Treatment of Pyoderma Gangrenosum with Topical Cromoglycate: A Forgotten Treatment Option? Presentation of Own Case and Review of Literature
Abstract
A challenging case of a patient with pyoderma gangrenosum dating back to 1990s is presented. After multiple and prolonged treatment attempts for 1.5 years, extensive ulcers started to heal rather fast within weeks with topical 2% cromolycate solution, which is more commonly used as an eye allergy drug. By the literature, the use of topical cromolycate has not been published for treatment of pyoderma gangrenosum after 2000.
Keywords: pyoderma gangrenosum; cromoglycate; treatment
Introduction
Pyoderma
gangrenosum (pg) is a rare ulcerative neutrophilic dermatosis with features of
vasculitis-like appearance. The etiology is unknown and histology not specific
for pg. The pathogenesis is complex, and dysregulation of innate and adaptive
immunity are involved1. The incidence is thought to be about 6,3
per 1,000,000 with the median age at presentation of 59 years. The sex
incidence is from equal, to females being predominant up to 76 % of cases2. Pg is often
associated with a systemic disease, such as ulcerative colitis, crohn's
disease, chronic active hepatitis, rheumatoid arthritis, monoclonal
gammopathy/myeloma, hematological malignancies, sarcoidosis, or a malignant or
other proliferative disease3, and syndromes such as papa, pash, papash
and sapho1. Infections caused by streptococci,
staphylococci or gram-negative bacteria have also been suspected as the
causative agents.
The
treatment of pyoderma gangrenosum is aimed at the established underlying
disease. Often, however, it cannot be diagnosed, and then the treatment is
aimed at soothing the wound process with different means. For example,
bacterial pyoderma, deep fungal infections, syphilitic gumma, necrotizing
vasculitis and treatment-related ulcerations must be ruled out4. The clinical
pictures of bacterial pyoderma and necrotizing vasculitis overlap with pyoderma
gangrenosum.
Treatments
Since the effectiveness of local treatments is usually
insufficient, systemic corticosteroid medication at doses of 40–120 mg/day is
the first-line treatment4,5. Intravenous pulse therapy (methylprednisolone 1,000
mg/day for 1–5 days6 has been given to reduce the side effects of
steroids. Other treatments have included plasmapheresis, tetracyclines,
vancomycin, metzocillin, dapsone, salazosulfapyridine, azathioprine, alkylating
agents (cyclophosphamide, melphalan, chlorambucil, clofazimine), which have had
varying degrees of efficacy4,5,7. Cyclosporine4,8,9 and tacrolimus (a
macrolide antibiotic,
which has an immunosuppressive effect similar to cyclosporine) have been shown
in a small data set4. Individual patients have been treated with
thalidomide and hyperbaric oxygen therapy4.
Gm-csf (granulocyte macrophage-colony stimulating factor) has been reported to
be beneficial10.
A
recent review with proposed algorithm for treatment of pg has been presented
for treatment of pg nowadays: systemic
corticosteroids, cyclosporine, methotrexate, mycophenolate mofetil,
azathioprine, systemic tacrolimus, dapsone, colchicine, thalidomide,
i.v.-immunoglobulin, granulocyte-macrophage adsorption apheresis, and the most
recent biologics such as inhibitors for tnf-alfa, il-1-beta, il-1alfa, il-17, il-23, c5a, il-6 , cd3, cd20, integrin, pde4, and jaks1.
Topical treatments
Local
topical treatments are aimed at alleviating pain and preventing secondary
infection. In mild forms of the disease, the effect of topical antimicrobial
treatments may be sufficient when they are continued with a topical or
intradermal corticosteroid (triamcinolone) or topical 5-aminosalicylic acid.
Individual cases have also been described treated with systemic cyclosporine
and topical mechlorethamine (topical nitrogen mustard)4,5.
A
recent description of topical treatments is presented for pg in a review:
corticosteroids, calcineurin inhibitors, miscellaneous basic wound care
treatments, topical timolol and phenytoin1.
Topical cromoglycate
A
total of 17 patients treated with cromoglycate have been described in the
literature from 1980 until 1998, and treatment response was achieved in 15
cases11-17. Of the two patients who did not respond to treatment,
one had monoclonal gammopathy18 and the other had recurrent idiopathic pg
for 14 years, in which cyclosporine was effective19. In the study of
five patients, only one was given topical cromoglycate as the only treatment;
four received oral steroids (prednisone 60 mg/day) at the same time and two of
them also received 5-aminosalicylate (2g/day)16. The response to 4% cromoglycate appeared
in 3 days and the wounds healed in about 3 months17.
Local
cromoglycate under occlusion with clobetasol dipropionate lead to only partial
healing but adding oral cyclosporin and triamcinolone injections led to
progressive complete healing after 7 months20.
Thus, the contribution of topical cromoglycate to overall healing cannot
be determined.
A
wide range of oral and topical treatments (including corticosteroids and
cromoglycate) were shown ineffective in the treatment of pg of a 68-year-old
woman but oral mycophenolate mofetil in combination with oral cyclosporine
showed an effect with thrombocytic growth factors followed 8 weeks later by
split thickness skin grafts21.
A
study described a series of 7 patients with peristomal pg. A 72-year-old female
with crohn’s disease got an effect by use of topical clobetasol propionate and
cromoglycate with intravenous infliximab. However, cromoglycate was ineffective
for a 64-year-old male with bladder cancer22.
Cromoglycate
can be used only locally, because the absorption of cromoglycate through the
mucous membrane of the gastrointestinal tract is poor; in rat and rabbit
experiments, absorption has been found to be only about 0.1–2.5%23. Absorption of 4%
cromoglycate emulsion cream through the skin is also very low at about
0.01–2.75%24. The mechanism of action of cromoglycate
has been shown to be based on the stabilization of the cell membrane of the
mast cell by indirectly inhibiting the function of calcium channels, as a
result of which the release of neurotransmitters and the inflammatory reaction
of the mast cells are inhibited25. The mechanism of action of cromolycate in
pyoderma gangrenosum is unclear. However, in several studies, cromoglycate has
been found to have direct effects on neutrophils and other inflammatory cells
at very low concentrations, even at 10 nm25-27.
Case presentation
The
patient was a 64-year-old woman who developed difficult-to-treat ulcers on her
lower legs in the spring of 1993. For years, she had hypercholesterolemia,
supraventricular arrhythmias, hypertension and coronary artery disease, and
left ventricular hypertrophy. In 1984, due to aortic enlargement and aortic
valve leakage, she underwent reconstruction and an aortic valve prosthesis
installation, and coronary artery bypass surgery was performed at the same
time. In 1985, a diagnosis of polymyalgia rheumatica was made. The treatment
was oral steroid medication, which had a quick response and the sr returned to
normal. The patient had been diagnosed with mild kidney failure related to
polycystic kidneys, and she also had many cysts in the liver. The medication was
verapamil, indapamide, lovastatin, potassium chloride, warfarin and
prednisolone (5 mg/day).
The
patient came to the dermatology clinic for examinations and treatments in
october 1993. On admission, several 1–3 cm-sized infected ulcers in the leg
area were found, with vasculitis-like redness at the edges, and purple, livedo
reticularis and darker blue-red macular patches around the wounds. Since then,
these ulcerated, multiplied and began to expand and merge into larger wounds (figure 1).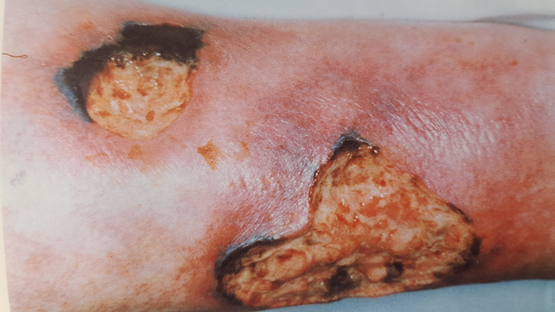
Figure 1.
After 6 months of disease (left leg lateral aspect).
Extensive
investigations did not reveal a clear underlying immunological disorder. The
pathological-anatomical diagnosis of the wound edge specimen was ulceration.
The reddened livedo reticularis area showed a microscopic examination of
chronic dermatitis; no specific findings were found in the immunofluorescence
study. Complete blood count, sr, crp, thyrotropin, ige, ast, creatine kinase,
aldolase, complement c3 and c4, and circulating immune complexes were
determined repeatedly with normal results. The result of the waaler-rose test
was also repeatedly normal. Cryoglobulins, hepatitis b and c antibodies, herpes
simplex, borrelia, nuclear and anc antibodies were not detected. Serum
creatinine concentration was 150–160 µmol/l. Fungal
cultures from test pieces at the edge of leg wounds were negative.
Electrophoretic fractionation of serum proteins did not reveal paraproteinemia,
but hypoalbuminemia and hypogammaglobulinemia were found, which was consistent
with renal failure and proteinuria of renal origin. X-rays of the lungs and
computed tomography of the abdomen showed no signs of cancer. No indications of
hematological abnormalities were found in the bone marrow aspiration sample.
From
the entry stage, staphylococcus aureus, proteus mirabilis, enterococcus
faecalis, acinetobacter and candida were detected in the wounds. Later, in
bacterial cultures, various bacteria were also found, such as citrobacter
freundii, xanthomonas maltophilia, klebsiella pneumoniae, enterobacter cloacae,
e. Coli, usually with combinations of 2–3 different bacteria and candida.
The
patient had osteoporosis and a mild moon-like face, and therefore prednisolone
in small doses (15 mg/day) and azathioprine (100 mg/day) were started to treat
the wounds. Cephalexin was chosen as the antibiotic and intrasite-gel and
iodosorb-cream were used as local treatment. Despite these treatments, however,
the condition progressed; the sizes of the wounds increased, the new ulcers had
a vasculitis-like feature, and the edges of the wounds had a bluish tint. The
prednisolone dose was increased to 40 mg/day and the azathioprine dose to 150
mg/day, and cephalexin, sulfadiazine-trimethoprim, bacampicillin and
ciprofloxacin were used as antibiotics according to bacterial culture results.
In addition, gm-csf was tried as a local treatment for a short time. Potassium
permanganate baths twice a week and 0.1 % silver nitrate baths twice a day were
given as drying and antimicrobial local treatment.
As
additional etiology exclusion, temporarily, warfarin was also changed to
phenindione, but later it was returned back to warfarin because the change had
no effect on the wounds; warfarin-induced ulcers were ruled out with this drug
change.
Due
to the lack of treatment response, azathioprine was replaced by
cyclophosphamide at doses of 100 mg/day and the prednisolone dose was kept at
35 mg/day. Due to osteoporosis, calcium supplements (1,000 mg/day) and
calcitonin nasal sprays were started. After a week of using cyclophosphamide, a
significant leukopenia (1.6 x10exp9/l) occurred, and this medication was
stopped. In the studies of increased back pain, osteoporotic collapse fractures
of the vertebral bodies of the spine at levels l3-l4 were found. After this,
plasmapheresis was performed on 5 consecutive days, but it had no clear effect
on pg. Next, cyclosporine medication was started at doses of 3 mg/kg/day, but
the dose was reduced by half about a month later due to an increase in the
serum creatinine concentration to 350 µmol/l, and increase in blood pressure
and swelling, which were caused by hypokalemia and hypomagnesemia from the
furosemide used. The steroid dose was gradually reduced over the course of
months to 15 mg/day, when the redness at the edges of the wounds had decreased
and the increase in size had stopped. After a stable period, the situation
began to deteriorate rapidly less than 3 months after the start of cyclosporine
medication. This medication was discontinued and replaced with dapsone at doses
of 100 mg/day, but after a week of use the dose was reduced by 50 % due to an
increase in the methemoglobin value. The prednisolone dose was further slowly
reduced to 7.5 mg/day. The situation remained somewhat stable for a few weeks,
but then the wounds started to get worse again quickly. The steroid dose was
not increased due to concurrent herpes zoster infection (figure 2).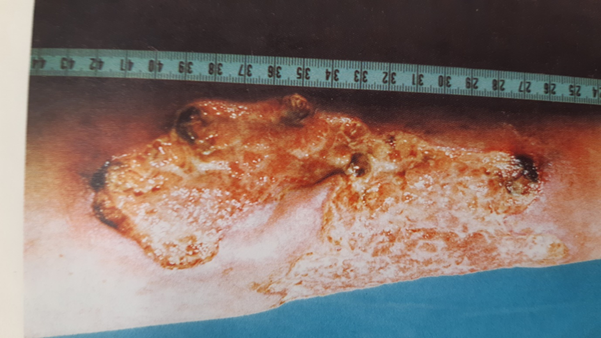
Figure 2. At 1.5 years of disease and at the time
topical treatment with cromoglycate was initiated.
At
this stage, in october 1994, local treatment with 2% cromoglycate (lecrolyn,
single-dose pipettes without preservatives) was started. The medicine was given
once a day (a pipette per wound area of about 4 cm) and at the same time the
wound areas were covered with duoderm sheets. In this case, the dose of dapsone
had been 50 mg/day for about 1.5 months, and this medication was continued at
the same time as the cromolycate treatment with prednisolone dose at 7.5
mg/day. The pain in the lower legs had been constant, and because of them,
ketoprofen (200 mg/day) and dextropropoxyphene (130 mg/day) had been given, and
later buprenorphine (0.4-0.8 mg/day) had been given instead. The pain caused by
the wounds that had been growing for months was clearly alleviated and the
redness reduced in 2-3 days, and then the wounds started to shrink in 3.5 weeks
noticeably after growing for about 1.5 years (figure 3). At the same time, the dose of dapsone was quickly
reduced to 100 mg/week and the use of buprenorphine for painkillers was
stopped. The use of ketoprofen also decreased. After rapid initial progress,
wound reduction slowed, and eventually final wound closure after approximately
14 months, and dapsone was stopped and prednisolone dose was further reduced to
5 mg/day.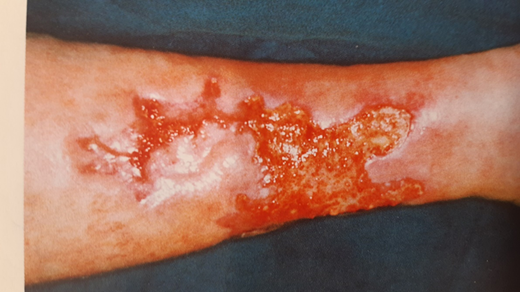
Figure 3. After 3.5 weeks with topical cromoglycate
treatment.
The
patient used continuous oral antibiotic therapy for more than two years. In the
follow-up at 2 years, the wounds have remained closed (figure 4).
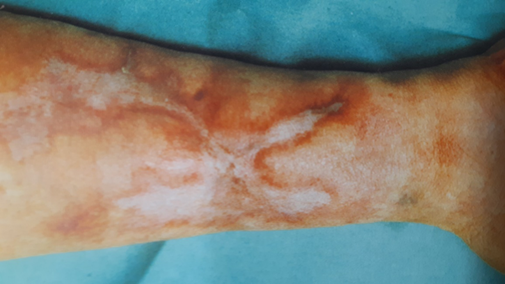
Figure 4. After 2-year follow up, wounds healed
fully 10 months earlier. The patient’s legs were saved from amputation.
Discussion
The patient
developed difficult-to-treat painful and constantly growing ulcers in the lower
legs. Based on the examinations and the clinical picture, the diagnosis was pg.
After several drug treatment trials that were ineffective or failed due to side
effects, we decided to proceed on local cromolycate treatment, with which a
clear response was achieved in just 2-3 days. This treatment was started in a
situation where treatments known to be effective were no longer available, and
thus the patient, who had been active until then, was threatened with
amputation of both lower limbs.
The literature
describes a response to cromolycate in 3 days and a wound healing in 5 weeks in
a patient who apparently had hepatitis c-based liver cirrhosis and portal
hypertension. However, the ulcers of the patient in question were clearly more
superficial16 than in our own patient. Other patients in the study16 also received high doses of prednisone (110 mg/day) and
two additional doses of 5-aminosalicylic acid (2 g/day). In these patients,
wound healing times were 5-8 weeks. In another patient case, a response was
also obtained in 3 days, and the final healing of the ulcers took about 3
months17.
The ulcers on our
patient's legs were very painful, deep, and extensive. Based on the discussion
and consideration with her, a conservative treatment line was chosen. It took a
little over a year to achieve a complete healing. High-dose steroid treatment
would obviously have been beneficial, but soon after the prednisolone dose was
increased to 40 mg/day, the patient developed osteoporotic collapse fractures
of the spinal vertebrae, which calcitonin and calcium supplementation could not
prevent. Giant-dose steroid pulse therapy may be associated with electrolyte
disturbances, which this patient already had due to treatment for kidney
disease and associated hypertension. At a dose tolerated by the patient,
cyclosporine remained ineffective, and renal toxicity began to emerge at a dose
of 3 mg/kg/day, at which level the efficacy of cyclosporine is insufficient9. Cyclophosphamide treatment caused severe leukopenia
after only one week and it had to be discontinued. There was no response to
plasmapheresis treatment. Dapsone at a dose of 100 mg/day was associated with
methemoglobinemia, which was corrected with a dose of 50 mg/day.
Dapsone had been
used for about 1.5 months when the condition of the wounds worsened, and local
cromolycate treatment was then started. This resulted in a very quick response
within a few days, which we attribute primarily to the effect of cromolycate,
although a synergistic effect with dapsone and prednisolon is also possible. At
that time, the dose of prednisolone was small (7.5 mg/day), but due to the
simultaneous herpes zoster infection, the steroid dose was not increased.
Prednisolone
inhibits the responses of both antibody and cell-mediated immunity and, in
large doses, can cause the infection to spread. The results from use of steroid
to prevent post-shingles pain have been variable, and according to the textbook
of dermatology28, one study had a favorable response in immunologically
normal herpes zoster patients. Instead, in pg, the cause has been considered a
process directly affecting the immunological system, the nature of which is
admittedly not known in more details. Thus, in the case of this patient, it was
decided to remain on low-dose steroid therapy for the duration of the zoster
infection. The importance of herpes zoster in the exacerbation of pg is not
known.
For local
treatment, cromolycate was given once a day until the wounds healed. Treatment
given 4 times a day might have been more effective17, but there were no practical possibilities for this. The
concentrations of topically used cromolycate described in the literature have
been 1-4%. Cromoglycate was used until the wounds closed. The etiology of pg is
unknown, although underlying diseases often affect the immune system. A
bacterial etiology has been suggested. Of the bacteria grown in wounds,
especially staphylococcus aureus and enterococcus are quite pathogenic, proteus
and klebsiella are less pathogenic. The patient was given antibiotic treatment
for a total of 2 years. The purpose of this was to prevent the worsening of the
situation caused by secondary bacterial infection, and, as prevention of
erysipelas, as cromolycate has no antimicrobial effect.
Numerous different
local and systemic treatments have been presented in the literature, and the
response to them has been varied. Thus, this skin disease may have several
etiological factors and mechanisms. The use of cromoglycate has been minimal,
but in the described cases – including ours - a positive response has been
found in 16 out of 18 patients.
Later, there was
not found the use of cromoglycate in pg-wounds by search from pubmed after
2000. In this context, it should be
emphasized that there have likely been a few treated patients, and it is
possible that only the cases with a positive result will be published. Also,
even positive outcomes may not have been presented.
In our clinic since
1999, only a few pg patients have been treated by topical cromoglycate combined
with various oral and topical treatments with variable outcome, and the role a
cromoglycate cannot be solely determined unlike in this our patient for whom practically
all possible treatments were given at the time in the 1990s, so we considered
the trial of cromoglycate treatment, which gave a clear fast response, to be
ethically acceptable, even though this form of treatment was not mentioned as a
treatment option for this disease at the time and not in the recent reviews1,2.
The later treatment
options by biologics during the last decades targeted on various interleukins that
are more
often used in the treatment of psoriasis might have also a good effect1,2. Cromoglycate
is cheap and has a few side effects and can be combined with systemic or
topical treatments. This option should be considered already in the early
stages of pg treatment, alone or combined with other treatments.
The treatment of
our patient was very challenging with the treatment options available in the
1990s, with drawbacks and side effects of used medications. In the end, the
wounds healed and have remained closed during the follow-up period of 2 years.
The patient had felt well and moved actively on her own feet.
Conflicts
of interest: author
declares none.
References
4. Chow rk, ho vc. Treatment of pyoderma gangrenosum. J am acad dermatol
1996;34:1047-1060.
7. Berger tg, elias pm, wintroub bu. Pyoderma gangrenosum. In: manual of
therapy for skin diseases. New york: churchill-livingstone 1990;256-258.
26. Patalano f, ruggieri f. Sodium cromoglycate: a review. Eur respir j 1989;556s-560s.
28. Sterling jc, kurtz jb. Varicella zoster. In: champion rh, burton jl,
burns da, breathnach sm, toim. Rook/wilkinson/ebling. Textbook of dermatology 6th
edition. Oxford: blackwell 1998;1015-1022.