Pulmonary Aspergilloma with DIPNECH; A Rare Case Report
Abstract
Introduction:
Pulmonary Aspergilloma
(PA) usually presents with a fungus ball or mycetoma as a result of saprophytic
colonization of Aspergillus fumigatus causing parenchymal damage and forming a
cavitary lesion. The incidence of pulmonary aspergilloma with tumor lesions is
rare. In our study, we aimed to present a patient diagnosed with diffuse
idiopathic neuroendocrine cell hyperplasia (DIPNECH) in a patient who underwent
anatomical resection for PA.
Case: A 46-year-old woman with no history
of comorbidities presented with a history of frequent pneumonia and recurrent
hemoptysis. Thorax CT showed a 47x42 mm cavitary lesion with irregular walls in
the lower lobe of the right lung, millimetric solid nodular lesions and fungus
ball appearance within the cavity, bronchiectatic changes and septal
thickening. Spirometry was performed and after 7 days antibiotherapy with
clinical improvement, the patient underwent lower lobectomy and mediastinal
lymph node dissection via right thoracotomy. Histopathologic examination
revealed a 3 cm cavitary lesion in the lobe which was compatible with pulmonary
aspergilloma. Histopathologic examination revealed a 3 cm cavitary lesion in
the lobe which was compatible with pulmonary aspergilloma. It was observed to be consistent
with diffuse idiopathic neuroendocrine cell hyperplasia.
Conclusion: In DIPNECH,
multifocal pulmonary nodules with mosaic attenuation are seen, which is rare. It is a generalized intramucosal
proliferation of pulmonary neuroendocrine cells clustered in monolayers or
small groups that can protrude into the bronchial lumen. DIPNECH was diagnosed
in 5.4% of patients operated for carcinoid tumor and the recommendation in the
literature is to perform surgical resection in case DIPNECH is detected. PA and
DIPNECH are two different entities and their coexistence is rare. DIPNECH,
which is also associated with other diagnoses, can often be detected
histopathologically in carcinoid tumors and surgical resection is the
appropriate treatment method as we have stated in our study.
Keywords: Pulmonary nodule, Pneumonia, Hemoptysis, Tumor
Abbrevations: PA: Pulmonary
Aspergilloma; DIPNECH: Diffuse Idiopathic Pulmonary Neuroendocrine Cell
Hyperplasia; CT: Computed Tomatoghraphy
1. Introduction
Pulmonary Aspergilloma (PA) usually
presents with a fungus ball or mycetoma as a result of saprophytic colonization
of Aspergillus fumigatus causing parenchymal damage and forming a cavitary
lesion1. It is known to affect
more than 200,000 people worldwide and can cause recurrent or massive
hemoptysis as a result of bronchial artery erosion and can be mortal in 2% to 50%2. Conditions such as chronic lung disease,
(bronchiectasis, sarcoidosis etc.) immunosuppression, hydatid cyst, malignancy,
diabetes mellitus, lupus, hypertension, coronary artery disease, lung abscess,
invasive ventilation, postoperative lung injury are frequently involved in the
etiology. On computed tomography of the thorax, demonstration of a fungus ball
in the cavity and ectatic bronchus are diagnostic3.
In addition, tumorlet lesions, which present as nodular lesions smaller than 5
mm, are rare neuroendocrine cell hyperplasias and are frequently seen in
carcinoid tumors4. The incidence
of pulmonary aspergilloma with tumor lesions is rare. In our study, we aimed to
present a patient diagnosed with Diffuse Idiopathic Pulmonary Neuroendocrine
Cell Hyperplasia (DIPNECH) in a patient who underwent anatomical resection for
PA.
2. Case Presentation
The patient with no history of
comorbidities who is 46 year-old, presented with a history of frequent
pneumonia and recurrent hemoptysis. Thorax CT showed a 47x42 mm cavitary lesion
with irregular walls in the lower lobe of the right lung, millimetric solid
nodular lesions and fungus ball appearance within the cavity, bronchiectatic
changes and septal thickening, as well as pathologic hilar and mediastinal
lymphadenopathies over 2 cm in size (Figure 1). Laboratory
tests on admission did not reveal any features except leukocytosis and elevated
C-reactive protein. Further tests were not performed because the Galactomannan
Agglutination test was not available in the center where we worked. Spirometry
was performed and after 7 days antibiotherapy with clinical improvement, the
patient underwent lower lobectomy and mediastinal lymph node dissection via
right thoracotomy (Figure 2)
Histopathologic examination revealed a 3 cm cavitary lesion in the lobe which was compatible with pulmonary aspergilloma. Hilar and mediastinal lymph nodes were evaluated as reactive. On microscopic examination, neuroendocrine hyperplasia and tumorlet foci, each 1-2 mm in diameter, were observed in the sections. It was observed to be consistent with DIPNECH. Mitosis was found to be less than 2 per 2 mm². Necrosis was not observed. Immunohistochemistry examination revealed CD56 (+), ChromograninA (+), Synaptophysin (+), PANCK (+), CDX2 (-), CD34 (-), D240 (-), CK7 (-), CK20 (-), TTF1 (focal +), NapsinA (-), ER (-), PR (-), PAX8 (-), WT1 (-), GATA3 (-) were detected and Ki67 proliferation index was 1%. (Figure 3). The chest tube was removed on postoperative day 5 and the patient was discharged on postoperative day 6. The patient was followed up with adjuvant voriconazole for 3 weeks as recommended in the literature and no pathology was observed at the 18th month postoperative follow-up1.
Figure 1: Preoperative computerized thorax CT image (A: axial view B:coronal view C: Sagital view).
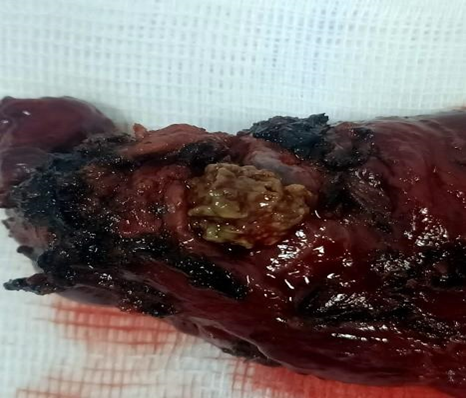
Figure 2: Section image (section was taken for diagnostic purposes after the section was removed from the lesion).
A B C
Figure 3: Histopathologic examination images. A: Cavitary lesion (aspergilloma) with a fungal ball in the lower right corner. B: Chronic inflammation with eosinophils is seen in the cavity wall. C: Bronchiole-limited neuroendocrine cells forming small nodules in the upper left corner (H&E, 40x).
3. Discussion
While medical or
surgical treatment was accepted as the standard of care in pulmonary
aspergilloma in the 1970s, combined treatment (anatomical resection with
adjuvant therapy) has been accepted as the gold standard since 20103.
Sublobar resections in peripherally located, <3 cm lesions are controversial
due to worse prognosis, more air leakage and risk of recurrence5.
In DIPNECH, multifocal pulmonary nodules with mosaic attenuation are seen,
which is rare6. It is a generalized intramucosal
proliferation of pulmonary neuroendocrine cells clustered in monolayers or
small groups that can protrude into the bronchial lumen. The cells do not cross
the mucosal basal lamina. (If they do, they are called “tumorlets.”) The cells
are round, oval or spindle-shaped, have moderate amounts of eosinophilic
cytoplasm and have round to oval nuclei with salt and pepper chromatin.
Histopathologically, the presence of ≥ 5 neuroendocrine cells distributed linearly or in
clusters within the basement membrane in ≥ 3 bronchioles and association with ≥ 3 tumorlets is diagnostic.
DIPNECH was diagnosed in 5.4% of
patients operated for carcinoid tumor and the recommendation in the literature
is to perform surgical resection in case DIPNECH is detected7. The coexistence of PA and DIPNECH is a
rare condition and in the case report of Yazgan et
al.8, a 67-year-old female patient underwent left lower
lobectomy due to fungus ball and the diagnosis of PA and DIPNECH could be shown.
In the case report of Moskovljevic, et al.4,
a 71-year-old female patient with a positive galactomannan agglutination test was
diagnosed with PA and DPNECH after right lower lobectomy.
If the lesions, which are considered
as nodular proliferation of neuroendocrine cells, are <5 mm from the
bronchiole wall, they are called tumorlets. If there is a relationship of 3 or
more airways, the diagnosis of DPNECH can be indicated radiologically6. It has been reported that the use of
somatostatin is beneficial in the treatment of these lesions, which are
considered premalignant by WHO9.
In the case report of Inomata et al.10, in a patient who
underwent chemotherapy for bilateral pulmonary nodules and a mass diagnosed as
primary adenocarcinoma in the right upper lobe, right upper lobectomy and right
lower lobe wedge resection were performed due to regression in the primary
tumor but no regression in the nodules and DIPNECH was diagnosed from nodules other
than the primary tumor.
4. Conclusion
PA and DIPNECH are two different entities and their coexistence is rare. DIPNECH, which is also associated with other diagnoses, can often be detected histopathologically in carcinoid tumors and surgical resection is the appropriate treatment method as we have stated in our study.
5. References
- Sagan D, Gozdziuk K. Surgery for
pulmonary aspergilloma in immunocompetent patients: no benefit from adjuvant
antifungal pharmacotherapy. Ann Thorac Surg. 2010;89(5): 1603-1610.
- Jiang C, Dai J, Bao Y, et al.
Surgical treatment of pulmonary aspergilloma: A 13-year experience from a
single clinical center. Ann Thorac Surg. 2022;114(1): 311-318.
- Denning DW, Cadranel J,
Beigelman-Aubry C, et al. European Society for Clinical Microbiology and
Infectious Diseases and European Respiratory Society. Chronic pulmonary
aspergillosis: rationale and clinical guidelines for diagnosis and management.
Eur Respir J. 2016;47(1): 45-68.
- Moskovljevic D, Colic N, Dimkic-Milenkovic
A, et al. Incidental finding of pulmonary tumorlet in a case of surgically
treated bronchiectatic cavity superimposed by aspergilloma. Srpski arhiv za
celokupno lekarstvo. 2022;150(11-12): 690-693.
- Lejay A, Falcoz PE, Santelmo N, et
al. Surgery for aspergilloma: time trend towards improved results? Interact
Cardiovasc Thorac Surg. 2011;13(4): 392-395.
- Ramirez RA, Cass AS, Das S, et al. A
multidisciplinary approach to the work up and management of pulmonary carcinoid
tumors and DIPNECH: a narrative review. Transl Lung Cancer Res. 2022;11(12): 2567-2587.
- Chassagnon G, Favelle O,
Marchand-Adam S, et al. DIPNECH: when to suggest this diagnosis on CT. Clin
Radiol. 2015;70(3): 317-325.
- Yazgan S, Gursoy S, Turk F, et al.
Conjunction of a fungus ball and a pulmonary tumourlet in a bronchiectatic
cavity. Korean J Thorac Cardiovasc Surg. 2018;51(2): 138-141.
- O'Brien C, Duignan JA, Gleeson M, et al. Quantitative Airway Assessment of Diffuse Idiopathic Pulmonary Neuroendocrine Cell Hyperplasia (DIPNECH) on CT as a Novel Biomarker. Diagnostics (Basel). 2022;12(12): 3096.
- Inomata S, Matsumura Y, Kobayashi Y, et al. Lung adenocarcinoma coexisting with diffuse idiopathic pulmonary neuroendocrine cell hyperplasia manifesting as multiple pulmonary nodules: A case report. Thoracic Cancer. 2022;13(21): 3076-3079.