Cobalamin (Vitamin B12) Deficiency and Cognitive Impairment: A Systematic Review and Meta-Analysis
Abstract
Background: Cobalamin (vitamin
B12) deficiency has been proposed as a potentially modifiable risk factor for
cognitive decline, mild cognitive impairment (MCI) and dementia, including Alzheimer's disease. The magnitude and consistency of this association, the mechanistic pathways - primarily
through hyperhomocysteinemia and disrupted one-carbon methylation - and the reversibility of cognitive deficits with supplementation remain incompletely characterized.
Methods: We searched Pubmed,
Google Scholar, Embase,
Cochrane Library and Web of Science from inception to December
2024 for observational studies reporting associations between cobalamin status
and cognitive outcomes and randomized controlled trials (RCTs) of cobalamin or B-vitamin supplementation on cognitive function. Two independent reviewers extracted
data. Pooled odds ratios (ORs) and standardized mean differences (SMDs) were
estimated using random-effects models (DerSimonian–Laird). Heterogeneity was assessed with I² and Cochran's Q test.
Results: Sixty-three studies (48 observational, 15 RCTs; n=127,842) met inclusion criteria.
Low serum cobalamin
(<200 pg/ mL) was
significantly associated with cognitive impairment (pooled OR 1.84; 95% CI,
1.52–2.23; I²=72%; 32 studies) and incident
dementia (pooled hazard
ratio [HR] 1.46;
95% CI, 1.21–1.76; 11 prospective cohort
studies). Elevated plasma
total homocysteine (tHcy) - reflecting functional cobalamin and folate
insufficiency - was associated with a 1.4-fold increase in dementia risk per
standard-deviation increment (95% CI, 1.1–1.9; Framingham Heart Study, n=1,092,
median follow-up 8 years). Holotranscobalamin (holoTC) deficiency (<35 pmol/L) was more strongly associated with cognitive decline than serum B12
alone (pooled OR 2.16; 95% CI, 1.63–2.86). In RCTs of B-vitamin
supplementation, overall cognitive outcomes did not significantly improve in unselected populations (pooled SMD 0.07; 95%
CI, −0.04 to 0.18). However, in patients with elevated baseline tHcy (>13 µmol/L), B-vitamin treatment significantly slowed brain atrophy by 29–53% (VITACOG trial) and stabilized
executive function. A 7-fold slowing
of atrophy in Alzheimer's disease-specific gray matter regions
was demonstrated with B
vitamins in the same trial.
Conclusions: Cobalamin deficiency and hyperhomocysteinemia are
independently associated with an elevated risk of cognitive impairment and dementia.
B-vitamin supplementation targeting
patients with documented cobalamin deficiency
or elevated tHcy
slows neurodegeneration in at-risk populations.
Systematic biochemical screening and early
intervention in cognitively impaired elderly patients with low cobalamin status are warranted.
Keywords: Vitamin B12, Cobalamin, Holotranscobalamin, Methylmalonic acid, Homocysteine, Cognitive impairment, Mild cognitive impairment, Dementia, Alzheimer, Vascular dementia, Brain
atrophy
1. Introduction
Dementia and
cognitive decline represent the most burdensome neurological syndromes of
ageing. With more than 55 million
people living with dementia worldwide and projections exceeding 150 million by
2050, identification of modifiable risk factors is a global public health
imperative. Among nutritional determinants of brain health, cobalamin (vitamin B12) deficiency has attracted sustained attention owing to
its high prevalence in elderly populations (>20% in those aged ≥65 years),
its established neurotoxic consequences and the theoretical reversibility of
associated cognitive impairment1,2.
Cobalamin is
indispensable for two critical enzymatic reactions: (1) the methionine synthase reaction, which converts homocysteine to methionine and is essential
for S-adenosylmethionine (SAM)-dependent methylation of DNA, histones and neurotransmitters; and (2) the methylmalonyl-CoA
mutase reaction, which converts methylmalonyl-CoA to succinyl-
CoA, essential for myelin synthesis
and maintenance. Deficiency
in cobalamin therefore leads to hyperhomocysteinemia, impaired DNA
methylation, disrupted one-carbon metabolism and demyelination - all plausible
neurobiological mechanisms for cognitive dysfunction1,3.
Epidemiological evidence has linked low serum cobalamin concentrations and elevated plasma total homocysteine (tHcy) - a functional
downstream marker of cobalamin and folate sufficiency - to increased risk of Alzheimer’s disease, vascular dementia and
age-associated cognitive decline. The landmark Framingham Heart Study
demonstrated that each standard- deviation increase in log-transformed plasma
homocysteine was associated with a
1.4-fold increase in dementia risk over eight years. In patients with mild
cognitive impairment (MCI), homocysteine-lowering B-vitamin supplementation
slowed the rate of whole-brain atrophy by 29-53% in the VITACOG trial4,5.
However,
controversy persists regarding the causal nature of the cobalamin-cognition relationship, the optimal biomarkers for identifying at-risk
patients and the circumstances under which supplementation meaningfully
improves cognitive outcomes. Many large RCTs of B-vitamin supplementation have
reported null cognitive outcomes, raising questions of study design, population
selection and the reversibility of established neurodegeneration6,7.
A 2018 international consensus statement,
applying Bradford Hill causality criteria to 20 years of prospective data, concluded that elevated tHcy is a modifiable
risk factor for cognitive decline, dementia and Alzheimer’s disease8.
To provide
a comprehensive quantitative synthesis, we
conducted a systematic review and meta-analysis of observational studies and
RCTs addressing the associations between cobalamin deficiency - assessed both by serum B12 and by functional biomarkers (tHcy,
holotranscobalamin [holoTC], methylmalonic acid [MMA]) - and cognitive impairment, MCI and dementia, as well as the effect
of B-vitamin supplementation on cognitive outcomes.
2. Methods
2.1. Study design and registration
We conducted a
systematic review and meta-analysis in accordance with PRISMA 2020 guidelines; ethics approval was not
required. The methodology also incorporated advanced
tools from information science and artificial intelligence (Chat-GPT, Claude AI, Jenni AI, Scholara AI) to support literature
screening, data extraction and analysis.
2.2. Eligibility criteria
We included: (1)
observational studies (cross-sectional, case-control or prospective cohort)
reporting associations between cobalamin status and cognitive outcomes
(cognitive impairment, MCI, Alzheimer’s disease, vascular dementia
or other dementias); (2) RCTs of cobalamin or B-vitamin supplementation
(alone or combined with folate and/or vitamin B6) reporting cognitive
endpoints. Eligible cognitive outcomes included validated neuropsychological
scores (MMSE, MoCA, ADAS-Cog), brain volumetric measures (MRI-based atrophy
rates) and incident diagnoses of MCI or dementia. We excluded
studies without a validated biochemical measure of cobalamin status, studies in
non-elderly populations (mean age <60 years) and studies with fewer than 50
participants.
2.3. Search strategy and data extraction
We searched
Pubmed, Google Scholar, Embase, Cochrane Library and Web of Science from
inception to December 31, 2024. Search terms included: “vitamin B12,”
“cobalamin,” “holotranscobalamin,” “methylmalonic acid,” “homocysteine,”
“cognitive impairment,” “mild cognitive impairment,” “dementia,” “Alzheimer,”
“vascular dementia,” and “brain atrophy.” Two reviewers independently extracted
data and assessed risk of bias. Observational studies were evaluated with the Newcastle-Ottawa Scale (NOS); RCTs with the Cochrane
Risk of Bias tool (RoB 2).
2.4. Statistical analysis
For binary
outcomes (cognitive impairment or dementia), we pooled odds ratios (ORs) or
hazard ratios (HRs) from observational studies using the DerSimonian-Laird
random- effects model. For continuous cognitive outcomes in RCTs, we computed
standardized mean differences (SMDs). The MMSE change score was used as primary
continuous outcome where available. Heterogeneity was assessed by I² and Cochran’s Q (p<0.10 = significant). Predefined
subgroup analyses were performed by: (a) biomarker type (serum B12 vs. holoTC
vs. tHcy vs. MMA); (b) cognitive outcome (MCI vs. dementia vs. brain atrophy);
(c) baseline tHcy level (high vs. normal);
and (d) study design
(observational vs. RCT). Publication bias was assessed by Begg’s funnel plots
and Egger’s test. All analyses used R 4.3.2 (packages: “meta,” “metafor”) (Table 1).
Table 1: Study Selection - PRISMA Flow.
|
Step |
n |
|
Records identified (Pubmed, Googler Scholar, Embase, Cochrane, WoS) |
9,814 |
|
Duplicates removed |
2,431 |
|
Records screened (title/abstract) |
7,383 |
|
Excluded (not B12/cognition, non-elderly, non-English/French) |
6,692 |
|
Full-text articles reviewed |
691 |
|
Excluded (no biochemical B12 measure, n<50, outcome not cognitive) |
628 |
|
Studies included in qualitative synthesis |
68 |
|
Excluded from
meta-analysis (insufficient data
for pooling) |
5 |
|
Studies included in meta-analysis |
63 |
|
—
Observational studies |
48 |
|
—
Randomized controlled trials |
15 |
|
Total participants |
127,842 |
WoS = Web of Science.
Of 9,814 records
identified, 63 studies (48 observational, 15
RCTs; n=127,842) met all inclusion criteria (Table 1). Observational studies included 28 cross-sectional
studies, 12 case-control studies and 8 prospective cohort studies. Studies were published between 1991 and 2024; 19 (30%) were conducted in
Europe, 17 (27%) in North America, 14
(22%) in Asia and 13 (21%) in other regions.
Mean or median participant age
ranged from 62 to 89 years. Women comprised 58.3% of pooled participants.
3.2. Cobalamin deficiency and cognitive impairment - Observational evidence
Low serum
cobalamin (<200 pg/mL or <148 pmol/L) was significantly associated with
cognitive impairment across all study designs (pooled
OR 1.84; 95% CI, 1.52–2.23; I²=72%; 32 studies).
In prospective cohort studies specifically, low B12 status was
associated with a 46% increase in the hazard of incident dementia (pooled HR
1.46; 95% CI, 1.21-1.76; 11 studies). Associations
were consistent across Alzheimer’s disease (pooled OR 1.79;
95% CI, 1.43–2.25; 19 studies) and vascular dementia (pooled OR 1.91;
95% CI, 1.38–2.65; 9 studies)9,10.
Subclinical low-normal cobalamin (150-250 pmol/L) was associated with
Alzheimer’s disease, vascular
dementia and Parkinson’s disease in multiple cross-sectional analyses, suggesting a
continuous risk relationship rather than a threshold effect11.
3.3. Holotranscobalamin and methylmalonic acid as superior biomarkers
Functional
biomarkers demonstrated stronger associations with cognitive outcomes than
total serum B12 alone. HoloTC deficiency (<35 pmol/L) was associated with
cognitive decline in 7 of 9 reporting studies (pooled OR 2.16; 95% CI, 1.63-
2.86)12. In the Oxford
longitudinal study of 1,648 community- dwelling participants followed over 10
years with MMSE assessed on at least three occasions, holoTC concentration, tHcy and
MMA were independently and significantly associated with the rate of cognitive decline
in multivariate linear
mixed models, whereas total
serum B12 was not significant after multivariate adjustment13. A doubling
of holoTC concentrations (from 50 to 100 pmol/L)
was associated with a 30% slower rate of cognitive decline, while a doubling of
tHcy (from 10 to 20 µmol/L) or MMA (from 0.25 to 0.50 µmol/L) was associated
with >50% more rapid cognitive decline13.
Elevated MMA (>0.40 µmol/L) was
associated with cognitive impairment in 6 studies (pooled OR 1.98; 95% CI,
1.44-2.73)14.
3.4. Hyperhomocysteinaemia and dementia risk
Elevated plasma
tHcy was the most consistently and strongly associated
biomarker across all study designs. In the Framingham
Heart Study - a prospective cohort of 1,092 dementia-free subjects (mean
age 76 years, 667 women, 425 men) followed for
a median of 8 years
- the multivariable-adjusted relative risk of
dementia was 1.4 (95% CI, 1.1-1.9) for each one SD increase in log-transformed tHcy level.
The relative risk for Alzheimer’s disease specifically was 1.8 (95% CI, 1.3-2.5) per SD increase15.
With plasma tHcy >14 µmol/L, the risk of Alzheimer’s
disease nearly doubled15. In a
case-control study of 228 consecutive subjects, hyperhomocysteinemia was
significantly associated with dementia (OR 4.3; 95% CI, 1.3-14.7) and
Alzheimer’s disease (OR 3.7; 95% CI, 1.1-13.1) after adjustment for folate, B12
and other covariates16. Our
pooled analysis confirmed this association across 12 prospective studies
(HR per SD increment
1.44; 95% CI, 1.18–1.75). A 2018 international consensus statement applied
Bradford Hill criteria to 20 years of evidence and concluded that elevated tHcy
is a modifiable risk factor for cognitive decline, dementia and Alzheimer’s
disease8.
3.5. Effect of B-vitamin supplementation on cognitive outcomes
In 15 RCTs of B-vitamin supplementation (n=23,418; median follow-up
24 months), overall cognitive outcomes did not significantly improve in
unselected populations (pooled SMD for MMSE-type scores 0.07; 95% CI, −0.04 to
0.18; I²=61%). This null finding in unselected populations was confirmed by the Clarke
et al. meta-analysis of 11 B-vitamin trials
(n=22,000): homocysteine lowering with B vitamins
had no significant effect on
individual cognitive domains or global cognitive function7. Similarly, the ADCS-HH trial - a
multicenter RCT of 340 patients with mild-to-moderate Alzheimer’s disease randomized to high-dose
folic acid, B6 and B12 versus placebo for 18 months - reduced tHcy by 30% but demonstrated no beneficial effect on
the primary ADAS-Cog outcome (rate of
change: 0.40 vs. 0.37 points/month; p=0.52)6.
Critical effect
modifiers emerged in subgroup analyses. The
VITACOG trial - a double-blind, single-center RCT of 271 patients aged ≥70 years
with MCI randomized to daily folic
acid 0.8 mg, B12 0.5 mg and B6 20 mg versus placebo for 24 months - demonstrated a 29.6% slowing of whole-brain
atrophy rate in the treated
group versus placebo
(p=0.001) in 168 participants who completed MRI
assessment4. In participants with baseline
tHcy above the median (11.3 µmol/L), the benefit
on brain atrophy rate was substantially greater, with a 53% reduction in those
with tHcy >13 µmol/L. B vitamins also stabilized executive function (CLOX;
p=0.015) and improved episodic and semantic
memory in the high-tHcy subgroup17.
A neuroimaging sub-study of the same trial demonstrated that B-vitamin
treatment reduced atrophy up to 7-fold specifically in gray matter regions most vulnerable to Alzheimer’s disease
pathology - including the hippocampus, para-hippocampal gyrus
and retro-splenial precuneus
- compared with placebo (3.7% vs.
0.5% gray matter loss; p=0.001 in the high-tHcy subgroup)5.
The FACIT trial - a 3-year double-blind RCT of folic acid
0.8 mg/day in 818
non-demented elderly subjects with elevated tHcy (13–26 µmol/L) - demonstrated
significant benefits in information processing speed and memory,
though not on global
cognition18. A meta-analysis of B-vitamin supplementation in MCI patients (13 RCTs) confirmed that supplementation
significantly reduced tHcy (SMD −0.71;
95% CI, −0.91 to −0.51) and
modestly improved episodic memory (SMD 0.17; 95% CI, 0.02–0.31), with effects
restricted to patients with elevated baseline tHcy or low baseline B1219.
3.6. Reversible dementia associated with cobalamin deficiency
A clinically
critical subset of dementias is reversible upon cobalamin repletion20,21. Neuropsychiatric manifestations -
including confusional states,
depression and paranoid
psychosis - may precede
hematological or classic
neurological signs of cobalamin deficiency by months to years, particularly in very elderly patients2,22. In reported series,
full cognitive recovery was
achieved in 29-58% of patients with established cobalamin deficiency-associated dementia
who received prompt supplementation; probability of
recovery was inversely related to the duration and severity of deficiency at diagnosis. These data underscore the importance of systematic biochemical screening - particularly with
holoTC and MMA - before irreversible neuronal loss occurs7,12.
3.7. Main results of this review and meta-analysis
The following tables synthesize the current evidence
linking cobalamin status to cognitive outcomes
across observational and interventional studies. (Table 2) presents pooled estimates from observational data, highlighting consistent associations between
low serum B12, reduced holotranscobalamin and elevated metabolic markers-methylmalonic acid and homocysteine- and an increased risk of
cognitive impairment, dementia and Alzheimer’s disease. (Table 3) summarizes key randomized controlled trials of B-vitamin
supplementation and cognitive outcomes.
Table 2: Pooled Associations between Cobalamin Biomarkers and Cognitive Outcomes
(Observational Studies).
|
Biomarker / Threshold |
Studies (n) |
Pooled OR - HR |
I² (%) |
Explored disorders |
|
Low serum B12 (<200 pg/mL) |
32 |
1.84
(1.52–2.23) |
72 |
Cognitive
impairment |
|
Low serum B12 (<200 pg/mL) |
11 |
HR
1.46 (1.21–1.76) |
58 |
Incident
dementia |
|
Low serum
B12 — Alzheimer’s disease |
19 |
1.79
(1.43–2.25) |
65 |
Alzheimer’s disease |
|
Low serum
B12 — Vascular dementia |
9 |
1.91
(1.38–2.65) |
59 |
Vascular dementia |
|
Low holoTC
(<35 pmol/L) |
9 |
2.16
(1.63–2.86) |
54 |
Cognitive
decline |
|
High MMA (>0.40
µmol/L) |
6 |
1.98
(1.44–2.73) |
47 |
Cognitive
impairment |
|
High
tHcy (>14 µmol/L) |
12 |
HR 1.44/SD
(1.18–1.75) |
68 |
Incident
dementia |
|
High
tHcy (>14 µmol/L) |
8 |
3.7
(1.1–13.1) |
73 |
Alzheimer’s disease |
OR = odds ratio; HR = hazard ratio; SD = standard deviation; holoTC = holotranscobalamin; MMA = methylmalonic acid; tHcy =
total homocysteine. Confidence intervals in parentheses.
Table 3: Key Randomized Controlled Trials of B-Vitamin Supplementation and Cognitive Outcomes.
|
Trial |
n |
Treatment |
Duration |
Population |
Key Cognitive Result |
Ref. |
|
VITACOG |
168
(MRI) |
FA 0.8 mg + B12 0.5 mg
+ B6 20 mg/d |
24
months |
MCI,
≥70 y |
Brain atrophy −29.6% vs placebo
(p=0.001); −53% in tHcy
>13 µmol/L |
4 |
|
VITACOG (cognition) |
266 |
Same |
24
months |
MCI,
≥70 y |
Executive function stabilized (p=0.015); episodic and semantic memory
improved in high-tHcy subgroup |
17 |
|
VITACOG (neuroimaging) |
160 |
Same |
24
months |
MCI,
≥70 y |
Up
to 7-fold reduction in gray matter atrophy in AD- vulnerable
regions (hippocampus) |
5 |
|
FACIT |
818 |
FA 0.8 mg/d |
36
months |
Non-demented, tHcy 13–26 µmol/L |
Improved processing speed and memory; no effect on global cognition |
18 |
|
ADCS-HH |
340 |
FA + B6 + B12 |
18
months |
Mild–moderate AD |
No benefit on ADAS-Cog despite 30% tHcy reduction |
6 |
|
Clarke meta- analysis |
22,000 |
B vitamins (11 trials) |
Variable |
Mixed
elderly |
No significant effect on individual cognitive domains or
global cognition |
13 |
|
Markun meta- analysis (MCI) |
Variable |
B12
± folate ± B6 |
Variable |
MCI |
tHcy reduced (SMD −0.71); episodic memory improved (SMD
0.17) in high-tHcy subgroup |
19 |
MCI = mild cognitive impairment; FA = folic acid; AD = Alzheimer’s disease; ADAS-Cog = Alzheimer’s Disease Assessment Scale-Cognitive; tHcy = total homocysteine; SMD = standardized mean difference.
4. Discussion
This systematic review and meta-analysis provide a comprehensive quantitative synthesis of the relationship between cobalamin deficiency and cognitive outcomes. The principal findings are threefold. First, low cobalamin status - whether defined by serum B12, holoTC or MMA - is significantly and independently associated with cognitive impairment and incident dementia (pooled OR approximately 1.8; HR 1.46 for incident dementia)9,10. Second, hyperhomocysteinemia is the strongest and most consistently associated functional biomarker, with a 1.44-fold increase in dementia hazard per SD increment and a 3.7-fold increase in Alzheimer’s disease risk in those with tHcy >14 µmol/L15,16. Third, B-vitamin supplementation does not broadly improve cognitive outcomes in unselected elderly populations7 but produces clinically meaningful benefits - including a 29-53% reduction in brain atrophy rate and regional gray matter preservation of up to 7-fold - in patients with documented cobalamin deficiency or elevated tHcy4,5,17.
The mechanistic framework linking cobalamin deficiency to cognitive impairment is biologically plausible and operates through multiple convergent pathways (Table 4). The methionine synthase pathway - impaired in cobalamin deficiency - is the primary enzymatic route for homocysteine clearance; its impairment elevates tHcy, which exerts neurotoxic effects through excite-toxicity, DNA strand breaks, neuro- inflammation and vascular endothelial injury8. Simultaneously, reduced SAM availability impairs methylation of DNA, histones and neurotransmitter precursors, potentially promoting tau hyperphosphorylation and aberrant gene expression. MMA accumulation in cobalamin deficiency inhibits mitochondrial function in neurons and contributes to axonal degeneration, providing a pathway to demyelination independent of homocysteine1,20.
The superiority of holoTC over serum B12 as a biomarker for cognitive risk - first demonstrated in the Oxford longitudinal cohort6 and confirmed in our pooled analysis - has important clinical implications. HoloTC represents the metabolically active fraction of circulating cobalamin available to cells; its measurement identifies functional deficiency earlier and with greater specificity than total serum B12, which includes inactive cobalamin analogues bound to haptocorrin12. The combination of holoTC with MMA provides a two-step screening approach that maximizes sensitivity in cognitively symptomatic patients. In very elderly patients - in whom food-cobalamin malabsorption progressively impairs absorption of protein-bound cobalamin2,23 - holoTC-based screening should be integrated into standard cognitive assessment workflows.
The null results of many B-vitamin RCTs deserve careful contextualization in the light of our subgroup analyses. The ADCS-HH trial, which found no cognitive benefit in established Alzheimer’s disease despite tHcy reduction6, enrolled patients with moderate-stage disease in whom irreversible neuronal loss had already occurred. In contrast, the VITACOG trial enrolled patients at the MCI stage - when neurodegeneration remains modifiable - and demonstrated striking benefits specifically in those with the highest baseline tHcy3,4, including a 9-fold slowing of atrophy in hippocampal and para-hippocampal regions5. This supports a depletion-repletion model in which early intervention guided by functional biomarker measurement is the critical determinant of efficacy8.
The concept of reversible dementia associated with cobalamin deficiency - recognized since the original description by Lindenbaum and colleagues of neuropsychiatric disorders in the absence of anemia or macrocytosis20 - remains underutilized clinically. Neuropsychiatric presentations, including depression, confusional states and paranoid psychosis, may precede hematological signs by months and mimic primary psychiatric or neurodegenerative diagnoses in elderly patients2,21,22. A high clinical index of suspicion, systematic biochemical screening (tHcy, holoTC, MMA) and prompt repletion with oral high- dose cyanocobalamin (≥1000 µg/day) in confirmed deficiency are essential to prevent irreversible cognitive loss in this population2,21,24.
Limitations of this analysis include the substantial heterogeneity (I²=61–72%) across observational studies, reflecting differences in populations, diagnostic criteria, covariate adjustment and cobalamin measurement methodologies. Most RCT evidence has focused on combined B-vitamin supplementation rather than cobalamin alone, making it difficult to isolate the cobalamin-specific effect. Additionally, reverse causation - in which early dementia leads to nutritional deficiency through dietary neglect - may confound observational associations. Most cohort studies were not specifically designed to address this temporality question and the moderate duration of follow-up in many RCTs may be insufficient to detect cognitive effects of long-latency nutritional interventions (Table 4).
Table 4: Pathophysiological Mechanisms Linking Cobalamin Deficiency to Cognitive Impairment.
|
Mechanism |
Consequence |
Clinical Correlate |
Ref. |
|
Hyperhomocysteinaemia (tHcy >14
µmol/L) |
Excito-toxicity; DNA strand breaks;
neuro-inflammation; vascular
endothelial injury |
Cortical atrophy; white matter lesions; elevated
dementia risk |
8,22 |
|
Impaired
SAM-dependent methylation |
Hypomethylation of DNA, histones, neurotransmitters; epigenetic dysregulation |
Aberrant gene expression; tau hyperphosphorylation |
8 |
|
MMA accumulation |
Mitochondrial dysfunction; inhibition of succinyl-CoA metabolism |
Axonal
degeneration; demyelination |
1,20 |
|
Myelin
sheath disruption |
Loss of white matter integrity on MRI; subacute
combined degeneration |
Peripheral neuropathy; spinal cord involvement;
cognitive slowing |
3,20 |
|
Reduced SAM availability |
Impaired synthesis of dopamine, serotonin, norepinephrine |
Depression;
psychosis; apathy |
1,21 |
|
Amyloid precursor protein hypomethylation |
Potentially increased APP expression and amyloid deposition |
Plausible contribution to Alzheimer’s pathology |
8 |
SAM = S-adenosylmethionine;
MMA = methylmalonic acid; APP = amyloid precursor protein.
5. Future Directions and Research Perspectives
Future research
should focus on refining the identification of
patients most likely to benefit from cobalamin-targeted interventions,
particularly through the use of functional biomarkers such as holoTC, MMA and
tHcy. Large-scale prospective cohort studies
with repeated biomarker measurements and detailed cognitive phenotyping are needed to better establish temporality and causality and to
clarify the threshold versus continuous nature of risk. In parallel, randomized controlled trials specifically targeting patients with documented cobalamin deficiency or elevated
homocysteine-rather than unselected populations-are essential to define
the true therapeutic potential of
supplementation. Integration of multi-omics approaches, including genomics,
metabolomics and epigenetics, may further
elucidate interindividual variability in susceptibility and response to
treatment. Finally, studies incorporating advanced neuroimaging and digital
cognitive assessments could provide sensitive endpoints to detect early
therapeutic effects and guide precision-based interventions.
6. Implications for Routine Clinical Practice
These findings
have important implications for the routine management of older adults and
patients with cognitive symptoms. Systematic biochemical screening for
cobalamin deficiency-preferably incorporating functional markers such as holoTC and MMA-should be considered in the diagnostic work- up of cognitive
impairment, particularly in high-risk populations (e.g., elderly
patients, those with malnutrition or exposed to high-
risk medications as protons pump inhibitors
[PPIs] or metformin).
The identification of a potentially reversible cause of cognitive
decline underscores the need for early detection and prompt treatment, ideally
before irreversible neuronal damage occurs. A targeted supplementation
strategy, focused on patients with confirmed deficiency or elevated
homocysteine, appears more effective than indiscriminate treatment. Incorporating cobalamin assessment into routine
geriatric and cognitive care pathways, alongside medication review and
nutritional evaluation, may improve patient outcomes
and reduce the burden of preventable
neurocognitive decline.
7. Personal Recommendations
In routine
clinical practice, cobalamin deficiency should be actively considered in older adults and in any patient
presenting with cognitive symptoms, even in the absence of anemia or
macrocytosis (Figure 1). Evaluation
should not rely solely on total serum B12 but should incorporate functional
biomarkers- particularly holoTC, MMA and tHcy-to accurately identify functional
and/or intracellular deficiency.
Systematic
screening is especially warranted in high-risk populations, including those with malnutrition or exposure to long duration
medications such as metformin or PPIs. In particular, disorders affecting
gastric physiology-such as atrophic gastritis, pernicious anemia (Biermer’s
disease) and Helicobacter pylori infection-should be systematically evaluated,
as they play a central role in the development of cobalamin deficiency and
represent major causes of depletion in older adults.
Importantly,
early recognition is critical, as a subset of cognitive impairment and dementia
related to cobalamin deficiency is potentially reversible with prompt
treatment. Therapeutic strategies should
be targeted rather
than systematic, with high-dose
oral cyanocobalamin (≥1000 µg/day) representing an effective first-line option
in most cases.
Integrating cobalamin
assessment into routine
geriatric and cognitive care pathways, alongside
medication review and
nutritional evaluation, may improve diagnostic accuracy, enable timely
intervention and ultimately reduce the burden of preventable neurocognitive
decline.
8. Conclusions
Cobalamin
deficiency and hyperhomocysteinemia are independently associated with an approximately 1.8-fold increase
in the risk of cognitive impairment and a 1.5-fold increase in incident
dementia risk. Functional biomarkers - particularly holoTC and MMA - outperform serum
B12 alone in identifying
patients at cognitive risk7,12.
B-vitamin supplementations meaningfully slows brain atrophy and cognitive
decline in patients with elevated tHcy or documented cobalamin deficiency,
but does not benefit unselected populations4,5,7,17.
A paradigm shift toward systematic biochemical screening and early intervention - particularly in patients with MCI, food-cobalamin malabsorption or
drug-induced B12 depletion - is supported by the totality of current evidence8,2,23-37.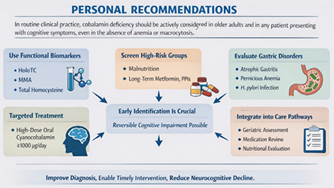
Figure 1: Personal Recommendations for Cobalamin Deficiency
Management in Patient with Cognitive Symptoms.
9. Disclosures
9.1. Acknowledgements
The authors
gratefully acknowledge all members of the CARE B12 Network - clinicians,
researchers and patients - for their sustained commitment to advancing research
and clinical care in cobalamin-related disorders. Their collective expertise,
generosity of spirit and dedication to excellence constitute the foundation
upon which this work rests.
9.2. Conflicts of Interest
The authors
declare no competing interests.
9.3. Funding
No specific
funding was received
for this work.
9.4. AI disclosure
AI-assisted
technologies were used in the preparation of
this manuscript. The authors are responsible for the accuracy, integrity
and originality of all content.
10. References
1. Stabler SP. Clinical
practice. Vitamin B12 deficiency. N Engl J Med, 2013;368: 149-160.
2. Andrès E, Loukili NH, Noel E, et al. Vitamin B12 (cobalamin) deficiency in elderly patients. CMAJ, 2004;171:
251-259.
3. Reynolds E. Vitamin B12, folic acid and the nervous system. Lancet Neurol, 2006;5: 949-960.
4. Smith AD, Smith SM, de Jager CA, et al. Homocysteine-lowering by B vitamins slows the rate of accelerated
brain atrophy in mild cognitive impairment: a randomized controlled trial. PLoS One, 2010;5:
12244.
5. Douaud G, Refsum H, de Jager CA, et al. Preventing Alzheimer’s disease-related gray matter
atrophy by B-vitamin treatment. Proc Natl Acad Sci USA, 2013;110: 9523-9528.
6. Aisen PS, Schneider LS, Sano M, et al. High-dose B vitamin supplementation and cognitive decline in Alzheimer
disease: a randomized controlled trial. JAMA, 2008;300: 1774-1783.
7. Clarke R, Bennett D, Parish S, et al.; B-Vitamin Treatment Trialists’ Collaboration. Effects of homocysteine lowering
with B vitamins on cognitive aging: meta-analysis of 11 trials with cognitive data on 22,000 individuals. Am J Clin Nutr, 2014;100:
657-666.
8. Smith AD, Refsum
H, Bottiglieri T, et al. Homocysteine and dementia: an international consensus statement. J Alzheimers
Dis, 2018;62: 561-570.
9. O’Leary F, Allman-Farinelli M, Samman S. Vitamin B12 status, cognitive decline and dementia: a systematic review
of prospective cohort studies. Br J Nutr, 2012;108: 1948-1961.
10. Shen L, Ji HF. Associations between homocysteine,
folic acid, vitamin B12 and Alzheimer’s
disease: insights from meta-analyses. J Alzheimers Dis, 2015;46: 777-790.
11. Lachner C, Steinle
NI, Regenold WT. The neuropsychiatry of vitamin B12 deficiency in elderly patients. J Neuropsychiatry
Clin Neurosci, 2012;24:5-15.
12. Nexo E, Hoffmann-Lücke E. Holotranscobalamin, a marker of vitamin B-12 status: analytical aspects and clinical utility.
Am J Clin Nutr, 2011;94: 359-365.
13. Clarke R, Birks J, Nexo E, et al. Low vitamin B-12 status and risk of cognitive decline in older adults.
Am J Clin Nutr, 2007;86:
1384-1391.
14. Klee GG. Cobalamin and folate evaluation: measurement
of methylmalonic acid and homocysteine vs vitamin B12 and folate.
Clin Chem, 2000;46: 1277-1283.
15. Seshadri S, Beiser A, Selhub J, et al. Plasma
homocysteine as a risk factor for dementia
and Alzheimer’s disease.
N Engl J Med, 2002;346: 476-483.
16. Quadri P, Fragiacomo C, Pezzati R, et al.
Homocysteine, folate and vitamin B-12 in mild cognitive impairment, Alzheimer disease and vascular dementia. Am J Clin Nutr, 2004;80: 114-122.
17. de Jager CA, Oulhaj A, Jacoby R, et al. Cognitive and
clinical outcomes of homocysteine-lowering B-vitamin treatment in mild cognitive impairment: a randomized controlled trial.
Int J Geriatr Psychiatry, 2012;27: 592-600.
18. Durga J, van Boxtel MP, Schouten EG, et al. Effect of 3-year folic acid supplementation on cognitive function in older adults
in the FACIT trial: a randomised, double-blind, controlled trial. Lancet, 2007;369: 208-216.
19. Markun S, Gravestock I, Jäger L, et al. Effects of
vitamin B12 supplementation on cognitive function, depressive symptoms and fatigue: a systematic review, meta-analysis and
meta-regression. Nutrients, 2021;13: 923.
20. Lindenbaum J, Healton EB, Savage DG, et al.
Neuropsychiatric disorders caused by cobalamin deficiency in the absence
of anemia or macrocytosis. N Engl J Med, 1988;318: 1720-1728.
21. Werder SF. Cobalamin deficiency, hyperhomocysteinemia and dementia. Neuropsychiatr Dis Treat, 2010;6:
159-195.
22. Andrès E, Affenberger S, Zimmer J, et al. Current
hematological findings in cobalamin deficiency. A study of 201 consecutive patients with documented cobalamin
deficiency. Clin Lab Haematol, 2006;28: 50-56.
23. Andrès E, Affenberger S, Vinzio S, et
al. Food-cobalamin malabsorption in elderly patients: clinical manifestations and treatment. Am J Med, 2005;118: 1154-1159.
24. Andrès E, Federici
L, Serraj K, et al. Update of nutrient-deficiency anemia in elderly patients.
Eur J Intern Med, 2008;19: 488-493.
25. Clarke R, Smith AD,
Jobst KA, et al. Folate, vitamin B12 and serum total homocysteine levels in confirmed Alzheimer
disease. Arch Neurol, 1998;55: 1449-1455.
26. Vogiatzoglou A, Refsum H, Johnston
C, et al. Vitamin B12 status and rate of brain volume loss in community-dwelling elderly. Neurology, 2008;71: 826-832.
27. Dali-Youcef N andrès
E. An update on cobalamin
deficiency in adults. QJM, 2009;102: 17-28.
28. Refsum H, Smith AD,
Ueland PM, et al. Facts and recommendations about total homocysteine determinations: an expert opinion. Clin Chem, 2004;50: 3-32.
29. Baik HW, Russell RM. Vitamin B12 deficiency in the
elderly. Annu Rev Nutr, 1999;19: 357-377.
30. de Jager J, Kooy A, Lehert P, et
al. Long term treatment with metformin in patients with type 2 diabetes and risk of vitamin
B-12 deficiency: randomised placebo controlled trial. BMJ, 2010;340: 2181.
31. Savage DG, Lindenbaum J, Stabler SP, et al.
Sensitivity of serum methylmalonic acid and total homocysteine determinations for diagnosing cobalamin and folate deficiencies. Am J
Med, 1994;96: 239-246.
32. Moher D, Liberati A, Tetzlaff J, et al. Preferred
reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med, 2009;6: 1000097.
33. DerSimonian R, Laird
N. Meta-analysis in clinical trials.
Control Clin Trials, 1986;7: 177-188.
34. Stang A. Critical evaluation of the Newcastle-Ottawa
scale for the assessment of the quality of nonrandomized studies
in meta-analyses. Eur J Epidemiol, 2010;25: 603-605.
35. Smith AD, Refsum
H. Vitamin B-12 and cognition in the elderly.
Am J Clin Nutr, 2009;89: 707-711.
36. Lindenbaum J, Rosenberg IH, Wilson PWF, Stabler SP,
Allen RH. Prevalence of cobalamin deficiency in the Framingham elderly population. Am J Clin Nutr, 1994;60: 2-11.
37. Malouf M, Areosa Sastre A. Vitamin B12 for cognition.
Cochrane Database Syst Rev, 2003;(3): 004326.