Photobiomodulation of Aging Tissues: Optimization of Dosimetric Parameters, Optical Modeling and Biphasic Dose Response
Abstract
Aging is characterized by progressive mitochondrial dysfunction, oxidative stress, chronic inflammation and impaired tissue regeneration, all of which culminate in degenerative conditions and poor healing capacity. Photobiomodulation (PBM), which utilizes red and near-infrared light (600-1000 nm), represents an emerging, non-invasive treatment modality that can be used to regulate cellular processes for maintaining tissue homeostasis. This review aims at providing a thorough overview of PBM with regards to aging, from cell biology to optics and from clinical applications to dosimetry. In terms of cellular biology, PBM involves the absorption of photons by mitochondrial photosensitizers resulting in ATP production stimulation, ROS signaling modulation, as well as activation of signaling cascades responsible for inflammation resolution and tissue repair. Nevertheless, PBM's therapeutic effect is greatly dependent upon optical properties of aging tissues, where light scattering increases along with the change in the chromophore concentration profile affecting light fluence distribution. The biphasic dose–response relationship is of particular interest here, as it demonstrates the presence of the optimal therapeutic window in which photobiomodulation stimulates biological effects, but low and high dosages can fail to work properly. The usage of modern computational techniques like the Monte Carlo simulation of light transport is suggested as a key factor for predicting energy deposition. From a clinical point of view, photobiomodulation shows promise in dealing with chronic wounds, musculoskeletal issues, neurodegenerative diseases and aging of the skin. Generally speaking, this paper focuses on the necessity to combine biophysical and biological knowledge to advance photobiomodulation in geriatric medicine.
Keywords: Aging, Wound healing, Photobiomodulation, Tissue regeneration, Low-level laser therapy, Dosimetry.
1. Introduction
The aging process itself is very complicated biologically and entails the occurrence of deterioration in cellular physiology, decreased regenerative capabilities and increased vulnerability to illnesses and other ailments. Such changes occur as a result of mitochondrial abnormalities, oxidative stress, chronic inflammatory response and decreased reparative properties of tissue structures. These findings indicate that problems such as chronic wounds, joint degeneration, neurodegeneration and skin aging constitute serious issues in contemporary medicine1.
Recent developments in photobiomodulation (PBM), formerly referred to as low-level laser therapy (LLLT), have identified it as a potential non-invasive intervention that involves modulating cellular function to promote tissue regeneration. This form of intervention involves the use of photons from red and near-infrared light ranging from 600–1000 nm to induce photochemical processes within cells, unlike other methods involving heat. On a cellular level, photons are absorbed by mitochondrial chromophores, especially cytochrome c oxidase, thereby improving electron transport, generating ATP and influencing ROS signaling2.
However, while these effects appear to be significant, it should be emphasized that their effectiveness heavily relies on the interaction between light and tissue. The latter involves processes such as absorption, scattering and attenuation of optical energy, all of which may be considerably affected by aging. Such changes are mainly connected to the alteration in composition and structure of biological tissues, which include, among other things, the formation of cross-links in collagen fibers, lower content of water and differences in chromophores’ distribution3.
An important feature of PBM is its biphasic dose-response pattern wherein lower dosages cause stimulation, whereas higher dosages may induce inhibition. Such non-linearity underscores the need for proper dosimetry, especially when applying PBM on geriatric patients since tissue sensitivity and optics have changed. Nevertheless, there is a lack of standardization in PBM techniques today and the ideal dosing protocol remains underdeveloped4.
In such cases, computational methods, especially those based on Monte Carlo simulation for photon transport, have proven to be vital tools that help better understand light propagation, fluence and energy deposition processes in biological tissue. In this regard, computer models will provide a way to predict accurately the dose distribution in target tissue layers and allow for optimization of PBM parameters for elderly patients.
Although several quantitative dosimetry schemes have been developed for PBM, no scheme that is specific to the optical properties of aged tissue and fluence attenuation has yet been established.
For this reason, this review attempts to give an integrated look at photo biomodulation processes in aged tissue in terms of biological mechanism, optics and dosimetry optimization and clinical applications. Special attention will be paid to fluence attenuation and biphasic dose-response effects on therapy efficiency. This way, the connection between biological and biophysical aspects of the problem under consideration will be made in order to facilitate the creation of new PBM protocols for geriatric patients.
2. Principles of Photo Biomodulation
Photo biomodulation (PBM), also called low-level laser therapy (LLLT), is a noninvasive treatment approach that employs light from red and near-infrared wavelengths for the modulation of biological responses without thermal effects. PBM does not rely on thermal effects but instead operates through photochemical and photobiological processes unlike high-power laser applications that make use of thermal or ablative effects in achieving biological objectives. The basic processes involved in PBM, including mitochondrial activation, redox processes, regulation of inflammatory responses and tissue repair, are depicted schematically in (Figure 1). The mechanism of action of PBM begins with photon absorption by intracellular chromophores in the tissues leading to series of biochemistry and signaling events. As indicated in (Figure 1), photon absorption causes the activation of mitochondria, increase in ATP formation, controlled ROS formation and activation of transcription factors involved in the control of inflammation and cell proliferation. Thus, the response achieved depends on optical dose and tissue characteristics5.
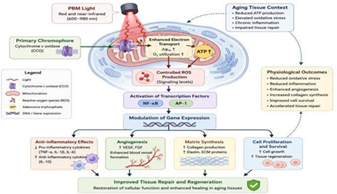
Figure 1: Mechanisms of photobiomodulation (PBM) at the cellular and molecular level. Diagrammatic representation of the basic mechanisms of PBM. Photobiomodulation involves absorption of red and near infrared light (600 - 980 nm) by mitochondrial chromophores such as cytochrome c oxidase (CCO), which leads to an increase in electron transport activity and increased levels of ATP formation. ROS formation that occurs in a controlled manner acts as signaling molecules responsible for the activation of transcription factors including NF-kB and AP-1, thereby regulating gene expression. As a result, down-regulation of pro-inflammatory cytokines like TNF-α and IL-1β, increased expression of anti-inflammatory cytokines such as IL-10, angiogenesis through VEGF up-regulation and stimulation of collagen synthesis take place.
2.1. Light-tissue interactions
The physics of light-tissue interaction is described by processes such as absorption, scattering, reflection and transmission. As soon as light reaches a biological sample, part of it gets absorbed due to the presence of various chromophores including melanin, hemoglobin and water, whereas the rest is subjected to scattering from tissue inhomogeneity.
As far as photobiomodulation (PBM) applications are concerned, the wavelength region from 600 to 1000 nm or an optical window, is preferable because of low levels of absorption and scattering of photons. Nevertheless, the behavior of light in tissues depends strongly on the characteristics of the latter and age-related changes may affect it dramatically6.
2.2. Mitochondrial photoreception
It has been widely reported that mitochondria act as the chief cellular target in PBM therapy. Cytochrome c oxidase (CCO), a crucial component in the mitochondria’s electron transfer chain, serves as a prime candidate for photoacceptor. The absorption of photon results in electron transfer, mitochondrial membrane depolarization and eventually ATP production.
Furthermore, PBM therapy contributes to the photodissociation of NO from CCO, thus ensuring better oxygen metabolism. While the involvement of CCO is well- documented, several other processes have been suggested as being responsible for photobiomodulation including photon interaction with water bound within mitochondria or other photosensitizers. As depicted in Figure 1 below, these processes suggest that PBM works via a multi-level mechanism rather than a single one7,8.
2.3. Cellular and molecular responses
The primary photochemical effects triggered by PBM cause a chain reaction of other secondary cellular effects, such as increased ATP production, ROS modulation and activation of transcription factors, like NF-κB and AP-
1. In regulated amounts, ROS function as signalers, responsible for the control of gene expression, cell proliferation and adaptation to stressors9.
Photo biomodulation is also effective in reducing inflammatory cytokines (for instance, TNF-α and IL-1β) and stimulating anti-inflammatory cytokine production (for example, IL-10). Additionally, PBM stimulates angiogenesis through VEGF pathways and increases the activity of fibroblasts, causing an increase in collagen formation and extracellular matrix remodeling. The interrelated processes are illustrated in (Table 1) and synthesized in (Figure 1).
Table 1: Cellular and molecular mechanisms of photobiomodulation (PBM) and their biological importance.
|
Mechanism |
Primary Target |
Molecular/Cellular Effect |
Biological
Outcome |
Relevance in Aging Tissues |
References |
|
Photon
absorption |
C y t o c h r o m e coxidase (CCO) |
Activation of electron transport chain |
Initiation of PBM response |
Reduced
efficiency in aged mitochondria → requires optimized dosing |
8,10 |
|
ATP production ↑ |
Mitochondria |
Increased oxidative phosphorylation |
Enhanced cellular energy metabolism |
Restores impaired bioenergetics in aged
cells |
11,12 |
|
ROS
modulation |
Mitochondria / cytoplasm |
Transient increase in
signaling ROS |
Activation of redox- sensitive pathways |
Helps rebalance oxidative stress in aging |
13 |
|
Gene activation |
NF-κB, AP-1 |
Upregulation of transcription and
protein synthesis |
Cell survival and proliferation |
Supports regeneration in aged tissues |
14 |
|
Anti- inflammatory response |
Cytokines (TNF-α, IL-1β, IL-10) |
↓ Pro-inflammatory,
↑ anti- in- flammatory mediators |
Reduced inflammation |
Counteracts “inflammaging” |
12 |
|
Angiogenesis |
VEGF, endothelial cells |
Increased vascular signaling |
New blood
vessel formation |
Improves oxygenation in aged tissues |
15,16 |
|
Collagen synthesis |
Fibroblasts |
Increased extracellular matrix
production |
Tissue remodeling and repair |
Enhances skin and wound
healing in elderly |
17 |
2.4. Biphasic dose response
One of the main features of PBM therapy is the presence of the
biphasic dose-response relation, which is well described by the Arndt-Schulz law. According to the law, small doses
of light activate biological processes, while large
doses may suppress the physiological functions of cells. Thus, the nonlinear
dependency reflects the presence of a special window of therapeutic
action when phototherapy shows
its positive effect.
At lower energies, no effects may be observed
since there is no adequate response from the biological system, while at high energies, cell activation
is suppressed.
Biphasic behavior is especially important
in elderly patients whose metabolism and optical
properties differ significantly from those in young organisms. Thus, strict
regulation of all parameters of irradiation (wavelengths, irradiance, fluence
and time of irradiation) is critical for success in PBM therapy18.
3. Mechanisms of Photo biomodulation in Aging
The mode of action for Photo biomodulation therapy relies on several
mechanisms, which include photochemical reactions, biochemical reactions and
cellular actions. In the context of aging tissue, PBM acts through the
restoration of bioenergetics in cells, regulation of redox signaling, modulation of the immune
response and stimulation of regeneration in tissues. (Table 2) provides an overview of some of the physiological
pathways involved in the action mechanism of photo biomodulation therapy, while
Figure 2 describes how the optical energy distribution affects aging tissues.
3.1. Mitochondrial activation and restoration of cellular bioenergetics
A major pathway in PBM is the uptake of photons at wavelengths
between 600 nm and 1,100 nm into mitochondria by photo acceptors, specifically cytochrome c oxidase. This facilitates enhanced
electron transport and increases mitochondrial membrane potential19.
As aging occurs, mitochondrial metabolism decreases, reducing energy levels and repair rates for tissues.
PBM acts on the mitochondria to help regenerate
cell division and migration as well as metabolic reactions. Also, PBM can
result in the separation of NO from cytochrome c oxidase, thus facilitating
oxidative phosphorylation and oxygen metabolism20.
3.2. Modulation of reactive oxygen species and redox signaling
ROS have a dual impact on biological organisms, as they can act as
destructive substances as well as messengers. PBM induces a controlled increase
in ROS levels that will activate redox-responsive signaling pathways and
transcription factors, including NF-κB and AP-1. Oxidative stress is often high
in older organisms; however, their protective antioxidants may be weakened. PBM works by balancing ROS levels and improving
cell adaptation as well as antioxidant activity in general. It is essential to
maintain control over ROS because this process helps regulate gene expression
and other functions21,22.
3.3. Anti-inflammatory and immunomodulatory effects
One of the features of aging is the chronic low-grade state of inflammation in the body, called
“inflammaging.” PBM downregulates the production of pro-inflammatory molecules,
such
as, TNF-α, IL-1β and cyclooxygenase-2, while upregulating
anti-inflammatory cytokines such as, IL-10.
Moreover, PBM regulates the activity of immune cells, mainly
affecting the process of macrophage polarization and inducing the conversion of
macrophages from M1 phenotypes to M2 phenotypes. This leads to the resolution of inflammation
and remodeling of tissues, which
is extremely important
in cases when the body is aged23,24.
3.4. Stimulation of proliferation, angiogenesis and Tissue regeneration
PBM improves the function of important cell types responsible for tissue repair, such
as fibroblasts, keratinocytes, endothelial cells and stem or progenitor cells. This results in enhanced cell
proliferation, migration and production of extracellular matrix, which in turn
increases tissue regeneration
capacity25,26. Additionally, PBM
induces angiogenesis by increasing the expression of growth factors
like VEGF and TGF-β. Such
actions increase blood flow, oxygen supply and
nutrition delivery, which can be hampered in aging tissues. All of these biological
mechanisms contributing to tissue regeneration are outlined in (Table 2).
Table 2: Mechanistic pathways of photobiomodulation (PBM) and their biological relevance
in aging tissues.
|
Mechanism |
Primary Target |
Key Biological Effects |
Relevance in Aging Tissues |
References |
|
M i t o c h o n d r i a l activation |
Cytochrome c oxidase (CCO) |
Enhanced
electron transport, ↑ ATP production, improved mitochondrial membrane potential |
Restores impaired cellular bioenergetics and
metabolic activity in aged cells |
27 |
|
Nitric oxide photodissociation |
CCO–NO complex |
Release of inhibitory NO, improved oxygen utilization and respiration |
Enhances mitochondrial efficiency under
hypoxic or aged
conditions |
28 |
|
ROS modulation |
Redox-sensitive
signaling pathways |
Transient ROS increase → activation of NF-κB, AP-1,
antioxidant response |
Rebalances
oxidative stress and restores
redox homeostasis |
20,29,30 |
|
Anti-inflammatory regulation |
Cytokines (TNF-α, IL-1β, IL-10) |
↓ Pro-inflammatory mediators, ↑ anti- inflammatory
cytokines |
Reduces chronic inflammation (inflammaging) |
31 |
|
M a c r o p h a g e polarization |
Immune cells
(M1 → M2) |
Shift toward
reparative phenotype, enhanced tissue remodeling |
Promotes resolution of inflammation and healing in elderly tissues |
32 |
|
Cellular proliferation |
Fibroblasts, keratinocytes |
↑ Cell
proliferation, migration, collagen synthesis |
Accelerates delayed wound healing
in aging |
33-35 |
|
Angiogenesis |
VEGF, TGF-β
pathways |
↑ Neovascularization, improved blood supply |
Enhances oxygen
and nutrient delivery to aged tissues |
36-38 |
|
Stem cell
activation |
Mesenchymal stem cells (MSCs) |
↑ Differentiation and regenerative capacity |
Compensates for age-related decline in stem cell
function |
7,39,40 |
|
Optical–tissue interaction |
Tissue chromophores (melanin, Hb,
water) |
Light absorption, scattering, depth- dependent fluence
distribution |
Alters effective dose delivery in aged
tissues |
41-43 |
|
Biphasic dose response |
Cellular metabolic response |
Stimulation at optimal dose, inhibition at excessive dose |
Requires precise
dosimetry in geriatric applications |
18,44-46 |
3.5. Optical and biophysical considerations in aging tissue
Moreover, besides biological factors, the success of PBM therapy is
significantly dependent on the physics of light transport through tissue. With
age, the physical parameters of tissue change due to alterations in collagen structure, hydration, vascularity and chromophore concentration. This
impacts the optical properties of tissues by affecting absorption, scattering
and anisotropy.
(Figure 2(A)) clearly indicates that the intensity of light diminishes from
maximum values at the surface to lower levels at increasing depths. Moreover,
in (Figure 2(B)), it can be observed that fluence is exponentially related
to depth according to the effective attenuation
coefficient (μeff). Consequently, optical changes with age impact the depth of light
penetration into tissue47-49.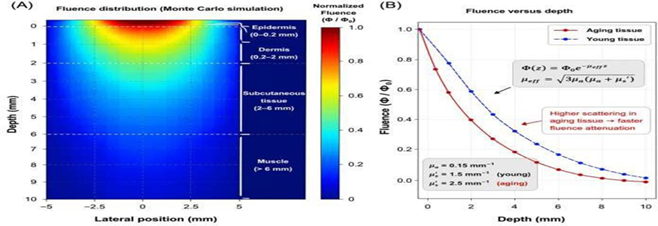
Figure 2: Penetration and fluence
distribution of light
used for photobiomodulation therapy in aging tissues.
Fluence distribution map obtained by Monte Carlo simulation demonstrating light intensity
distribution in a multilayer tissue model, where the absorption is maximal at
the surface layer while the light intensity decreases with depth.
Fluence profile as a function of depth depicting the exponential
decrease in light intensity due to the effective attenuation coefficient (μeff).
3.6. Biphasic dose-response relationship
PBM exhibits a biphasic dose-response relationship response
where small doses cause increased cell activity while large doses can cause
inhibition, with the optimum dose giving the maximum effect. The biphasic
effect of PBM is especially important during use on aged individuals who have
lower metabolic abilities, hence
altering their sensitivity to light. Thus, proper optimization of optical
parameters is necessary for an efficient and
optimized application during treatment of older individuals. Understanding the
physiology of aging and using this information along with optical theory helps
determine the correct treatment procedure50-53.
Despite considerable research into the physiological pathways
involved in photobiomodulation, notable variations can be seen among various
experimental setups. While a large number of researchers have observed improved
mitochondrial function and ATP generation due to exposure to PBM, others have
reported limited or no significant effects based on the wavelength, light intensity and biological conditions of cells54,55.
In addition to the above, the involvement of cytochrome c oxidase as the main photo acceptor
is also disputed, with certain
researchers proposing that it could
involve other molecules such as the water bound to the mitochondria or other
chromophores. The differences suggest that PBM physiology varies among
different tissues and ages.
Hence, there is a need for an extended mechanism that will factor in
the aforementioned differences in order to achieve accurate predictions of PBM
results in aging tissues.
4. Optical Interaction with Aging Tissue
The therapeutic effectiveness of PBM is regulated by both cellular
responses as well as light propagation inside biological tissues. Optical
changes related to aging can have a significant impact on the way in which
light propagates inside biological tissues,
thus impacting the amount of light that can be effectively
delivered, as seen in Figure 2 below. Parameters of light propagation are
highlighted in (Table 3).
4.1. Age-related changes in tissue optical properties
Tissue composition is significantly altered with the process of
aging. These changes in tissue composition directly impact the optical
characteristics. The changes
include decreased water content, changes in the structure of collagen and changes in the concentration of chromophores such as melanin and hemoglobin.
All these changes impact the basic optical parameters such as the absorption coefficient (μₐ), scattering coefficient (μ↑) and the anisotropy factor (g)56.
Changes in the structure of collagen with the process
of aging include increased
collagen cross-linking and disorganization. These changes impact the scattering
characteristics. Similarly, changes in tissue pigmentation and vascularization impact
the absorption characteristics57,58. The changes
in tissue composition with the process of aging
increase the optical heterogeneity of the tissue, which impacts the propagation
of photons.
4.2. Light attenuation and fluence distribution
The propagation of light in biological tissues is affected by
absorption and scattering phenomena. Consequently, there is exponential
attenuation as light travels deeper into tissues. This can be characterized as follows:
|
Where I (z) represent light intensity at a depth z, I0 represents incident light intensity and μeff represents effective attenuation coefficient. The effective attenuation coefficient can be characterized as follows:
|
Where μs′ = μs (1-g) is the reduced
scattering coefficient.
As seen from Figure 2(B), fluence falls off sharply with depth,
suggesting that a substantial fraction of optical energy is lost to absorption
and scattering in the outer layers of tissue. In aged tissues, the enhanced
scattering effect causes even greater attenuation of the beam.
4.2. Monte carlo modeling of photon transport
accurate description of the phenomenon. Therefore, the Monte Carlo
(MC) simulation has been recognized
as the gold standard in the simulation of photon transport59,60.
This approach considers
the simulation of the path of
single photons as they experience multiple scattering and absorption. The
spatial distribution of the MC simulation is presented in Figure 2(A), where
the distribution of the photon fluence decreases from the surface with depth.
Moreover, the MC simulation offers the possibility of simulating the realistic
multi-layer model of the tissue involved. This makes the MC simulation highly
appropriate for the simulation of the aging tissues involved.
4.3. Implications for PBM dosimetry in aging
The optical properties of aging tissues have significant implications for PBM
dosimetry. As illustrated in Figure 2(A-B), the energy deposition becomes
superficial, while the fluence
at the deeper layer is greatly reduced. Hence, it is crucial for PBM protocols
in geriatric medicine to consider these changes in optical properties by appropriately
modifying the parameters used in PBM, including the wavelengths, power density
and exposure time52,61. The
optical parameters relevant for light propagation and their implications in
aging tissues are listed in (Table 3).
4.4. Optimization strategies
Optimization methods for dealing with the problem
of light attenuation in aging
tissues include:
• Utilization of long wavelength light (800-980 nm) for
increased penetration
• Modification of surface fluence to account for attenuation loss at increasing depths
• Utilization of Monte Carlo based simulation models
for treatment planning
•Tailored PBM therapy according to tissue or patient specificity
All of these optimization techniques lead to greater
light control and PBM efficacy.
Table 3: Optical parameters and their impact on photo
biomodulation in aging tissues.
|
Parameter |
Description |
Effect in Aging Tissues |
Impact on PBM |
References |
|
(µa) (absorption coefficient) |
Light absorption by chromophores (melanin, hemoglobin, water) |
Altered chromophore distribution
with age |
Modifies energy deposition profile |
43,62,63 |
|
(µs) (scattering coefficient) |
Photon scattering within tissue microstructure |
Increased due
to collagen cross-
linking and structural changes |
Reduces penetration depth |
62,64,65 |
|
(g) (anisotropy factor) |
Directionality of scattering |
Slight variation with tissue aging |
Influences photon propagation direction |
62,66,67 |
|
(µeff) (effective attenuation coefficient) |
Combined absorption and scattering effects |
Increased in aging tissues |
Accelerates light attenuation |
67,68 |
|
(Φ)
Fluence |
Optical energy
per unit area within tissue |
Reduced at deeper layers |
Limits therapeutic effectiveness in deeper tissues |
68,69 |
5. Clinical Applications of Photobiomodulation in Geriatrics
Photobiomodulation (PBM) has become a highly prospective non-invasive method of treating various aging- related pathologies. Due to its capacity to affect mitochondrial functioning, oxidative stress and inflammation, photobiomodulation becomes an effective way to influence the most important pathological mechanisms of tissue dysfunction related to aging. The connection between biological effects caused by photo biomodulation and clinical results can be seen in (Figure 3); whereas clinical uses of PBM are described in (Table 4).
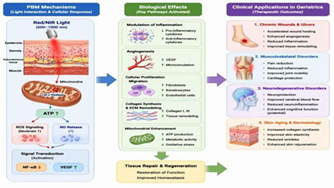
Figure 3: Photobiomodulation (PBM) applications in geriatrics
medicine. Scheme illustrating the connections between the biological
processes initiated via PBM and its therapeutic effects.
Light activation of mitochondrial chromophores increases the
production of ATP and ROS signaling, which results in regulation of
inflammatory responses, neovascularization and tissue regeneration.