Urban-Rural Disparities and Gender Dynamics in Obesity: Pioneering Strategies for Tailored Prevention and Treatment
Abstract
Background: Obesity is a complex, multifactorial
health crisis with significant disparities influenced by gender and environment,
particularly across urban and rural
settings worldwide. Understanding
these disparities is vital for
developing targeted prevention and
intervention strategies.
Objectives: This review aims to explore the biological, behavioral, environmental and socio-cultural factors contributing to urban-rural
and gender disparities in obesity. It emphasizes the importance of personalized
and culturally sensitive approaches for effective management.
Methods: A comprehensive synthesis of current
epidemiological data, biological mechanisms, environmental influences and policy strategies was
conducted, integrating quantitative figures, models and recent advances in
genomics, metabolomics and digital health.
Results: Findings reveal that women generally exhibit higher obesity prevalence
in high-income countries, largely due to hormonal
influences and sociocultural norms, whereas disparities in low- and middle-income countries are influenced by urbanization and
dietary transitions. Biological
differences in fat distribution and hormonal regulation, coupled with environmental and behavioral factors,
underpin these patterns. Urbanization exacerbates risk through sedentary lifestyles and processed food availability, with significant gender-specific implications. Emerging technologies and policy measures offer promising avenues for personalized
and equitable obesity management.
Conclusion: Addressing obesity
disparities requires an integrated approach that considers biological,
environmental and psychosocial factors. Advances in precision medicine, digital health and multisectoral policies are critical for developing effective, culturally appropriate interventions aimed at reducing global obesity burden and health inequalities.
Keywords: Obesity, Gender
disparities, Urban-rural differences,
Adipose tissue distribution, Hormonal regulation,
Environmental factors
1. Introduction
1.1. Background
Obesity has
emerged as a formidable global health crisis, transcending geographical,
socio-economic and cultural boundaries. Characterized by an abnormal or
excessive accumulation of adipose tissue, obesity poses significant risks to
individual and public health, contributing to the etiology and progression of numerous chronic
conditions. According to the
World Health Organization (WHO), in 2020, more than 650 million adults
worldwide were classified as obese, a figure that has approximately tripled
since 1975 when global obesity prevalence was considerably lower1. This alarming trend underscores the urgent
need for comprehensive understanding and targeted intervention strategies2.
Obesity is a
complex, multifactorial disease influenced by an interplay of genetic,
hormonal, behavioral, environmental and socio-economic factors. The
pathophysiology involves dysregulation of energy homeostasis, adipocyte
hypertrophy and hyperplasia and alterations in metabolic signaling
pathways. The consequences extend beyond mere excess weight, leading to
metabolic syndrome, type 2 diabetes mellitus (T2DM), cardiovascular diseases (CVD), certain cancers and psychosocial impairments.
The global burden of obesity is substantial; the WHO estimates that
obesity-related conditions account for 4 million deaths annually and contribute
to increased healthcare costs, diminished quality of life and reduced life
expectancy3.
Despite the universal nature of obesity,
its prevalence and
manifestation exhibit significant disparities based on demographic, geographic
and socio-economic factors. Among these, the influence of gender and
residential environment has garnered increasing attention due to their profound
impact on epidemiological patterns, biological mechanisms, behavioral
determinants and therapeutic outcomes4.
1.2. Definitions and basic concepts
Obesity is
typically quantified using the Body Mass Index (BMI), a simple anthropometric measure calculated as weight in kilograms divided by height in meters squared (kg/m²). The WHO classifies
adult obesity as a BMI ≥30 kg/m², with overweight defined as BMI between 25 and
29.9 kg/m². BMI alone does not
adequately capture adipose tissue distribution or metabolic health; hence,
additional metrics such as waist circumference, waist-to-hip ratio and body
composition analyses are employed to assess central versus peripheral fat
accumulation5.
1.2.2. Gender differences:
Biological sex and hormonal milieu significantly determine adipose tissue distribution and function. Women generally exhibit higher total body fat percentages (25-30%) compared to men (20-25%) at similar BMI levels, with a propensity for gluteofemoral fat accumulation. Men tend to develop central obesity, which correlates with higher cardiometabolic risk. Postmenopausal women experience hormonal shifts leading to increased visceral fat and metabolic disturbances7.
1.2.3. Environmental factors: Urbanization and rurality influence lifestyle behaviors, dietary patterns, physical activity levels and exposure to environmental pollutants all of which modulate obesity risk. Urban environments often favor sedentary lifestyles and greater availability of calorie-dense foods, whereas rural settings may involve more physical labor but are increasingly affected by dietary westernization. This introduction underscores the complexity of obesity as a multifaceted disease influenced by an interplay of individual biological characteristics and environmental exposures. Recognizing the disparities rooted in gender and residential environment is essential for advancing personalized medicine, improving health outcomes and designing effective public health interventions8.
The necessity of understanding the complex interactions between gender and environment in the context of obesity cannot be overstated. Such insights are vital for the development of targeted prevention strategies, personalized treatment modalities and equitable health policies aimed at curbing the global obesity epidemic9.
1.3. Gender and residential area-specific advances in obesity
Extensive investigations have also explored associations between anthropometric measures such as Body Mass Index (BMI), Body Surface Area (BSA) and Body Shape Index (BSSI) and cardio-metabolic risk factors, including obesity and Type 2 Diabetes Mellitus (T2DM). These indices demonstrate promising utility as predictive markers for metabolic syndrome and related comorbidities, thereby serving as valuable tools for early diagnosis and targeted intervention in the context of personalized medicine20,11-13,17-19. Given the biological and environmental disparities discussed in recent literature such as gender-specific fat distribution, hormonal influences and environmental pollutants these anthropometric indices can enhance risk stratification tailored to regional and gender- specific profiles19.
Several studies have critically assessed the performance and applicability of various percentile estimation techniques and anthropometric indices across different age groups and sexes, underscoring the importance of developing gender- and age-specific reference standards11-13,21,16,19. Recent research advocates for the incorporation of BSSI as a superior prognostic marker for mortality risk and metabolic disturbances, aligning with the trend toward precision medicine and personalized health assessment19. These investigations contribute significantly to advancing anthropometric science within the South Asian context, providing a robust foundation for implementing population-specific growth assessment protocols. Such tailored approaches are essential for early detection of growth abnormalities, metabolic risks and obesity-related complications, ultimately improving health outcomes through more accurate, culturally relevant clinical evaluations.
1.4. Significance of study
Understanding the heterogeneity in obesity patterns is critical for developing effective, culturally sensitive and equitable prevention and treatment strategies. Evidence indicates that obesity prevalence is not uniform across populations but varies markedly based on sex, age, socio-economic status and geographic location. In high-income countries such as the United States and many European nations, women often demonstrate higher obesity prevalence compared to men, with estimates reaching approximately 40-45% among women versus 35-40% among men22,23. Conversely, in low- and middle-income countries, the gender gap may be narrower or even reversed, reflecting divergent socio-cultural norms, occupational shifts and lifestyle transitions.
The urban-rural divide significantly influences obesity epidemiology. Urban populations tend to exhibit higher obesity prevalence, attributed to sedentary lifestyles, increased access to processed foods and environmental factors conducive to weight gain. For example, data from Pakistan suggest that urban residents have an obesity prevalence of approximately 25-30%, compared to 15-20% among rural residents. Rural areas are not immune to rising obesity rates, especially amidst dietary westernization and mechanization of labor. Elucidating the intricate relationship between gender and residential environment is essential because these factors do not operate independently; rather, their intersectionality can compound disparities, thereby necessitating nuanced, targeted interventions.
1.5. Objectives of the study
The epidemiology of obesity reveals notable disparities influenced by gender and residential environment, with prevalence rates varying significantly across regions and settings. Globally, women tend to have higher obesity rates than men in high-income countries, driven by factors such as hormonal differences, cultural norms and lifestyle behaviors, whereas in low- and middle-income nations, the patterns are more heterogeneous. Urban populations generally exhibit elevated obesity prevalence due to sedentary lifestyles, increased access to processed foods and environmental factors conducive to weight gain, while rural areas are experiencing a rising trend linked to dietary westernization and mechanization of labor. These disparities underscore the importance of understanding intersectional influences to develop targeted, effective public health interventions aimed at reducing obesity-related health burdens worldwide.
2. Global Trends and Gender Disparities in Obesity Prevalence
Obesity has ascended to the status of a global health crisis, characterized by an alarming increase in prevalence across diverse populations worldwide. Recognized as a complex and multifactorial disease, obesity results from intricate interactions amongst genetic, hormonal, behavioral, environmental and socio-cultural factors. Its epidemiology exhibits pronounced disparities across different regions, income levels and demographic groups, with gender playing a significant role in shaping these patterns. Understanding the global trends and gender-specific differences in obesity prevalence is essential for informing targeted prevention strategies, optimizing clinical management and guiding public health policies aimed at reducing the burden of obesity and its associated comorbidities such as type 2 diabetes mellitus (T2DM), cardiovascular disease (CVD) and certain cancers7,24.
This comprehensive analysis aims to delineate the global epidemiological patterns of obesity with an emphasis on gender disparities. It integrates current data, epidemiological trends, biological underpinnings and sociocultural influences, supported by quantitative figures and mathematical models where appropriate, to provide a nuanced understanding of these disparities. It also discusses how these factors intersect with environmental and lifestyle determinants across different regions, highlighting the importance of context-specific, culturally sensitive interventions.
2.1.Epidemiological Overview of Global Obesity Trends
2.1.1. Prevalence patterns across income levels and regions:
Globally, obesity prevalence has more than doubled over the past four decades. According to the World Health Organization, in 2016, more than 650 million adults (13% of the world population aged ≥18 years) were classified as obese (body mass index [BMI] ≥30 kg/m²). The distribution of obesity is markedly heterogeneous, with high-income countries demonstrating the highest prevalence rates, yet low- and middle-income countries (LMICs) experiencing the most rapid increases25. In high- income nations such as the United States, Australia and many European countries, obesity prevalence among adults exceeds 35-40%. For instance, data from the CDC (2021) indicate that 42.4% of U.S. adults are obese, with a higher prevalence among women (44%) compared to men (37%). Similarly, European countries like the United Kingdom report adult obesity rates of approximately 27-35%, with women slightly surpassing men in prevalence26.
In LMICs, obesity has historically been less prevalent but is now rising rapidly, especially in urban areas. In South Asia, for example, urban Indian women exhibit obesity prevalence of approximately 20-25%, with rural populations traditionally showing lower rates (10%), though these are increasing due to dietary transitions and urban migration. Sub-Saharan Africa and parts of Southeast Asia show emerging obesity trends, often with a higher prevalence among women27.
2.2. Gender disparities in obesity prevalence: Biological and sociocultural perspectives
Quantitatively, the waist-to-hip ratio (WHR) serves as a marker of fat distribution. Women usually present with WHR below 0.85, indicating peripheral fat accumulation, whereas men often have WHR exceeding 0.90, reflecting central adiposity. These distinctions have substantial implications for disease risk stratification30. Hormonal influences are pivotal. Estrogen promotes subcutaneous fat deposition and exerts vasoprotective effects, while testosterone favors visceral fat accumulation. Postmenopausal women experience a decline in estrogen levels, leading to increased visceral adiposity and heightened metabolic risk. Conversely, men with higher testosterone levels tend to develop more visceral fat, amplifying the risk of metabolic syndrome31.
2.2.2. Sociocultural and behavioral factors:
Sociocultural norms and behavioral patterns profoundly influence gender disparities in obesity. Women in high-income countries often face societal pressures related to body image, which can influence dieting behaviors, physical activity and healthcare engagement32. Physical activity levels are generally lower among women, especially in urban settings, due to safety concerns, cultural restrictions or caregiving responsibilities. For example, global data from the World Health Organization1 indicate that women are approximately 20-30% less likely to meet physical activity guidelines compared to men.
Dietary behaviors are also gender-specific. Women may be more prone to emotional eating or dieting, leading to cycles of weight fluctuation. In LMICs, women often have limited access to nutritious foods due to socio-economic constraints, leading to both undernutrition and obesity a paradoxical coexistence33. Psychosocial stress and mental health factors contribute variably. Urban women report higher levels of stress, which can trigger cortisol-mediated visceral fat accumulation. Emotional eating, driven by stress or depression, further exacerbates adiposity among women34.
2.3. Regional disparities in gender-specific obesity trends
2.3.2. Low- and middle-income countries: In LMICs, the gender gap in obesity prevalence is often narrower, with some regions reporting higher rates among men. In urban South Asia, male obesity rates reach approximately 15-20%, with women at similar or slightly higher levels. Cultural factors such as gendered food allocation, physical activity patterns and reproductive health influence these trends37. In sub-Saharan Africa, rural men engaged in mechanized agriculture exhibit obesity rates of 8-12%, whereas women, often involved in household activities, display lower rates (~10%), although urbanization and dietary westernization are increasing these figures rapidly38.
Mathematical modeling of these patterns indicates that the odds ratio (OR) for obesity associated with female gender in high-income countries exceeds 1.5, while in LMICs ors vary between 1.0 and 1.3, depending on urbanization level39.
2.4. The impact of urbanization on gender disparities
Urban environments significantly influence obesity trends, with profound gender-specific implications. Urbanization involves lifestyle changes such as decreased physical activity due to mechanized transport and sedentary occupations, increased access to processed foods and altered social norms31. Urban women often experience higher rates of central obesity, partly due to reduced physical activity and increased caloric intake. Data from developing countries reveal that urban women’s prevalence of abdominal obesity (waist circumference ≥88 cm) can reach 45-50%, compared to 20-25% in rural women40.
Urban men also experience increased obesity rates, often driven by dietary westernization and reduced occupational physical activity. The urban-rural obesity prevalence ratio (PR) in men is typically around 1.2-1.4, whereas for women, it can be as high as 2.0, signifying a more pronounced urban effect on women24.
2.5. Biological and physiological variations influencing obesity
2.5.2. Hormonal regulation: Menopause in women results in decreased estrogen, leading to increased visceral fat and a shift toward a male-pattern fat distribution, thereby elevating cardiometabolic risk. Conversely, testosterone deficiency in men is linked to increased adiposity43. Emerging evidence suggests that environmental endocrine-disrupting chemicals (EDCs), prevalent in urban pollution, interfere with hormonal pathways, further influencing adipogenesis and fat distribution in a gender- specific manner. For example, phthalates and bisphenol A (BPA) can mimic estrogen or interfere with androgen signaling, affecting fat accumulation patterns44.
2.6. Sociocultural and lifestyle determinants
2.6.1. Dietary patterns and food environment : Dietary behaviors are deeply rooted in cultural norms and economic conditions. In high-income countries, women’s diets often include higher carbohydrate and sugar intake, influenced by marketing and social norms. Fast food and processed snacks are prevalent, contributing to positive energy balance45. In LMICs, rapid urbanization and globalization have introduced Western dietary patterns characterized by energy-dense, nutrient-poor foods. Women’s increased participation in the workforce and urban migration have led to higher caloric intake, often exceeding energy expenditure46.
2.6.2. Physical activity and sedentary behavior: Physical activity levels are inversely related to urbanization. Data from the WHO indicate that only 25-30% of women in urban settings meet recommended physical activity levels, compared to 45-50% of men. Cultural restrictions, safety concerns and caregiving responsibilities limit women’s mobility and recreational activity47. In rural areas, physical activity is primarily occupational, with agricultural work providing substantial energy expenditure. However, mechanization and urban migration are reducing these physical activity levels, especially among women48,49.
2.6.3. Psychosocial factors and mental health: Urban stressors, including social isolation, noise pollution and economic pressures, influence eating behaviors and metabolic health. Women in urban environments report higher levels of stress-related eating, which involves increased intake of high- calorie, palatable foods. Cortisol production during chronic stress enhances visceral fat deposition, further exacerbating cardio metabolic risk50.
2.7. Quantitative data and mathematical modeling of disparities
Table 1: Regional Disparities in Obesity Prevalence and Gender Odds Ratios.
|
Region |
Female Obesity
Prevalence (%) |
Male Obesity Prevalence (%) |
Gender Odds
Ratio (OR) |
References |
|
North America (USA) |
42.4 |
36.2 |
1.54 |
CDC,
2021 |
|
Europe (EU countries) |
40 |
35 |
1.33 |
Eurostat,
2022 |
|
South Asia (urban India, Pakistan) |
20-25 |
15-20 |
1.25-1.30 |
Haque
et al., 2021 |
|
Sub-Saharan Africa |
15-Oct |
12-Aug |
1.2-1.25 |
WHO Regional Reports |
|
Middle
East |
45-50 |
40-45 |
1.2-1.3 |
WHO Eastern
Mediterranean Data |
The odds ratios
(ORs) indicate that women are significantly more likely to be obese than men in
high-income countries, whereas in LMICs,
the ORs are lower but still indicate
a gender disparity favoring
higher female prevalence (Table 1,
Figure 1&2)1,22,23,51,52.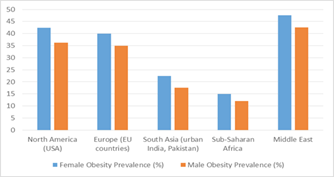
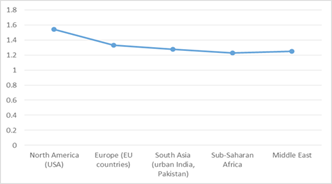
Figure 1: Male versus
Female Obesity Prevalence.
2.7.2. Temporal trends: Longitudinal data
reveal that obesity prevalence has increased by approximately 2-3% annually over the past two
decades in most regions, especially among women in urban settings. For example,
in India, female obesity increased from 10% in 1990 to 22% in 2015,
representing a doubling within 25 years53. The global landscape of obesity
is characterized by distinctive gender-specific patterns shaped by biological, behavioral and sociocultural factors. Women in
high-income countries exhibit higher prevalence rates driven by hormonal influences, sociocultural
expectations and lifestyle factors, whereas in LMICs, rising obesity among both
genders reflects rapid urbanization, dietary westernization and mechanization
of labor54.
Figure 2: Gender Odds Ratio (OR) across different
Regions.
Mathematical
modeling and epidemiological data underscore
the dynamic and evolving nature
of these disparities, necessitating culturally sensitive, gender-responsive and region-
specific interventions. Recognizing the complex interplay of these factors is
crucial for effective public health strategies aimed at curbing the obesity
epidemic and its associated health burdens globally55.
3. Biological and Physiological Differences in Obesity
The
pathophysiology of obesity is intricately linked to biological and
physiological differences that vary significantly between genders. These
differences influence fat distribution, adipocyte function, hormonal regulation and metabolic pathways,
ultimately affecting the risk profiles
for obesity-related diseases. Recognizing these
gender-specific biological factors
is essential for
understanding disparities in obesity prevalence, phenotypic expression and
associated metabolic complications. This section
provides a comprehensive exploration of the biological and physiological distinctions, emphasizing adipose tissue distribution, hormonal
regulation and environmental influences on these mechanisms (Table 2)56.
Table 2: Summary of Key Biological
and Physiological Differences in Obesity by Gender.
|
Aspect |
Women |
Men |
Notes/Implications |
|
Body Fat Percentage |
25-30% at similar BMI |
20-25% at similar BMI |
Women have
higher total fat
percentages |
|
Fat Distribution |
Gynoid (peripheral:
hips, thighs) |
Android (central:
visceral fat) |
Different risk
profiles |
|
Waist-to-Hip Ratio (WHR) |
<0.85 |
>0.90 |
Marker of fat
distribution and risk |
|
Hormonal Influence |
Estrogen promotes subcutaneous fat |
Testosterone promotes visceral fat |
Postmenopause increases visceral fat in women |
|
Response
to Environmental Endocrine Disruptors |
Sensitive to BPA, phthalates |
Less sensitive but affected by hormonal changes |
Impact on fat
accumulation and distribution |
Men predominantly accrue central or android adiposity, characterized by visceral fat accumulation within the abdominal cavity. Visceral adipose tissue (VAT) is metabolically active, secreting pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), which promote insulin resistance, dyslipidemia and atherosclerosis. Quantitatively, imaging studies utilizing magnetic resonance imaging (MRI) or computed tomography (CT) have demonstrated that men possess approximately 30-50% more visceral fat area than women at comparable BMI levels, translating into a higher cardiometabolic risk profile. Studies report that men often exhibit ratios exceeding 0.5, indicating dominant visceral fat, whereas women typically maintain ratios below 0.358.
3.1.2. Hormonal regulation of fat depot specificity: The differential fat distribution pattern is primarily governed by sex hormones, notably estrogen and testosterone, which modulate adipocyte differentiation, lipolysis and storage capacity in specific tissues. Estrogen enhances the proliferation and activity of preadipocytes in subcutaneous fat depots, favoring peripheral fat storage59. Estrogen’s influence involves activating estrogen receptors (ERα and ERβ) in adipocytes, leading to increased expression of genes conducive to lipid storage in subcutaneous tissues. This effect is particularly evident during reproductive years, where estrogen levels are high, correlating with gynoid fat predominance. Postmenopause, with declining estrogen, women often experience a shift toward central adiposity, increasing their risk for metabolic syndrome60.
Testosterone in men promotes visceral fat accumulation by stimulating adipocyte differentiation within the visceral region. Testosterone inhibits subcutaneous fat proliferation, favoring a visceral distribution. It also influences lipolytic activity, with higher testosterone levels correlating with lower overall fat mass but increased visceral adiposity61. Quantitative data suggest that estrogen decreases lipolysis in peripheral fat, with a reduction of approximately 10-15% in lipolytic activity compared to premenopausal levels, contributing to fat accrual in the lower body. Conversely androgen excess in women (e.g., polycystic ovary syndrome) is associated with increased visceral fat and metabolic disturbances62.
3.2. Environmental and lifestyle influences on fat distribution
Research indicates that stress-induced cortisol elevation in urban environments promotes visceral adiposity. Cortisol, a glucocorticoid hormone, enhances lipogenesis in visceral fat depots via activation of the glucocorticoid receptor (GR), increasing VAT volume by approximately 20-30% in chronically stressed individuals65. In rural populations engaged in physically demanding activities, fat distribution tends to favor peripheral depots, with a lower V/S ratio and decreased risk of metabolic syndrome. However, mechanization and dietary westernization are leading to shifts toward central adiposity even in these populations66.
3.3. Hormonal regulation and metabolic variability
Postmenopause, with estrogen levels declining from an average of 200-300 pg/mL to below 50 pg/mL, women experience increased visceral fat accumulation up to 30-40% of total adiposity elevating their risk for metabolic syndrome and cardiovascular disease69. Testosterone, prevalent in males at approximately 300-1000 ng/dL, influences adiposity by enhancing visceral fat deposition. Testosterone deficiency in men (e.g., hypogonadism) correlates with increased visceral fat, with visceral fat mass increasing by approximately 15-20% per 10% decrease in circulating testosterone70.
3.3.2. Endocrine disruptors and environmental chemicals: Environmental exposures, especially in urban pollution, include endocrine-disrupting chemicals (EDCs) such as bisphenol A (BPA), phthalates and polychlorinated biphenyls (PCBs). These agents can interfere with hormonal signaling, affecting adipogenesis, fat distribution and metabolic health71. BPA mimics estrogen, binding to estrogen receptors and altering gene expression related to adipocyte differentiation. Studies demonstrate that BPA exposure correlates with a 12-20% increase in visceral fat area among exposed populations, with stronger effects observed in women with hormonal sensitivities72.
3.4. Environmental impact on biological mechanisms
Table 3: Behavioral and Socioeconomic Determinants.
with hormonal
susceptibilities, such as postmenopausal women or those with hormonal
imbalances, due to their heightened sensitivity to environmental perturbations9.
Chronic low-grade
inflammation driven by environmental pollutants can increase the adipose tissue macrophage infiltration
by 40-60%, further promoting insulin resistance and metabolic
dysfunction, especially in visceral depots74.
4. Behavioral and Socioeconomic Determinants of Obesity
Obesity is a
multifaceted condition influenced not only by biological and environmental factors but also significantly shaped
by behavioral and socioeconomic determinants. These factors modulate
individual choices, access to resources and cultural norms, thereby affecting
the prevalence and distribution of obesity across different
populations. Understanding the intricate
interplay of dietary behaviors, physical activity patterns and socioeconomic
status (SES) is crucial for developing effective, targeted interventions. This section explores these determinants
in detail, emphasizing their regional variations, underlying mechanisms and
implications for public health (Table 3)75.
|
Determinant |
Urban |
Rural |
Impact/Notes |
|
Food
Environment |
Processed, calorie-dense foods abundant |
Traditional, whole
foods |
Urbanization increases obesogenic diet |
|
Physical Activity |
Reduced,
sedentary lifestyles |
High
occupational activity |
Mechanization reduces rural activity |
|
Socioeconomic
Status |
Low SES linked to poor diet,
less activity |
Similar
trends |
Food
deserts, limited resources |
|
Healthcare Access |
Better
in urban; limited in rural |
Scarce
facilities |
Affects diagnosis and management |
|
Cultural
Norms |
Shift towards slimness in urban
areas |
Body size as prosperity in some cultures |
Influences behaviors |
Fast-food marketing strategies often focus on women, especially those balancing work and family responsibilities, emphasizing convenience and affordability. These marketing campaigns leverage social media, celebrity endorsements and promotional discounts, shaping dietary choices that favor calorie-dense foods. The cumulative effect contributes to positive energy balance, with some studies estimating that urban residents consume an excess of 300-500 kcal/day compared to rural residents, significantly increasing obesity risk78. Rural populations traditionally rely on diets rich in whole grains, legumes, vegetables and lean meats components associated with lower caloric density and higher satiety. Globalization and market integration have led to the infiltration of processed foods into rural markets. Market surveys in Southeast Asia indicate that processed snack consumption in rural areas has increased by 20-25% over the last decade, contributing to shifts in dietary patterns. Rural food insecurity, paradoxically, can coexist with obesity a phenomenon termed the “hunger-obesity paradox” where reliance on inexpensive, calorie-rich, nutrient- poor foods leads to weight gain despite insufficient intake of micronutrients79.
4.1.2. Dietary quality and nutritional transition: The quality of diet is a critical determinant of obesity development. Urban diets often involve higher consumption of refined grains, sugar- laden beverages and processed snacks, whereas traditional diets emphasize unprocessed, fiber-rich foods. The shift towards Westernized dietary patterns is associated with increased intake of saturated fats and added sugars, which are linked to increased visceral adiposity and insulin resistance. For example, an increase in added sugar intake (from 10% to 20% of total calories) has been associated with a 15-20% increased risk of developing obesity-related metabolic disorders80.
4.2. Physical activity and sedentary lifestyle
4.2.2. Rural physical activity and its decline: In rural settings, physical activity has traditionally been high due to manual labor associated with agriculture, animal husbandry and household chores. This lifestyle contributes to higher energy expenditure, acting as a protective factor against obesity. Nonetheless, mechanization of farming tools, motorized transport and technological advances have resulted in a decline in physical activity levels among rural populations83. Recent surveys illustrate that rural adults’ physical activity levels have decreased by approximately 10-15% over the past decade, leading to rising obesity prevalence. Rural men in Southeast Asia now report average daily physical activity levels that are 20-25% lower than in previous generations, correlating with increased BMI and waist circumference79.
4.3. Socioeconomic factors and access to resources
In rural areas, socioeconomic disadvantages hinder access to healthcare, health education and healthy foods. Limited infrastructure results in fewer opportunities for physical activity, such as gyms or parks and healthcare services for screening and managing obesity-related conditions. Cultural perceptions of body weight also influence health-seeking behaviors; in some rural communities, higher body weight may be associated with prosperity and health, reducing motivation for weight management86.
4.3.2. Healthcare access and health literacy: Limited access to healthcare services hampers early detection, counseling and management of obesity. In rural settings, healthcare facilities are often sparse, with patient-to-provider ratios exceeding 1,000:1 in some regions. This gap delays diagnosis and intervention, allowing obesity and its complications to progress unchecked87. Health literacy a person’s capacity to obtain, process and understand basic health information is often lower in socioeconomically disadvantaged populations. Limited health literacy diminishes the likelihood of adopting healthy behaviors, such as balanced diets and regular physical activity or seeking timely medical advice88.
5. Cultural and Psychosocial Influences on Obesity
Cultural and psychosocial factors are fundamental determinants of obesity, shaping individual behaviors, perceptions and responses to environmental stimuli. These influences often operate through complex pathways involving societal norms, mental health and behavioral coping mechanisms. Understanding these facets is essential for designing culturally sensitive and psychologically informed interventions aimed at preventing and managing obesity across diverse populations (Table 4)33.
Table 4: Cultural and Psychosocial Influences.
|
Aspect |
Key Points |
|
Body
Image Norms |
Higher BMI seen as prosperity in some cultures; slimness valued
in others |
|
Cultural
Barriers |
Restrictions on women’s activity; traditional diets |
|
Stress &
Mental Health |
Chronic urban
stress promotes visceral fat via cortisol |
|
Social
Perceptions |
Stigma or acceptance varies
by culture |
Urbanization often shifts these norms toward valuing slimness, especially among women, driven by Western beauty standards propagated through media, fashion and celebrity culture. For instance, in urban China and India, over 70% of women aged 20-35 associate slenderness with attractiveness and success. These shifting norms influence gender-specific behaviors, with urban women engaging more actively in weight control practices, including dieting and exercise, driven by societal pressures and personal aspirations90.
5.1.2. Cultural barriers and facilitators: Cultural beliefs may also influence attitudes toward physical activity, dietary choices and health-seeking behaviors. For example, some cultures regard physical activity as unnecessary or even inappropriate for women, especially in conservative societies. Such norms reduce participation in physical activity; in certain Middle Eastern communities, less than 20% of women report engaging in regular exercise, primarily due to cultural restrictions91. Some cultures incorporate traditional food practices that promote balanced nutrition, such as the Mediterranean diet, which is associated with lower obesity rates. Recognizing these cultural strengths provides opportunities to reinforce healthy behaviors within culturally relevant frameworks92.
5.2. Psychosocial stress and mental health
1.5 times more prone to emotional overeating, with cortisol levels positively correlating (r=0.65, p<0.01) with increased waist circumference94.
5.2.2. Gender differences in stress responses: Gender differences in stress response and coping strategies influence obesity trajectories. Women tend to respond to stress through emotional overeating, a behavior driven by hormonal mechanisms involving cortisol, serotonin and dopamine pathways. Neuroimaging studies reveal that women with high stress levels show increased activation of the limbic system, associated with emotional regulation and reward processing, which correlates with increased intake of palatable, energy-dense foods95. Men may respond to stress via different behavioral pathways, such as increased alcohol consumption or reduced physical activity. Quantitative data suggest that men under stress are 35% more likely to engage in sedentary behaviors, including screen time, which contributes to decreased energy expenditure7.
5.2.3. Mental health and obesity: Psychosocial factors such as depression, anxiety and low self-esteem are both causes and consequences of obesity, creating a bidirectional relationship. Epidemiological data indicate that individuals with clinical depression have a 1.2-1.8 times higher risk of developing obesity. Conversely, obesity can exacerbate mental health issues through social stigmatization and reduced quality of life, forming a vicious cycle that complicates management96.
5.3. Impact of residential area on obesity management and treatment
5.3.2. Pharmacological and surgical interventions: Pharmacotherapy for obesity, including agents like liraglutide and naltrexone/bupropion, operates through mechanisms such as appetite suppression, delayed gastric emptying and modulation of reward pathways. These drugs’ pharmacokinetics and pharmacodynamics may be influenced by adipose tissue distribution and metabolic state, which are affected by gender and environment99. Lipophilic drugs like liraglutide, which are extensively distributed into adipose tissue, may exhibit variable efficacy depending on body fat composition. For instance, individuals with higher visceral fat may experience different drug absorption and metabolism profiles. While current guidelines do not specify gender-specific dosing, emerging evidence suggests that personalized dosing strategies considering individual biological and environmental factors could optimize outcomes100.
Bariatric surgery, such as Roux-en-Y gastric bypass and sleeve gastrectomy, requires thorough preoperative assessment that accounts for gender-specific anatomical and hormonal considerations. Postoperative care must be tailored to address differences in weight loss trajectories and nutritional needs, which vary by gender and residential context due to disparities in healthcare access101.
5.4. Lifestyle interventions and community-based programs
5.4.2. Rural settings: In rural areas, formal programs may be scarce or nonexistent. Nonetheless, community-based, culturally tailored initiatives can effectively promote healthy behaviors. These programs leverage local social networks, traditional practices and community leaders to foster engagement. For example, in rural India, village health volunteers have organized group physical activities and nutritional education sessions, resulting in a 15-20% reduction in obesity prevalence over two years103.
5.4.3. Tailoring interventions: Culturally and environmentally tailored interventions considering local dietary patterns, physical activity opportunities and social norms are more likely to succeed. Incorporating local food practices, respecting cultural perceptions of body image and addressing socioeconomic barriers can enhance adherence and sustainability104. Cultural and psychosocial influences are pivotal in shaping obesity risks and management strategies. Societal norms regarding body image, mental health and stress responses significantly modulate behaviors that contribute to or mitigate obesity. Recognizing the diversity of these influences across different residential settings and cultural contexts is essential for developing effective, culturally sensitive interventions. Tailoring approaches to address these psychosocial factors holds promise for improving obesity outcomes globally105.
6. Modern Perspectives and Future Directions in Obesity Management
The landscape of obesity management is rapidly evolving, driven by advances in biomedical science, technology and public health policy. The integration of personalized medicine, digital health tools and multisectoral policy initiatives holds promise for addressing the complex interplay of genetic, behavioral, environmental and social determinants of obesity. This section provides an in-depth analysis of emerging strategies and future directions, emphasizing the importance of tailoring interventions to individual and population-level needs and promoting sustainable, equitable health outcomes106.
6.1. Personalized and precision medicine in obesity
6.1.2. Gender-specific and environmentally informed interventions: Gender differences in hormonal profiles, adipose tissue distribution and gene expression impact both the pathophysiology of obesity and treatment response. For example, women tend to accumulate subcutaneous fat, which is metabolically less harmful than visceral fat predominant in men. Pharmacogenomics studies indicate that drug efficacy and side effect profiles may differ by gender; for instance, lipophilic drugs such as liraglutide may have different pharmacokinetics in individuals with varying fat distribution. Incorporating genetic and hormonal data into clinical decision- making fosters a move toward precision medicine, where interventions be they pharmacological, dietary or behavioral are customized109. Pharmacotherapy efficacy varies according to individual biological parameters. For instance, individuals with higher visceral adiposity may respond differently to GLP-1 receptor agonists like liraglutide, with some studies reporting up to 15-20% greater weight loss in this subgroup. Similarly, pharmacokinetic modeling suggests that dosing adjustments based on body composition could enhance outcomes. Bariatric surgery, such as sleeve gastrectomy or gastric bypass, requires preoperative risk stratification considering gender, age, BMI, comorbidities and psychological factors. Postoperative management must be personalized, including nutritional supplementation and behavioral support, to optimize weight loss and minimize complications110.
6.2. Digital health and telemedicine: Transforming obesity care
Data indicate that adherence to digital interventions can increase by 30-50%, with some programs demonstrating an average weight loss of 5-10% over six months. These tools are particularly valuable for women and rural residents who face barriers to traditional healthcare access, thereby promoting gender-sensitive and culturally appropriate care.
6.2.2. Personalized digital interventions: Artificial intelligence (AI) and machine learning algorithms can analyze large datasets to customize behavioral interventions. For example, AI-driven apps can identify patterns in individual data such as stress levels, sleep quality and activity and adapt recommendations accordingly. Mathematical models can predict the likelihood of adherence or relapse, enabling clinicians to intervene proactively.
Virtual coaching and tele-supervision can enhance motivation and accountability, addressing psychosocial barriers identified previously. The integration of digital health with electronic health records (EHRs) facilitates comprehensive, longitudinal management of obesity, encompassing biological, behavioral and social data.
6.3. Policy and public health interventions: Creating supportive environments
Regulations on the marketing of unhealthy foods and beverages, especially targeting children and adolescents, are critical. Evidence suggests that restricting advertising of high- sugar snacks and SSBs can reduce consumption by 15-25% among youth.
In rural settings, policies should focus on improving access to nutritious foods, such as subsidizing fruits and vegetables and establishing community-based programs that leverage local resources. For example, school-based nutrition programs can reduce childhood obesity prevalence by 10-15%, especially when culturally tailored.
6.3.2. Gender-sensitive policy initiatives: Gender disparities in healthcare access and resource allocation demand targeted policies. Ensuring equitable access to weight management clinics, counseling services and educational programs is vital. For instance, implementing community outreach that addresses cultural norms and gender roles can increase participation among women, who often face social constraints.
The future of obesity management rests on integrating cutting-edge scientific insights with innovative technological solutions and comprehensive policy measures. Emphasizing personalized and precision approaches allows for interventions that consider individual genetic, hormonal, behavioral and environmental factors, thereby enhancing efficacy and sustainability. Digital health tools, including telemedicine and AI-driven applications, have the potential to bridge healthcare disparities, particularly in rural and underserved populations and facilitate gender-sensitive, culturally appropriate care. Simultaneously, multisectoral policies that promote healthy environments such as active transportation infrastructure, healthy food accessibility and regulatory measures are essential for creating systemic change.
Research must continue to explore the complex interactions among biological, environmental and psychosocial determinants, prioritizing longitudinal, multi-ethnic studies that can inform tailored interventions. Mathematical modeling and data analytics will play a pivotal role in predicting outcomes, optimizing resource allocation and personalizing care. Achieving equitable and sustainable reductions in obesity prevalence demands a collaborative effort among clinicians, policymakers, researchers and communities. By embracing innovative strategies grounded in scientific evidence and cultural sensitivity, the global health community can make significant strides toward reversing the obesity epidemic and improving health outcomes for diverse populations worldwide.
7. Conclusion
This comprehensive review underscores the multifaceted nature of obesity, highlighting the critical influence of gender disparities and environmental context on its epidemiology, pathophysiology and management. Biological differences, including adipose tissue distribution and hormonal regulation particularly the roles of estrogen and testosterone play pivotal roles in shaping distinct obesity phenotypes and associated cardio metabolic risks across sexes. Sociocultural norms, lifestyle behaviors and environmental exposures further modulate these biological predispositions, contributing to regional and urban- rural variations in prevalence. Recognizing the intersectionality of biological, behavioral and environmental determinants is essential for developing precision medicine approaches, enabling tailored interventions that optimize therapeutic efficacy and mitigate health disparities.
Looking forward, integrating advancements in genomics, metabolomics and digital health technologies offers promising avenues for personalized, gender-sensitive obesity management. Precision medicine, coupled with culturally competent behavioral and community-based strategies, can enhance adherence and outcomes. Multisectoral policy initiatives aimed at creating health-promoting environments such as active urban infrastructure, regulation of obesogenic marketing and equitable access to nutritious foods are crucial for sustainable population-level impact. Addressing the complex interplay of biological, psychosocial and socio-economic factors through innovative, evidence-based and culturally adapted interventions is imperative to stem the global obesity epidemic and reduce its associated morbidity and mortality.
8. Conflict of Interest Statement
Authors have no conflict of interest.
9. Acknowledgments
The authors like to express their sincere gratitude to all the individuals who participated in this study, as well as the organizations and institutions that provided their support and resources. The authors acknowledge the contributions of their colleagues and peers who have inspired and guided them throughout this research journey. The authors extend their appreciation to the reviewers for their valuable feedback, which has significantly improved the quality and clarity of this work.
10. Grant Support & Financial Disclosures
11. Funding Statement
12. Informed Consent / Patient Consent
13. Data availability statement
14. Authors Contribution
15. Other Journal Specific Statements
16. Abbreviations
BMI : Body Mass Index
WHO : World Health Organization
T2DM : Type 2 Diabetes Mellitus
CVD : Cardiovascular Diseases
VAT : Visceral Adipose Tissue
WHR : Waist-to-Hip Ratio
EDCs : Endocrine Disrupting Chemicals
MRI : Magnetic Resonance Imaging
CT : Computed Tomography
HDL : High-Density Lipoprotein
LDL : Low-Density Lipoprotein
GWAS : Genome-Wide Association Studies mHealth : Mobile Health
AI : Artificial Intelligence
LMS : Lambda Mu Sigma
QR : Quantile Regression
BSSI : Body Shape and Size Index
BSA : Body Surface Area
PI : Ponderal Index
CDC : Centers for Disease Control
17. References
1. World Health Organization. Obese and overweight, 2024.
2. Rathomi HS. Aligning Behaviour with Physiology: The Potential of Time-Restricted Eating for Rural Obesity Control, 2024.
3. Spinelli A, Buoncristiano M, Nardone P, et al. Thinness, overweight and obesity in 6 to 9-year-old children from 36 countries: The World Health Organization European Childhood Obesity Surveillance Initiative-COSI 2015-2017. Obesity Reviews, 2021;22: 13214.
4. Yu Y, Liang Z, Zhou Q, et al. Decomposition and Comparative Analysis of Urban-Rural Disparities in eHealth Literacy Among Chinese University Students: Cross-Sectional Study. Journal of Medical Internet Research, 2025;27: 63671.
5. Khanna D, Peltzer C, Kahar P, et al. Body mass index (BMI): a screening tool analysis. Cureus, 2022;14(2).
6. Rabadán-Chávez G, de la Garza RID, Jacobo-Velázquez DA. (2023). White adipose tissue: Distribution, molecular insights of impaired expandability and its implication in fatty liver disease. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 2023;1869(8): 166853.
7. Cooper AJ, Gupta SR, Moustafa AF, et al. Sex/gender differences in obesity prevalence, comorbidities and treatment. Current obesity reports, 2021: 1-9.
8. Lamas B, Evariste L, Houdeau E. Dysregulation along the gut microbiota-immune system axis after oral exposure to titanium dioxide nanoparticles: A possible environmental factor promoting obesity- related metabolic disorders. Environmental Pollution, 2023;330: 121795.
9. Sørensen TI, Martinez AR, Jørgensen TSH. Epidemiology of obesity. In From obesity to diabetes. Cham: Springer International Publishing, 2022: 3-27.
10. Hussain WG, Shehzad F, Ahmad R, et al. Establishing growth charts for proposed body shape and size index of the Pakistani population, using quantile regression approach. SAGE Open Medicine, 2021;9: 20503121211036135.
11. Hussain WG, Akbar A, Shehzad F. Gaussian (Z-Score) percentiles of ponderal index (pi) in Pakistani children and adults: a quantitative approach to human growth analysis. MOJ Gerontology & Geriatrics, 2024;9(3).
12. Hussain WG, Shehzad F, Akbar A. Comparison of Quantile Regression and Gaussian (Z-scores) Percentiles to BSA in Growth Charts with a Pakistani Population. Gerontology and Geriatric Medicine, 2024;10: 23337214241273189.
13. Hussain WG, Shehzad F, Akbar A. Examining the Relationship Between Obesity and Income Distribution Using Body Mass Index (BMI) and Body Shape and Size Index (BSSI): A Case Study of Pakistan. Gerontology and Geriatric Medicine, 2024;10: 23337214241288795.
14. Shehzad F, Hussain WG, Akbar A. A Comparative Evaluation of Quantile Regression Percentiles, Gaussian Percentiles and Raw Percentiles to Body Shape and Size Index (BSSI) in Growth Charts: A Case Study of Pakistan. Open Access Journal of Surgery, 2024;16(2).
15. Shehzad F, Hussain WG, Akbar A. Quantifying Human Growth Using Quantile Regression Percentiles for Body Surface Area (BSA): A Case Study of Pakistan. Curr Res Diabetes Obes J, 2024;18(1).
16. Qureshi MA, Hussain WG. Assessment of Body Shape and Size Index (BSSI) in Relation to Gaussian Percentiles Growth Charts: A Gender- Specific Analysis across Age Cohorts in South Punjab, Pakistan. Curr Res Diabetes Obes J, 2025;17(5).
17. Hussain WG. A New Standard for Mortality Prediction: The Body Shape and Size Index (BSSI) Emerges as a Superior Alternative. Open Access Journal of Surgery, 2025;16(3).
18. Hussain WG. From Data to Insights: Analyzing Body Shape and Size Index (BSSI) Trends in Pakistani Population through Growth Charts of Gaussian (Z-Scores) Percentiles. J Community Med Public Health 2025;9(2): 516.
19.Hussain WG, Qureshi MA. A comprehensive review of Body Shape and Size Index (BSSI) in relation to obesity: Insights from recent studies in Pakistan. J of Gyne Obste & Mother Health, 2025;3(2): 1-10.
20. Aisha R, Akbar A, Hussain WG. Effect of pre-delivery body mass index and gestational weight gain on infant weight. International Journal of Biosciences, 2019;15(5): 218-226.
21. Atique H, Hussain WG. Quantile Regression Analysis for Examining Gender Variations in Obesity Prevalence in Pakistan Using Body Surface Area Percentiles in Growth Charts. Curr Res Diabetes Obes J, 2025;17(5).
22. Centers for Disease Control and Prevention, & National Center for Health Statistics. Underlying Case of Death 1999-2019 on CDC WONDER Online Database, released in 2020. Data are from the Multiple Cause of Death Files as compiled from data provided by the 57 vital statistics jurisdictions through the Vital Statistics Cooperative Program, 2021.
23. Eurostat A. Characteristics of the Spanish Exporting Company, 2021.
24. Wang Y, Huang Y, Al Jawish MM, et al. Rising obesity-associated mortality in men: Exploration of gender disparity from the global burden of disease study, 1990–2019. Journal of general internal medicine, 2024: 1-10.
25. Kang S, Kang M, Lim H. Global and regional patterns in noncommunicable diseases and dietary factors across national income levels. Nutrients, 2021;13(10): 3595.
26. Mathieu◻Bolh N. The elusive link between income and obesity. Journal of Economic Surveys, 2022;36(4): 935-968.
27. Hemmingsson E, Ekblom Ö, Kallings LV, et al. Prevalence and time trends of overweight, obesity and severe obesity in 447,925 Swedish adults, 1995-2017. Scandinavian journal of public health, 2021;49(4): 377-383.
28. Koceva A, Herman R, Janez A, et al. Sex-and gender-related differences in obesity: from pathophysiological mechanisms to clinical implications. International Journal of Molecular Sciences, 2024;25(13): 7342.
29. Muscogiuri G, Verde L, Vetrani C, et al. Obesity: a gender-view. Journal of endocrinological investigation, 2024;47(2): 299-306.
30. Culbert KM, Sisk CL, Klump KL. A narrative review of sex differences in eating disorders: is there a biological basis? Clinical therapeutics, 2021;43(1): 95-111.
31. Kapoor N, Arora S, Kalra S. Gender disparities in people living with obesity-an unchartered territory. Journal of mid-life health, 2021;12(2): 103-107.
32. Min J, Goodale H, Xue H, et al. Racial-ethnic disparities in obesity and biological, behavioral and sociocultural influences in the United States: a systematic review. Advances in Nutrition, 2021;12(4): 1137- 1148.
33. Agustina R, Susiloretni KA, Shankar AH, et al. Psychosocial, eating behavior and lifestyle factors influencing overweight and obesity in adolescents. Food and Nutrition Bulletin, 2021;42(1): 72-91.
34. Medina FX, Solé-Sedeno JM, Bach-Faig A, et al. Obesity, Mediterranean diet and public health: A vision of obesity in the Mediterranean context from a sociocultural perspective. International journal of environmental research and public health, 2021;18(7): 3715.
35. Segal AB, Huerta MC, Aurino E, et al. The impact of childhood obesity on human capital in high-income countries: a systematic review. Obesity Reviews, 2021;22(1): e13104.
36. Eskandari F, Lake AA, Rose K, et al. A mixed-method systematic review and meta-analysis of the influences of food environments and food insecurity on obesity in highz-income countries. Food science & nutrition, 2022;10(11): 3689-3723.
37. Abay KA, Ibrahim H, Breisinger C. Food policies and
obesity in low-and middle-income countries. World Development, 2022;151: 105775.
38. Mahumud RA, Sahle BW, Owusu-Addo E, et al. Association of dietary intake, physical activity
and sedentary behaviors
with overweight and obesity among 282,213 adolescents in 89 low and middle income to high-income countries. International Journal of Obesity,
2021;45(11): 2404-2418.
39. Romano E, Ma R, Vancampfort D, et al. Multimorbidity and obesity in older adults
from six low-and
middle-income countries. Preventive Medicine, 2021;153: 106816.
40. He J, Xie Y. The sociocultural mechanism of obesity: The influence of gender role attitudes on obesity and the gender
gap. Social Science
& Medicine, 2022;293: 114655.
41. Lustig RH, Collier
D, Kassotis C, et al. Obesity I: Overview and molecular and biochemical mechanisms. Biochemical pharmacology, 2022;199: 115012.
42. Porro S, Genchi VA, Cignarelli A, et al. Dysmetabolic adipose tissue in obesity: morphological and functional
characteristics of adipose stem cells and mature adipocytes in healthy and unhealthy obese subjects. Journal of Endocrinological Investigation, 2021;44: 921-941.
43. Roh E, Choi KM. Hormonal gut–brain signaling for the treatment of
obesity. International journal of molecular sciences, 2023;24(4): 3384.44. Khanna D, Welch
BS, Rehman A. Pathophysiology of obesity, 2021.
45. Pineda E, Barbosa Cunha D, Taghavi Azar Sharabiani M, et al. Association of the retail food environment, BMI,
dietary patterns and socioeconomic position in urban areas of Mexico. PLOS Global Public Health, 2023;3(2): 0001069.
46. Atanasova P, Kusuma D, Pineda E, et al. The impact of the consumer and neighborhood food environment on dietary intake
and obesity- related outcomes: A systematic review
of causal impact
studies. Social science & medicine, 2022;299: 114879.
47. Curran F, Davis ME, Murphy K, et al. Correlates of
physical activity and sedentary behavior
in adults living
with overweight and obesity: a systematic review. Obesity Reviews, 2023;24(11): 13615.
48. Silveira EA, Mendonça CR, Delpino FM, et al.
Sedentary behavior, physical inactivity, abdominal obesity and obesity in adults and older adults: A systematic review and meta-analysis. Clinical nutrition ESPEN, 2022;50: 63-73.
49. Leitzmann M. Physical activity, sedentary behaviour
and obesity, 2021.
50. Andrie EK, Melissourgou M, Gryparis A, et al. Psychosocial factors and obesity in adolescence: A case-control study.
Children, 2021;8(4): 308.
51. Pérez-Rodrigo C, Bárbara GH, Citores MG, et al.
Prevalence of obesity and associated cardiovascular risk factors in the Spanish population: the ENPE study. Revista Española de Cardiología (English Edition), 2022;75(3): 232-241.
52. Haque R, Keramat SA, Rahman SM, et al. Association of
maternal obesity with fetal and neonatal
death: Evidence from South and South- East Asian countries. PloS one, 2021;16(9): 0256725.
53. Gao L, Peng W, Xue H, et al. Spatial–temporal trends
in global childhood overweight and obesity from 1975 to 2030: a weight mean center and projection analysis of 191 countries. Globalization and Health, 2023;19(1): 53.
54. Raisi-Estabragh Z, Kobo O, Mieres JH, et al. Racial disparities in obesity-related cardiovascular mortality in the
United States: temporal trends from 1999 to 2020. Journal
of the American Heart Association, 2023;12(18): 028409.
55. Aqeel M, Guo J, Lin L, et al. Temporal physical
activity patterns are associated with obesity in US adults. Preventive
medicine, 2021;148: 106538.
56. Friedenreich CM, Ryder-Burbidge C, et al. Physical activity, obesity and sedentary behavior in cancer etiology:
epidemiologic evidence and biologic mechanisms. Molecular oncology, 2021;15(3): 790-800.
57. Boulet N, Briot A, Galitzky J, et al. The sexual dimorphism of human
adipose depots. Biomedicines, 2022;10(10): 2615.58. Delaney KZ, Santosa S. Sex differences in regional adipose tissue depots pose different
threats for the development of Type 2 diabetes in males and females. Obesity Reviews, 2022;23(3): 13393.
59. Zhang Z, He Z, Yang H, et al. The Accumulation of Visceral Fat in Postmenopausal Women: The Combined
Impact of Prenatal Genetics, Epigenetics and Fat Depot
Heterogeneity-A Descriptive Review.
Clinical and Experimental Obstetrics & Gynecology, 2025;52(2): 26194.
60. Sahu B, Tikoo O, Pati B, et al. Role of distinct
fat depots in metabolic regulation and pathological implications. Reviews of Physiology, Biochemistry and Pharmacology, 2022: 135-176.
61. Wagner IV, Savchuk I, Sahlin L, et al. De novo and
depot-specific androgen production in human adipose tissue: A source
of hyperandrogenism in women with obesity. Obesity facts, 2022;15(2): 281-291.
62. Korac A, Srdic-Galic B, Kalezic A, et al. Adipokine signatures of subcutaneous and visceral abdominal fat in
normal-weight and obese women with different metabolic profiles. Archives
of medical science: AMS, 2021;17(2): 323.
63. Dun Q, Xu W, Fu M, et al. Physical
activity, obesity and hypertension among adults in a rapidly urbanised city. International journal of hypertension, 2021;2021(1): 9982562.
64. Boakye K, Bovbjerg M, Schuna J, et al. Urbanization and physical activity in the global Prospective Urban and Rural
Epidemiology study. Scientific reports, 2023;13(1): 290.
65. Bose C, Syamal AK, Bhattacharya K. Pattern of dietary
intake and physical activity among obese adults in rural vs
urban areas in West Bengal: A cross-sectional study. Research Journal of Pharmacy and Technology, 2022;15(9): 3924-3930.
66. Hossain MB, Khan JR, Adhikary AC, et al. Association
between childhood overweight/obesity and urbanization in
developing countries: Evidence from Bangladesh. Journal of Public Health, 2022;30(12): 2819-2828.
67. Singh P, Covassin
N, Marlatt K, et al. Obesity, body composition and sex hormones: implications for cardiovascular risk. Comprehensive Physiology, 2022;12(1): 2949-2993.
68. Dubey P, Reddy SY, Singh V, et al.
Association of exposure to phthalate metabolites with sex hormones, obesity and metabolic syndrome in US women. JAMA network open, 2022;5(9):
2233088-e2233088.
69. Brown KA. Metabolic
pathways in obesity-related breast cancer. Nature Reviews Endocrinology, 2021;17(6): 350-363.
70. Santos-Marcos JA, Mora-Ortiz M, Tena-Sempere M, et
al. Interaction between gut microbiota and sex hormones and their relation to sexual dimorphism in metabolic diseases. Biology of sex differences, 2023;14(1): 4.
71. Lobstein T, Brownell KD. Endocrine-disrupting
chemicals and obesity risk: A review of recommendations for obesity prevention policies. Obesity Reviews, 2021;22(11): 13332.
72. Amato AA, Wheeler HB, Blumberg B. Obesity and
endocrine- disrupting chemicals. Endocrine connections, 2021;10(2): 87-105.
73. Masood B, Moorthy M. Causes of obesity: a review. Clinical Medicine, 2023;23(4): 284-291.
74. Lin X, Li H. Obesity: epidemiology, pathophysiology
and therapeutics. Frontiers in endocrinology, 2021;12: 706978.
75. Lee GY, Um YJ. Factors affecting obesity in urban and
rural adolescents: Demographic, socioeconomic
characteristics, health behavior and health
education. International Journal
of Environmental Research and Public Health, 2021;18(5): 2405.
77. Van Minh H, Khuong DQL, Tran TA, et al. Childhood overweight and obesity in Vietnam: a landscape analysis of the extent and risk factors. INQUIRY: The Journal of Health Care Organization, Provision and Financing, 2023;60: 00469580231154651.
78. Saavedra JM. The changing landscape of children’s diet and nutrition: new threats, new opportunities. Annals of Nutrition and Metabolism, 2022;78(2): 40-50.
79. Gülü M, Yapici H, Mainer-Pardos E, et al. Investigation of obesity, eating behaviors and physical activity levels living in rural and urban areas during the covid-19 pandemic era: a study of Turkish adolescent. BMC pediatrics, 2022;22(1): 405.
80. Popkin BM, Ng SW. The nutrition transition to a stage of high obesity and noncommunicable disease prevalence dominated by ultra- processed foods is not inevitable. Obesity reviews, 2022;23(1): 13366.
81. Ghosh S, Paul M, Mondal KK, et al. Sedentary lifestyle with increased risk of obesity in urban adult academic professionals: an epidemiological study in West Bengal, India. Scientific reports, 2023;13(1): 4895.
82. Motomura M, Koohsari MJ, Lin CY, et al. Associations of public open space attributes with active and sedentary behaviors in dense urban areas: A systematic review of observational studies. Health & place, 2022;75: 102816.
83. Okobi OE, Ajayi OO, Okobi TJ, et al. The burden of obesity in the rural adult population of America. Cureus, 2021;13(6).
84. Musić Milanović S, Buoncristiano M, Križan H, et al. Socioeconomic disparities in physical activity, sedentary behavior and sleep patterns among 6 to 9-year old children from 24 countries in the WHO European region. Obesity Reviews, 2021;22: 13209.
85. Fismen AS, Buoncristiano M, Williams J, et al. Socioeconomic differences in food habits among 6 to 9-year old children from 23 countries-WHO European Childhood Obesity Surveillance Initiative (COSI 2015/2017). Obesity reviews, 2021;22: 13211.
86. Al Yazeedi B, Berry DC, Crandell J, et al. Family influence on children’s nutrition and physical activity patterns in Oman. Journal of Pediatric Nursing, 2021;56: 42-48.
87. Chrissini MK, Panagiotakos DB. Health literacy as a determinant of childhood and adult obesity: a systematic review. International journal of adolescent medicine and health, 2021;33(3): 9-39.
88. Lovrić B, Placento H, Farčić N, et al. Association between health literacy and prevalence of obesity, arterial hypertension and diabetes mellitus. International journal of environmental research and public health, 2022;19(15): 9002.
89. Mohajan D, Mohajan HK. Body mass index (BMI) is a popular anthropometric tool to measure obesity among adults. Journal of Innovations in Medical Research, 2023;2(4): 25-33.
90. Lister NB, Baur LA, Felix JF, et al. Child and adolescent obesity. Nature Reviews Disease Primers, 2023;9(1): 24.
91. Anand VV, Zhe ELC, Chin YH, et al. Barriers and facilitators to engagement with a weight management intervention in Asian patients with overweight or obesity: a systematic review. Endocrine Practice, 2023;29(5): 398-407.
92. Proaño GV, Moreno LMR, Arciniegas MJ, et al. Effectiveness, barriers and facilitators of overweight and obesity prevention strategies in Latin America; a scoping review and qualitative study in Colombia. The Lancet Regional Health–Americas, 2024;29.
93. Krishna J. Interrelationship Between Stress and Obesity Among Adults (Doctoral dissertation, St Terasa’s (autonomous) Ernakulam), 2023.
94. Dakanalis A, Voulgaridou G, Alexatou O, et al. Overweight and obesity is associated with higher risk of perceived stress and poor sleep quality in young adults. Medicina, 2024;60(6): 983.
95. Kautzky A, Heneis K, Stengg K, et al. Biological and psychological stress correlates are linked to glucose metabolism, obesity and gender roles in women. Neuroendocrinology, 2022;112(2): 130-142.
96. Melamed OC, Selby P, Taylor VH. Mental health and obesity during the COVID-19 pandemic. Current Obesity Reports, 2022;11(1): 23-31.
97. Jiang Y, Wang S, Ren L, et al. Effects of built environment factors on obesity risk across three types of residential community in Beijing. Journal of Transport & Health, 2022;25: 101382.
98. Blüher M, Aras M, Aronne LJ, et al. New insights into the treatment of obesity. Diabetes, Obesity and Metabolism, 2023;25(8): 2058-2072.
99. Topaloglu O, Sahin I. Pharmacological treatment of obesity in clinical practice. Med-Science, 2021;10(2): 651-657.
100. Kosmalski M, Deska K, Bąk B, et al. Pharmacological support for the treatment of obesity—present and future. In Healthcare, 2023;11(3): 433.
101. Tchang BG, Aras M, Kumar RB, et al. Pharmacologic treatment of overweight and obesity in adults. Endotext, 2024.
102. de Bont J, Márquez S, Fernández-Barrés S, et al. Urban environment and obesity and weight-related behaviours in primary school children. Environment international, 2021;155: 106700.
103. Beverly EA. Obesity management solutions in rural communities. Current Cardiovascular Risk Reports, 2024;18(1): 13-23.
104. Fu ES, Berkel C, Merle JL, et al. A Scoping Review of Tailoring in Pediatric Obesity Interventions. Childhood Obesity, 2025;21(1): 3-21.
105. Alemán JO, Almandoz JP, Frias JP, et al. Obesity among Latinx people in the United States: A review. Obesity, 2023;31(2): 329-337.
106. Baker JS, Supriya R, Dutheil F, et al. Obesity: treatments, conceptualizations and future directions for a growing problem. Biology, 2022;11(2): 160.
107. Woldemariam S, Dorner TE, Wiesinger T, et al. Multi-omics approaches for precision obesity management: Potentials and limitations of omics in precision prevention, treatment and risk reduction of obesity. Wiener Klinische Wochenschrift, 2023;135(5): 113-124.
108. Paczkowska-Abdulsalam M, Kretowski A. Obesity, metabolic health and omics: Current status and future directions. World Journal of Diabetes, 2021;12(4): 420.
109. McKenzie BL, Pinho-Gomes AC, Woodward M. Addressing the global obesity burden: a gender-responsive approach to changing food environments is needed. Proceedings of the Nutrition Society, 2024: 1-9.
110. Seiferth C, Färber T, Pape M, et al. Differential effects of the individualized gender-sensitive mHealth intervention I-GENDO on eating styles in individuals with overweight and obesity–a randomized controlled trial. BMC Digital Health, 2023;1(1): 46.