly, sildenafil has
also been explored in the setting of acute pulmonary embolism (PE).
Experimental models and case reports suggest that sildenafil may reduce
pulmonary vascular resistance and improve RV function in acute PE by promoting
pulmonary vasodilation11,12. A randomized trial in patients with
intermediate-high risk PE found that a single dose of sildenafil did not
significantly improve cardiac index but did lower systemic blood pressure,
highlighting the need for further research13. In our patient, the combination
of sub-segmental PE and pre-existing PH likely precipitated acute RV
decompensation. The favorable response to sildenafil suggests that pulmonary
vasodilation may have mitigated RV afterload and improved oxygenation, even in
the acute setting, especially in the presence of impaired RV-PA coupling.
This case underscores the
importance of individualized therapy in CPFE with severe PH. While guidelines
caution against routine use of vasodilators in Group 3 PH, our patient’s
hemodynamic profile and clinical trajectory justified a trial of sildenafil, which
resulted in marked improvement. The TAPSE/SPAP ratio, a noninvasive marker of
RV-PA coupling, has been shown to predict outcomes in PH and guided our
therapeutic decision. The combination of inhaled Treprostinil and sildenafil
may offer synergistic benefits in select patients, although further studies are
needed to define optimal treatment strategies.
Conclusion
This case illustrates the
diagnostic and therapeutic complexity of managing CPFE with superimposed PE and
severe pulmonary hypertension. While pulmonary vasodilators are not routinely
recommended for WHO Group 3 PH, this case supports their use in select patients
with evidence of RV-PA uncoupling and hemodynamic compromise. Further research
is needed to identify which patients may benefit from such therapies.
References
1. Dias OM, Baldi BG, Costa AN, Carvalho CR. Combined
pulmonary fibrosis and emphysema: an increasingly recognized condition. J Bras
Pneumol 2014;40(3):304-312.
2. Jankowich
MD, Rounds S. Combined pulmonary fibrosis and emphysema syndrome: a review.
Chest 2012;141(1):222-231.
3. Cottin V, Le Pavec J, Prévot G, Mal H, Humbert M,
Simonneau G, Cordier JF; GERM"O"P. Pulmonary hypertension in patients
with combined pulmonary fibrosis and emphysema syndrome. Eur Respir J
2010;35(1):105-111.
4. Vitulo
P, Stanziola A, Confalonieri M, et al. Sildenafil in severe pulmonary hypertension associated
with chronic obstructive pulmonary disease: A randomized controlled multicenter
clinical trial. J Heart Lung Transplant 2017;36(2):166-174.
5. Humbert
M, Kovacs G, Hoeper MM, Badagliacca R. 2022 ESC/ERS Guidelines for the
diagnosis and treatment of pulmonary hypertension: Developed by the task force
for the diagnosis and treatment of pulmonary hypertension of the European
Society of Cardiology (ESC) and the European Respiratory Society (ERS).,
European Heart J 2022;43(38):3618-3731.
6. Waxman A, Restrepo-Jaramillo R, Thenappan T, et al.
Inhaled Treprostinil in Pulmonary Hypertension Due to Interstitial Lung
Disease. N Engl J Med 2021;384(4):325-334.
7. Guazzi
M, Bandera F, Pelissero G, et al. Tricuspid annular plane systolic excursion and pulmonary
arterial systolic pressure relationship in heart failure: an index of right
ventricular contractile function and prognosis. Am J Physiol Heart Circ Physiol
2013;305(9):1373-1381.
8. Trip P, Rain S, Handoko ML, et al. Clinical relevance
of right ventricular diastolic stiffness in pulmonary hypertension. Eur Respir
J 2015;45(6):1603-1612.
9. Ghofrani
HA, Wiedemann R, Rose F, et al. Sildenafil for treatment of lung fibrosis and pulmonary
hypertension: a randomised controlled trial. Lancet 2002;360(9337):895-900.
10. Han MK, Bach DS, Hagan PG, et al. IPFnet
Investigators. Sildenafil preserves exercise capacity in patients with
idiopathic pulmonary fibrosis and right-sided ventricular dysfunction. Chest
2013;143(6):1699-1708.
11. Wang H, Li W, Yan Y, et al. Sildenafil improves
hemodynamic changes caused by acute pulmonary embolism by inhibiting Rho kinase
activity. J Int Med Res 2024;52(4):3000605241240938.
12. Galea M, Quiney N.
Sildenafil in Acute Pulmonary Embolism: Case Report and Review of Literature.
Journal of the Intensive Care Society 2009;10(1):44-45.
13. Andersen A, Waziri F, Schultz JG, et al. Pulmonary
vasodilation by sildenafil in acute intermediate-high risk pulmonary embolism:
a randomized explorative trial. BMC Pulm Med 2021;21(1):72.