Magnitude of Hearing Loss and Associated Factors Among Children with Sleep- Disordered Breathing Attending St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia
Abstract
Background: Worldwide, sleep-disordered
breathing is an important cause of morbidity in children, affecting various
systems. Its prevalence is increasing due to different factors, including
Ethiopia. Among others, it causes auditory consequences if not timely managed.
However, its estimates regarding hearing problems along with its contributing
factors are not adequately investigated in the study setting.
Objective: To assess the magnitude and the
association of hearing loss among children with sleep-disordered breathing
attending otorhinolaryngology clinic of St. Paul’s Hospital Millennium Medical
College, Addis Ababa, Ethiopia.
Methods: A cross-sectional study design
employing retrospective chart review was conducted. Data were collected using
structured checklist. Systematic random sampling technique was employed to
recruit medical records of children diagnosed with sleep-disordered
breathing. Data were cleaned
via Epi-info and analyzed using SPSS version 27. Data were
summarized using descriptive statistics. Multiple logistic regression was performed
to identify association between dependent and independent variables, computing odds ratio. A p-value
<0.05 was considered significant. Texts, tables and figures were used to present
the results.
Results: Out of 211
studied children, 122 (57.8%) were males and children’s age ranged from three
months to fourteen years, with median (interquartile range) of 4 (3-6) years. Of all children, 22.3% (95% CI: 17-28)
were diagnosed of hearing loss. After adjusting for sociodemographic
and clinical characteristics, it was found that patients with sleep apnea were
more likely to develop hearing loss than their counterparts (AOR, 2.10; 95% CI,
1.04,4.21).
Conclusion: Hearing loss is a common health problem in patients with sleep-disordered breathing. There is a serious need for better ear care and screening programs for early detection and management of this problem.
Keywords: Sleep-disordered breathing, Hearing loss, Children, Ethiopia
Abbreviations: AOR: Adjusted Odds Ratio; CHL: Conductive Hearing Loss; COR: Crude Odds Ratio; ENT: Ear, Nose and Throat; ET: Eustachian Tube; HL: Hearing Loss; MHL: Mixed Hearing Loss; OME: Otitis Media Effusion; OSAS: Obstructive Sleep Apnea Syndrome; SDB: Sleep Disordered Breathing; SNHL: Sensorineural Hearing Loss; SPHMMC: St. Paul’s Hospital Millennium Medical College; WHO: World Health Organization
1. Introduction
1.1. Background of the study
Sleep-disordered breathing is a
syndrome of upper airway dysfunction during sleep that is characterized by
snoring and/or increased respiratory effort as a result of greater upper airway
resistance and pharyngeal collapsibility. They are grouped into Obstructive
Sleep Apnea Syndrome (OSAS), central sleep apnea, sleep-related hypoventilation
and sleep-related hypoxemia disorder. OSAS is characterized by intermittent partial
and/or complete upper airway obstruction during sleep (hypopnea or obstructive
apnea, respectively) that may compromise normal ventilation and sleep pattern.
Primary snoring is defined as noisy breathing (snoring) with-out obstructive
sleep apnea, frequent arousals from sleep or gas exchange abnormalities1,2.
OSAS is a common disorder encountered
in clinical practice that is increasingly being recognized because of the
obesity epidemic and greater public and physician awareness2. Aden tonsillar hypertrophy
is the most common cause of OSAS in children and obesity, hypotonic
neuromuscular diseases and craniofacial anomalies are other major risk factors3,4. Its main symptoms include
snoring, sleepiness and significant reports of sleep apnea episodes1. Although snoring is the most
common presenting complaint in children, but the clinical presentation varies
according to age4.
Pediatric OSAS has become widely
recognized only in the last few decades as a likely cause of wide range of
morbidities among children1.
The prevalence of parent-reported ‘habitual snoring ranged from 1.5 to 6% while
parent-reported apneic events during sleep span from 0.2% to 4%. Moreover,
prevalence of SDB by varying constellations of parent-reported symptoms was
estimated to range 4% to 11%; and OSA diagnosed by varying criteria on
diagnostic studies is approximately 3% in children4, with the range extending from 1% to 4%. The overall
prevalence of parent-reported snoring by any definition is estimated to stand at
7.455. Mean
prevalence of mild to severe possible OSAS and severe possible OSAS in children
across all grade levels was 9.5% and 1.6%, respectively6.
Pediatric OSAS prevalence has two
peak periods. The first peak occurs in children from 2 to 8 years of age, with
the presence of enlarged adenoids and/or tonsils. A second peak arises during
adolescence in relation to weight gain4. Aden tonsillar hypertrophy, obesity, craniofacial
anomalies and abnormal neuromotor tone are the main conditions predisposing to
OSA in childhood7.
SDB and particularly in the form of
OSA, affects many systems, including the cardiovascular, endocrine,
neuropsychiatric, cognitive and vestibular systems8-11. The auditory system is affected by
Eustachian tube dysfunction and hypoxia-induced cochlear and vestibular
dysfunction12,13.
In general, risk factors for hearing
impairment include host-related and environmental factors. Host factors include
male sex, genetic susceptibility, craniofacial deformities, immunosuppression
and adenoid hypertrophy. Environmental variables include low socioeconomic status,
URIs, the fall and winter months, daycare, having older siblings, exposure to
tobacco smoke, allergies, obesity and GERD14,15. Despite the possibility of burden of
hearing impairment among patients with SDB, there is scarcity of data from
Ethiopian perspective. Hence, the present study was designed with the aim
assessing the magnitude of hearing loss among children with sleep-disordered
breathing attending otorhinolaryngology clinic of St. Paul’s Hospital
Millennium Medical College, Addis Ababa, Ethiopia.
1.2.
Statement of the problem
The
adverse consequences of pediatric OSAS may not simply be confined to the
child’s immediate well-being and development but may continue to be detrimental
to the patient’s long-term health in adulthood14. Children with OSAS may experience a range of
problems, including delayed growth, failure to thrive, cardiovascular issues,
enuresis, attention-deficit/hyperactivity disorder, poor academic performance
and emotional instability. Furthermore, it results in hearing loss in children3,4,16. Hearing loss in
children causes lifelong deficits in speech and language acquisition, poor
academic performance, personal-social mal-adjustments and emotional
difficulties17.
An
estimated 1.57 billion people globally had hearing loss in 2019, accounting for
one in five people18.
The World Health Organization (WHO) reported that hearing loss is the fourth
highest cause of disability globally, affecting around 6.1% of the world's
population. Current data suggest that approximately 466 million people of the
world’s population have disabling hearing loss, therefrom, 34 million children,
thereby impacting their quality of life. It has been estimated that 8.6% of
children in Sub-Saharan areas have hearing problems (i.e. almost 18 million
children19.
Untreated
hearing loss is estimated to cost over 660 billion euros annually, including
expenditures for the health care and education systems, lost productivity and
costs for accessibility, adaption and social inclusion for individuals with
impairments, according to a report by the WHO19. In 2000 in the United States, severe to
profound hearing impairment was estimated to cost society $297 000 over the
lifetime of an affected individual, mostly due to reduced work productivity.
The average overall medical expense for a child in South Africa for the first
five and ten years following cochlear implantation in 2015 was $27,000 and
$40,00020. Additionally,
hearing loss is independently associated with impairment of executive function
and behavioral difficulties among such patients21.
The WHO
research states that South Asia, Asia Pacific and Sub-Saharan Africa are the
main global regions afflicted by hearing loss that is debilitating, with
prevalence rates that are over four times higher than in high-income regions. Under
15 years of age, 60% of hearing impairment cases result from preventable
causes, 31% of which are related to infections19.
Therefore,
it is important to invest in the early detection, diagnosis and rehabilitation
of hearing impairment in children so that the proper interventions can be
carried out.
In Ethiopian
context, despite the fact that there is scarcity of data regarding the exact
magnitude and impact of SDB, otic complications are usually neglected
conditions due to insufficient funds, work force, facilities and knowledge. Thus,
the audiological aspect of SDB has received only little attention although it
has been shown to result in profound negative impact. Hence, there is a need to
conduct such studies to its magnitude and its contributing factors in Ethiopian
context. This paper aims to give data about the prevalence and association of
pediatric hearing loss among SDB patients, by taking case of children visiting
otorhinolaryngology clinic of St. Paul’s Hospital Millennium Medical College,
Addis Ababa, Ethiopia.
1.3.
General objective
To
assess the magnitude of hearing loss and associated factors among children with
sleep-disordered breathing attending otorhinolaryngology clinic of St. Paul’s
Hospital Millennium Medical College, Addis Ababa, Ethiopia, 2022.
1.4. Specific objectives
To find out the magnitude of hearing loss among
children with sleep-disordered breathing attending otorhinolaryngology clinic
of St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia, 2022.
To identify factors associated with hearing loss among
children with sleep-disorder breathing attending otorhinolaryngology clinic of
St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia.
1.5.
Significance of the study
The
findings of this study will be an input to the country’s health sector
transformation plan, which aims to improve equity, coverage and utilization of
essential health services. Understanding disease prevalence, that is, the
proportion of a population with the condition, is critical to anticipating
health care needs and allocating appropriate resources. In addition,
comparisons of the burden by demographic factors may yield etiological clues
and identify subgroups at particularly high risk for targeted case finding.
Moreover, this study is expected to contribute to the existing body of
knowledge regarding magnitude of hearing impairment and its associated factors
among children with SRBD attending otolaryngology clinics at the study setting,
thereby shedding some light to the potential areas for evidence-based
mitigation measures. Furthermore, the findings are expected to be of particular
importance to local planners and health-policy makes to design policies that
would alleviate this growing health burden among vulnerable age groups.
Finally, this study will also serve as a baseline information to other
interested investigators in similar research endeavors.
2. Methods and Materials
2.1. Study area and period
St. Paul’s Hospital Millennium Medical College (SPHMMC) was
founded bin July 1947 during the reign of Emperor Haile Selassie and it is the
second largest hospital in Ethiopia. The hospital has more than 2500 clinical,
academic and administrative staff. While the inpatient capacity is more than
700 beds, the college sees more than 2000 emergency and outpatient clients
daily. The clinic is run five times a week by a team of seniors, residents,
nursing and other staff. The hospital was selected for presence of organized
ENT clinics providing services for a relatively large population size belonging
both to pediatric and adult age groups. Data were
collected from October 1st to October 31st,
2022.
2.2. Study design
A cross-sectional study design
employing retrospective chart review was conducted.
Children with sleep-disordered breathing visiting Ear, Nose and Throat
clinic of St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia.
Randomly selected children with sleep-disordered
breathing visiting Ear, Nose and Throat clinic of St. Paul’s Hospital
Millennium Medical College, Addis Ababa, Ethiopia from September 1st, 2021 to August 31st, 2022 and who fulfill the inclusion criteria.
2.5.1. Inclusion criteria:
Children with clinical diagnosis of sleep-disordered
breathing.
Children aging 14 years or younger by the time of data
collection
Children with pre-existing or congenital hearing disorder
Children whose charts are lost or incomplete
2.6. Sample size determination
The sample size for this study is calculated using a single population proportion formula. Considering the population proportion of estimate proportion of patients with sleep-disordered breathing that, develop hearing problem to be 50% due to absence of previous study done at the current study setting and with the intention of obtaining maximum sample size:
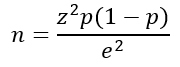
n = the required sample size
p = proportion of patients with SDB that
develop hearing loss =0.5
Zα\2
= the critical value at 95% confidence level = 1.96
e = precision (margin of error) = 5%
Accordingly,
Hence, to compute the final sample size using the correction formula as the total source population from the hospital is less than 10,000 (N=465)
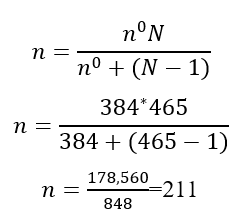
Therefore, adding 5% for contingency on the calculated sample size, the final sample size obtained was 222.
2.7.
Sampling procedures
The study
participants were selected by means of systematic random sampling technique, as
the total number of children with SDB was estimated to be 465 during the study
period. The first patient was selected via
lottery method and then after every other patient was approached for data
collection, with K being 2 (that is, 465/222).
2.8. Data collection tools and
procedures
Patients’ medical records were
retrieved and reviewed. Data were collected from charts using structured checklist.
Caregivers were contacted virtually via phone calls whenever need arises. The
data collection format includes questions divided into three parts (background
information, clinical variables and otoscopic, endoscopic and audiometric
parameters) and it was adapted from related literatures22. Data were gathered from logbook
records of ENT patient registry over one year (from September 1st, 2021 to August 31st, 2022). Two professional healthcare workers were
recruited and trained on data collection procedures.
2.9. Study variables
2.9.1. Dependent variable
- Hearing loss
2.9.2. Independent variables
-
Age of the child
-
Sex
-
Residence
-
History of SDB symptoms
-
Subjective hearing loss
-
Ear discharge
-
Adenoid size
-
Tonsillar size
Sleep-disordered breathing: Represent those sleep disorders, which are characterized by abnormal
respiration during sleep. It includes Obstructive Sleep Apnea Syndrome (OSAS),
central sleep apnea, sleep-related hypoventilation and sleep-related hypoxemia
disorder2.
Hearing loss: Refers to any degree of hearing impairment as assessed clinically including history and physical examination (tympanometry included), with or without audiometric confirmation.
2.11. Data processing and analysis
Data entering, coding and cleaning were
performed using Epi-info version 7.2.2.2 and then exported to SPSS version 27
to carry out statistical analysis. Frequency and cross tabulation were used to
check for missed value and variables. The demographic and clinical
characteristics of patients were computed by using descriptive statistics such
as mean, median, percentage, frequencies, interquartile range and standard
deviation. Multiple logistic regression was used to determine
associations between the independent and dependent variables. Those variables
with p value of ≤0.25 in bivariable logistic
regression were included in the multivariable logistic regression model to
compute adjusted odds ratio at 95% confidence interval. Hosmer-Lemeshow goodness-of-fit test was run to test the fitness of the model, where p value
of >0.05 was used to declare data fitness. Variables with p-value ≤0.05 were considered as statistically associated with
the outcome variable. Finally, the study findings were presented using
diagrams, tables and figures.
2.12. Data quality management
To ensure data quality, data
collection tool was prepared after thorough review of relevant literatures and
related studies. An English version, pretested questionnaire was used to
collect data. Brief training for the data collectors (two health professionals)
about the process of data collection was given before the actual data
collection. The data collection procedure was closely supervised and each day,
filled questionnaires were double-checked manually for consistency and
completeness by data collectors and principal investigator before proceeding to
statistical analysis.
2.13. Dissemination of the study
findings
The findings of this study will be submitted to Department of Otorhinolaryngology, Head and Neck Surgery, St Paul’s Hospital Millennium Medical College as a partial fulfillment of the requirements for the specialty certificate in Otorhinolaryngology-Head and Neck Surgery. The outcome of this study will be presented to the higher officials of the college. Additionally, the manuscript will be submitted to a peer-reviewed scientific journal for possible publication.
3. Results
3.1. Socio-demographic
characteristics
This study included data derived from two hundred and eleven patients with sleep-disordered breathing, making a response rate of 95%. Among the studied children, more than two-thirds (122; 57.8%) were males and the remaining 89 (42.2%) were females. Children’s age ranged from three months to fourteen years, with median (interquartile range) of 4 (3-6) years. Regarding current residence, about three-fourths (158; 74.9%) of the study population were urban whereas the remaining quarter (53; 25.1%) were documented to rural residents. (Table 1).
Table 1: Socio-demographic characteristics of children with sleep disordered breathing attending otorhinolaryngology clinic of St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia, 2022.
|
Variable |
Frequency |
Percent (%) |
|
Child’s age |
||
|
<2 years |
19 |
9 |
|
2 to 5 years |
129 |
61.1 |
|
>5 years |
63 |
29.9 |
|
Child’s sex |
||
|
Male |
122 |
57.8 |
|
Female |
89 |
42.2 |
|
Residence |
||
|
Urban |
158 |
74.9 |
|
Rural |
53 |
25.1 |
3.2. Clinical characteristics
With regard to clinical characteristics of the studied children, all had snoring episodes during sleeping while two-third (136; 64.5%) of the children were described to be mouth breathers. A third (70) were documented to have sleep apnea. Close to half (98;46.4%) of the children had history of night-time awakenings whereas only two (0.9%) had enuresis. Seventy-four (35.1%) had history of restless sleep, while 22 (10.4%) had caregiver-reported subjective hearing loss and only three (1.4%) had some form of aural discharge. Regarding adenoid size, more than half (107;50.7%) had grade III on endoscopic evaluation while 83 (39.3%) and 59 (28%) children had grade III and grade IV tonsillar hypertrophy based on Brodsky grading scale (Table 2).
Table 2: Clinical characteristics of children with sleep disorder breathing attending otorhinolaryngology clinic of St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia, 2022.
|
Variable |
Frequency |
Percent (%) |
|
Snoring |
||
|
Yes |
211 |
100 |
|
No |
0 |
0 |
|
Mouth breathing |
||
|
Yes |
136 |
64.5 |
|
No |
75 |
35.5 |
|
Sleep apnea |
||
|
Yes |
70 |
33.2 |
|
No |
141 |
66.8 |
|
Nocturnal awakening |
||
|
Yes |
98 |
46.4 |
|
No |
113 |
53.6 |
|
Enuresis |
||
|
Yes |
2 |
0.9 |
|
No |
209 |
99.1 |
|
Restless sleep |
||
|
Yes |
74 |
35.1 |
|
No |
137 |
64.9 |
|
Subjective hearing
loss |
||
|
Yes |
22 |
10.4 |
|
No |
189 |
89.6 |
|
Ear discharge |
||
|
Yes |
3 |
1.4 |
|
No |
208 |
98.6 |
|
Adenoid grade |
||
|
Grade 0 |
6 |
2.8 |
|
Grade I |
14 |
6.6 |
|
Grade II |
47 |
22.3 |
|
Grade III |
107 |
50.7 |
|
Grade IV |
37 |
17.5 |
|
Tonsillar grade |
||
|
In situ |
9 |
4.3 |
|
Grade I |
12 |
5.7 |
|
Grade II |
48 |
22.7 |
|
Grade III |
83 |
39.3 |
|
Grade IV |
59 |
28 |
3.3. Magnitude of hearing loss
Of the total, 22.3% (95% CI: 17-28) of the study
participants were diagnosed of hearing loss, as depicted in (Figure 1). Moreover, most (39; 83.0%) of the hearing
losses were bilateral while the remaining 8 (17.0%) were unilateral. Additionally,
audiometric evaluation showed conductive hearing loss in all of the children
who had the test done.
3.4. Factors affecting hearing loss
In this study, thirteen variables,
namely child’s age, child’s sex, residence, snoring, mouth apnea, breathing,
sleep nocturnal awakening, enuresis, restless sleep, subjective hearing loss,
ear discharge, adenoid grade and tonsillar grade were considered in the
regression analysis. In order to determine the factors associated with hearing
loss while controlling possible confounders, independent variables that yielded
p value of ≤0.25 in binary logistic regression
were exported to multiple regression model to compute adjusted odds ratio in
determining odds of hearing loss.
Accordingly, five factors, namely child’s sex, child’s age, residence, sleep apnea and adenoid grade were observed in the bivariable analysis to be associated with presence of HL among the studied children. Following multivariable logistic analysis, the only variable that showed statistically significant association with HL was presence of sleep apnea. Specifically, this study showed that when compared to those who were described not to have sleep apnea, children with sleep apnea were more likely to have hearing loss [AOR=2.10 (95%CI:(1.04,4.21)] (Table 3).
Table 3: Factors associated with hearing loss among children with sleep-disordered breathing attending otorhinolaryngology clinic of St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia, 2022
|
Variable |
Hearing loss |
COR (95%CI) |
AOR (95%CI) |
|
|
Yes |
No |
|||
|
Child’s sex |
||||
|
Male |
31 |
91 |
1.55(0.79,3.06) |
1.58(0.75,3.31) |
|
Female |
16 |
73 |
1 |
1 |
|
Child’s age |
||||
|
<2 years |
9 |
10 |
3.83(1.28,11.47) |
2.42(0.69,8.56) |
|
2 to 5 years |
26 |
103 |
1.07(0.50,2.30) |
0.83(0.36,1.90) |
|
>5 years |
12 |
51 |
1 |
1 |
|
Residence |
||||
|
Urban |
41 |
117 |
2.75(1.09,6.90) |
2.75(1.01,7.51) |
|
Rural |
6 |
47 |
1 |
1 |
|
Sleep apnea |
||||
|
Yes |
21 |
49 |
1.90(0.98,3.69) |
2.10(1.04,4.21)* |
|
No |
26 |
115 |
1 |
1 |
|
Adenoid grade |
||||
|
Grade 0 |
3 |
3 |
3.11(0.53,18.22) |
3.12(0.52,18.79) |
|
Grade I |
7 |
7 |
3.11(0.86,11.29) |
3.07(0.83,11.35) |
|
Grade II |
11 |
6 |
0.95(0.36,2.78) |
0.99(0.36,2.78) |
|
Grade III |
17 |
90 |
0.59(0.24,1.46) |
0.54(0.22,1.37) |
|
Grade IV |
9 |
28 |
1 |
1 |
Only variables with p value <0.25 in bivariable logistic regression are shown here
4. Discussion
The present study aimed to assess the
magnitude of hearing loss among children with sleep-disordered breathing by
analyzing children attending otorhinolaryngology clinic of St. Paul’s Hospital
Millennium Medical College, Addis Ababa, Ethiopia. Subsequently, it has
discovered several important findings. The study showed that a significant
portion of children with SDB have hearing impairment, affecting more than one-fifth
of them. Moreover, it was revealed that hearing loss was independently contributed
by the presence of sleep apnea.
Specifically, this study demonstrated
that more than a fifth of children with SDB have hearing loss. This finding was
marginally higher than the observation in American setting, where 14.8% of
patients with sleep-disordered breathing had hearing loss23. The current finding is supported by the
works of Hill and co-authors, who observed that snoring children had
significantly poorer hearing and greater past exposure to hearing loss than
non-snoring controls21.
This finding is much higher than the Taiwanese report in which only 0.33% of
OSAS patients were observed to experience subsequent hearing loss in 9 years16.
The large burden of HL among patients
with SDB is plausible since there is hypoxia/hypercapnia, chronic noise
exposure to the cochlea and persistent inflammatory status in sleep apnea,
which in turn cause damage to the cochlea, which is highly sensitive to
circulatory alterations due to its lacks of adequate collateral blood supply24,25. Yet, this finding is of serious concern
given the fact that hearing loss can be irreversible, with long lasting impact
on the lives of the victims26.
The large difference in the magnitude
of hearing loss between this study and the previous estimates in other settings
could be attributed to the relative abundance of poverty-related factors and poor
access to health care including medical and surgical intervention among
children in resource-challenged regions such as ours. Moreover, the
inter-regional variation can be explained by differences in length of
follow-up, sample size, the characteristics of the studied population and methodological
design. For example, the current study employed a one-year chart review while
the Taiwanese study followed patients for nine years. Besides, it should be
remembered that this study was done during Coronavirus disease (COVID-19)
pandemic, which was quoted to be associated with the development of Sudden
Sensorineural Hearing Loss (SSNHL)27.
On the other hand, the current study highlighted that SDB children with caregiver reported sleep apnea have higher risk of developing hearing loss. This is more or less in line with the South Korean report that demonstrated that lowest oxyhemoglobin saturation be an independent factor influencing auditory function in severe obstructive sleep apnea28. The possible explanation for this finding can be multiple and it includes the fact that chronic periodic hypoxia associated with sleep apnea can provoke oxidative stress and impaired vascular endothelial function and it may also harm the vasa nervorum, with collective damage to the hearing function of the auditory system29.
5. Strengths and Limitations
5.1. Strengths
The study was done at one of the
leading tertiary hospitals of the country where the highest numbers of
sleep-disordered breathing patients with varying socio-demographic
characteristics are expected to be represented.
5.2. Limitations
This study is expected to
be prone for the limitation of a cross-sectional study design and as a
consequence, establishing causal relationship would be impossible.
The current study was
done at a single otorhinolaryngology center, making it difficult to generalize
to other populations attending different public health facilities.
Being a chart-based
retrospective study, some important information in patients’ clinical records
were missing due to improper filling of patients’ particulars.
There was lack of confounder
method adopted to measure hearing loss (E.g. ABR).
6. Conclusion
This study showed that hearing loss
is a common health problem in patients with sleep-disordered breathing, affecting
about one in four children with SDB have hearing impairment. Moreover, presence
of sleep apnea was the only factor that contributed to hearing loss in the
studied population.
7. Recommendation
Based on the findings obtained in the
study, the following recommendations can be forwarded.
For health professionals: Whenever possible, trained health
professionals should screen children with sleep-disordered breathing for
auditory complication, with emphasis to those with predisposing factors such as
sleep apnea.
For local health policy makers: Local policy makers, along with
concerned stakeholders, should consider awareness creation modalities via mass
media and health extension workforce to consolidate the caregivers’ knowledge
on the care of affected children on top of the need for early screening and
diagnosis of such vulnerable population.
For researchers: Future studies should be done to validate the current findings and they should employ stronger study designs including comprehensive set of variables to have deeper understanding.
8. Deceleration
Ethical clearance was obtained from Ethical Review Committee of SPHMMC. Clearance letter was submitted to the medical director before carrying out the actual data collection for the study. This was followed by introduction and familiarization of the data collectors with the study subunits. Before interviewing clients, the purpose of the study was briefly explained and written informed consent was sought. During data collection, study participants were informed that the information collected will be kept anonymous and confidential. Consent for publication was obtained from the patient and is available upon request from the corresponding author.
9. Authors' Contributions
Dr. Mohammedsefa Arusi
and Dr. Martha Driba contributed substantially to the conception, design,
drafting of the manuscript and final approval of the version to be published.
Dr. Addis Asfaw, Dr. Wale
Limeh and Dr. Zeyneb Saleh contributed to data acquisition, interpretation of
data, critical revision of the manuscript and final approval of the version to
be published. All authors reviewed and approved the manuscript.
10. References
- Mannarino MR, Di
Filippo F, Pirro M. Obstructive sleep apnea syndrome. Eur J Intern Med.
2012;23: 586-593.
- Burman D. Sleep Disorders: Sleep-Related
Breathing Disorders. FP Essent. 2017;460: 11-21.
- Katz ES, D’Ambrosio CM. Pediatric
obstructive sleep apnea syndrome. Clin Chest Med. 2010;31: 221-234.
- Chang SJ, Chae KY.
Obstructive sleep apnea syndrome in children: Epidemiology, pathophysiology,
diagnosis and sequelae. Korean J Pediatr. 2010;53: 863-871.
- Lumeng JC, Chervin RD. Epidemiology of
pediatric obstructive sleep apnea. Proc Am Thorac Soc. 2008;5: 242-252.
- Tsukada E, Kitamura S, Enomoto M, et al. Prevalence of childhood obstructive
sleep apnea syndrome and its role in daytime sleepiness. PLoS One. 2018;13: 1-12.
- Verhulst S, Kaditis
A. Obstructuve sleep apnoea in children. Breathe. 2011;7: 240-247.
- Jean-Louis G, Zizi
F, Clark LT, Brown CD, McFarlane SI. Obstructive sleep apnea and cardiovascular
disease: Role of the metabolic syndrome and its components. J Clin Sleep Med.
2008;4: 261-272.
- Milkov MP.
Vestibular and hearing disorders in obstructive sleep apnea patients. Известия На Съюза На Учените - Варна Серия Медицина И Екология. 2018;23: 45.
- Vahedi E, Khosravi
A, Alizadian R, Badri T. Cardiac function and cognitive function in patients
with obstructive sleep apnea. Sleep Sci Pract. 2021;5: 1.
- Yahia S, Ali H, Sallam Y. Effect of
tonsillar hypertrophy on velopharyngeal closure and resonance of speech. J
Recent Adv Med. 2020.
- Kasemsuk N, Chayopasakul V, Banhiran W, et
al. Obstructive Sleep Apnea and Sensorineural Hearing Loss: A Systematic Review
and Meta-analysis. Otolaryngol - Head Neck Surg (United States). 2022.
- Kayabasi S, Hizli O,
Yildirim G. The association between obstructive sleep apnea and hearing loss: a
cross-sectional analysis. Eur Arch Oto-Rhino-Laryngology. 2019;276: 2215-2221.
- Banda FM, Powis KM,
Mokoka AB, et al. Hearing impairment among children referred to a public
audiology clinic in gaborone, botswana. Glob Pediatr Heal. 2018;5: 1-8.
- Sallavaci S.
Prevalence and Factors associated with Hearing Impairment in Preschool Children
in Albania. Arch Med. 2016;8: 1-8.
- Chen CK, Shen SC, Lee LA, et al. Idiopathic
sudden sensorineural hearing loss in patients with obstructive sleep apnea. Nat
Sci Sleep. 2021;13: 1877-1885.
- Nunes AD da S,
Pereira RN, Junior AP, Barbosa IR, Balen SA. Sensitivity and specificity of
three hearing screening protocols in the school setting. 2020;22: 1-8.
- GBD Hearing Loss
Collaborators. Hearing loss prevalence and years lived with disability , 1990-
2019 : findings from the Global Burden of Disease Study 2019 2021;397: 996-1009.
- World health
orgnization (WHO). Addressing The Rising Prevalence of Hearing Loss. World
Health Organization: Geneva, Switzerland. 2018: 655-658.
- Mohr PE, Feldman JJ,
Dunbar JL, McConkey-Robbins A, Niparko JK, Rittenhouse RK. The Societal Costs
of Severe to Profound Hearing Loss in the United States. Int J Technol Assess
Health Care. 2000;16: 0266462300103162.
- Hill CM, Bucks RS,
Kennedy CR, et al. Hearing loss mediates executive function impairment in
sleep-disordered breathing. Sleep Med. 2017;34: 18-23.
- Zhang Y, Xu M, Zhang
J, Zeng L, Wang Y, Zheng QY. Risk factors for chronic and recurrent otitis
media-A meta-analysis. PLoS One. 2014;9.
- Nathan AS, Hubbell RD, Levi JR. Management
of children with co-occurring sleep disordered breathing and hearing loss. Int
J Pediatr Otorhinolaryngol. 2022;163: 111367.
- Chopra A, Jung M,
Kaplan RC, et al. Sleep apnea is associated with hearing impairment: The
Hispanic community health study/study of Latinos. J Clin Sleep Med. 2016;12: 719-726.
- Lazarini PR, Kfouri
AC. Idiopathic sudden sensorineural hearing loss: etiopathogenic aspects. Braz
J Otorhinolaryngol. 2006;72: 554-561.
- Olusaniya BO, Baimgboye BA, Somefun AO.
Review Article The Burden and Management of Infants with Life-long and
Irreversible Hearing Impairment in Nigeria. 2011;20: 310-321.
- Meng X, Wang J, Sun
J, Zhu K. COVID-19 and Sudden Sensorineural Hearing Loss: A Systematic Review.
Front Neurol. 2022;13.
- Olusaniya BO, Baimgboye BA, Somefun AO. Review Article The Burden and Management of Infants with Life-long and Irreversible Hearing Impairment in Nigeria. 2011;20: 310-321.
- Gozal D, Kheirandish-Gozal L. Cardiovascular morbidity in obstructive sleep apnea: Oxidative stress, inflammation and much more. Am J Respir Crit Care Med. 2008;177: 369-375.
Annex
001. Data collector: code ____/___/___ Name ______________________
002. Date of data collection ____/___/____ Time ___________
003. Checked by Supervisor: Signature ________day _______month ______ year____
|
S.No |
Questions |
Response |
Remark |
|
1 |
Sex |
1. Male 2. Female |
|
|
2 |
Age |
_____ years |
|
|
3 |
Address |
1. Urban 2. Rural |
|
Section II presenting symptoms
|
S.No |
Question
|
Response
|
Remark
|
|
1 |
Snoring
|
1.Yes
2.No |
|
|
2 |
Mouth
breathing |
1.Yes
2.No |
|
|
3 |
Sleep
apnea |
1.Yes
2.No |
|
|
4 |
Restless
sleep |
1.Yes
2.No |
|
|
5 |
Frequent
awakenings |
1.Yes 2.No |
|
|
6 |
Enuresis |
1.Yes 2.No |
|
|
|
Parental reported Hearing loss |
1.Yes 2.No |
|
|
|
Duration
of hearing loss |
1.<3month
2.3-6month 3.>6month |
|
|
|
Unilateral
or bilateral hearing loss |
1.Unilateral
2.Bilateral |
|
|
|
Progression
|
1.Progressive 2.Suddene
|
|
|
|
Ear
discharge |
1.Yes
2.No |
|
Section III Endoscopic /Otoscopic/Physical finding
|
S.No |
Question
|
Responses |
Remark |
|
1 |
Tympanic
membrane |
1.Normal
2.Bulged 3.Erythematous
4.Retracted 5.perforated |
|
|
2 |
Grade
of tonsil |
1.grade I 2.grade II 3.grade III 4 .grade IV |
|
|
3 |
Endoscopic Grades of adenoid |
1.grade I 2.grade II 3.grade III 4 .grade IV |
|
|
4 |
Pure
tone average |
1.CHL
2. SNHL 3. MHL |
|
|
5 |
Tympanometry |
1.B-type
2.AS-type 3.AD-type
4.A –type |
|
|
6 |
Objective Hearing loss |
1.
Yes 2. No |
|