Larvicidal Activity of Sweet Wormwood (Artemisia annua L.) Extracts on Aedes aegypti (Tiger Mosquito)
1.
Introduction
Artemisia
annua (Family-Asteraceae) has been used in traditional medicine
for treating fever and malaria. There are several species of Artemisia known as aromatic fragrance
plants that have a characteristic scent and taste1. The herb
of Artemisia has been used medicinally
to treat fevers for more than 2,000 years and to treat malaria for more than
1,000 years in China. Artemisia used in Chinese traditional medicine for
centuries, is today considered part of the solution where malaria has become
resistant to other medicines. Artemisinin-based combination therapies (ACTs)
have been recommended by World Health Organization (WHO) since 2001 in all
countries where falciparum malaria -
the most resistant form of the disease - is endemic2. Zhang et
al. 3 also proved that Artemisia showed the strongest biological
activity in July, and found that the acaricidal activity varied significantly
with the development of the individual plant. Mosquitoes have long been
known for their importance as vectors of diseases4. Despite
their small size, mosquitoes are of economic and medical importance. The genera
Aedes, Anopheles, and Culex are
important vectors of mosquito-borne diseases worldwide. Mosquito-borne diseases
cause economic loss and are commonly found in tropical rather than temperate
region5. Aedes aegypti, the
primary carrier of the dengue virus, which is predominant in tropical regions,
also transmits yellow fever in Africa and South America6. Cases
of dengue fever and dengue hemorrhagic fever have increased every year and
resulted in high number of deaths in Malaysia7. World
Health Organization also reported that mosquitoes are one of the deadliest
insects in the world. Their ability to carry and spread disease to humans
causes millions of death every year. In 2015, malaria alone caused 438, 000
deaths. The worldwide incidence of dengue has risen 30-fold in the past 30 years,
and more countries are reporting first outbreak of the disease. Zika, dengue,
Chikungunya, yellow fever are all transmitted to humans by the Aedes aegypti mosquito, according to the
World Health Organization8. Thus, this study was to evaluate the larvicidal activity of the
leaves of sweet wormwood (Artemisia annua
L.) on Aedes aegypti mosquito.
2.
Materials and Methods
2.1. Study area
The larvicidal activities of sweet
wormwood (Artemisia annua L.) on Aedes aegypti was carried out in the
Insectary of the Department of Science Laboratory Technology, University of
Jos, Nigeria (Latitude 09°57’ 01’’N and Longitude 08°53’ 21’’ E).
2.2. Collection of
Plant Materials
The leaves of A. annua were collected from the Botanical Garden of the Department
of Plant Science and Biotechnology, University of Jos, Nigeria.
2.3. Preparation of Plant Extracts
The collected leaves were dried at
room temperature (27-37°C) for 10 -15 days. The dried leaves were crushed using
electric blender. Twenty four grams (24 g) of the powdered leaves were orderly
poured into three different dried and clean reagent bottles containing 200 ml
of ethanol, distilled water and methanol, respectively. The samples were placed
on a mechanical shaker (1000 rmm/min) for 3 hrs, after which the samples were
left over night to settle and filtered the next day using funnel and filter
paper to obtain the extracts. The extracts were then used to test for
larvicidal activities on Aedes aegypti larvae.
2.4. Collection of Mosquito Larvae
Larvae of Ae. aegypti were collected from abandoned flower pots in Bauchi
road campus of the University of Jos. The larvae were collected using standard
dipping technique. The dipper was lowered at an angle of 450 to
minimize disruption and the top of the water was skimmed so as to cause the
nearby larvae to flow into the dipper. Care was taken not to spill water when
raising the dipper from the water. However, if the flower pots have emerging
vegetation, the water was disturbed so as to cause the larvae to swim
downwards. Some of the vegetation were then removed using the dipper to create
a clear spot for sampling. Larvae collected were transferred into a gallon for
transportation to the insectary.
2.5. Larvicidal Bioassay
Concentration of 0.1 ml, 0.2 ml and
0.3 ml of extracts were tested. Two replications were done to ensure the
validity of result. The larval mortality bioassay was carried out according to
the test method for larval susceptibility proposed by the World Health
Organization9. Twenty five fourth instar larvae of Ae. aegypti were placed in plastic bowls containing 40 ml of
aqueous suspension of tested material at various concentrations. A set of
control experiments using only the solvents were also set up. Mortality was
recorded from first fifteen minutes to 24 hours of exposure and the larvae were
not starved of food over this period.
Dose
responses of larvicidal bioassay
The numbers of death larvae were
counted from the first 15 minutes to 24 hours of exposure. The LC50
was calculated using probit analysis to find out the acute toxicity of the
extracts.
Statistical
Analyses
Data was analyzed using R Console
software (Version 3.2.2). The observed mortality and proportions of observed
mortality of Aedes aegypti larvae in
relation to different concentrations of solvents used to extract the leaves of Artemisia annua were compared using
Pearson’s Chi-squared test and the difference between the three solvents were
compared using one way ANOVA. Result from the acute toxicity test using
different concentrations of Artemisia
annua extracts were subjected to probit analysis to determine the LC50
values for the different solvents used in the extraction of the plant. The
p-values <0.05 were considered statistically significant.
2.6. Corrected mortality: mortality
was corrected using Abbott’s formula.
E’= [(E-C)/ (100-C)] x100
Where E is the (uncorrected)
exposure mortality expressed in percentage and C is the control mortality
expressed in percentage.
3.
Results
Determination of the
acute toxicity of extract of A. annua
leaves on larvae of Aedas. aegypti
Ethanol: The acute toxicity of different
concentrations of ethanol extracts of A.
annua against larvae of Ae. aegypti
show a significant difference (c2 = 6, df = 2, P =
0.04979) in the mortality rate of Ae.
aegypti larvae at 0.1 ml, 0.2 ml and 0.3 ml respectively (Figure 1). The
result revealed that there was 83.33% mortality at 0.1 ml, 94% mortality at 0.2
ml after 105 minutes and 96.66% mortality at 0.3 ml after 45 minutes (Figure 1).
Methanol: There was a
significant difference (c2 = 4, df = 2, P =
0.03451) in the mortality rate of larvae of Ae.aegypti
in relation to concentrations of methanolic extracts of A.annua (Figure 2). The breakdown of the
results showed 70% mortality at 0.1 ml after 24 hours, 75% mortality at 0.2 ml
after 24 hours and 95% mortality at 0.3 ml after 24 hours, respectively (Figure 2).
Water: There was no significant difference (c2 =4, df= 2, P = 0.1353) in the mortality rate of larvae of Ae. aegypti in relation to
concentrations of water extracts of A.annua
(Figure 3).The results depicted 76% mortality at 0.1 ml, 73.33% mortality
at 0.2 ml and 86.66% mortality at 0.3 ml after 24 hours, respectively (Figure
3).
Figure 1: Mortality rate of
Larvae of Ae. aegypti in relation to concentrations of ethanolic extract.
Figure 2: Mortality rate of
Larvae of Ae. aegypti in relation to concentrations of methanolic extract.
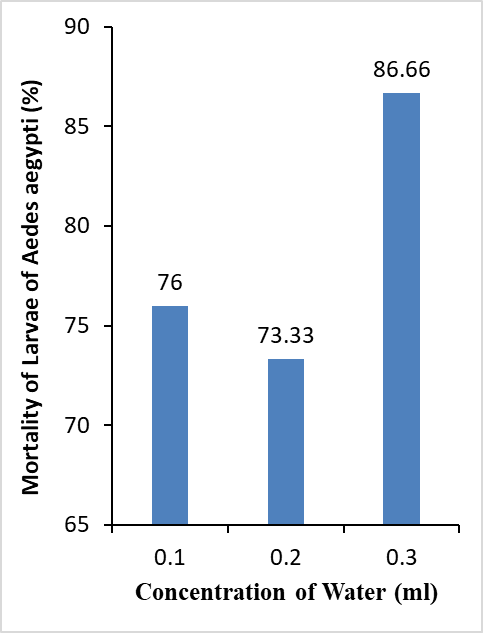
Figure 3: Mortality rate of
Larvae of Ae. aegypti in relation to concentrations of Water extract.
Determination
of the Lethal Concentration (Lc50) of the plant extract on larvae of
Ae. aegypti
The
larvicidal activity of Ethanol extracts of A.
annua gave an LC50 value of 0.131 ml (Table 1) indicating that ethanol extracts of A. annua exhibit larvicidal activity against Ae. aegypti larvae with a 24 hours LC50 value of 0.131
ml. However, the larvicidal activity of water extracts of A. annua recorded an LC50 value of 0.251 ml (Table1)
demonstrating that water extracts of A.
annua exhibit larvicidal activity against Ae. aegypti larvae with a 24 hours LC50 value of 0.25
ml. The larvicidal activity of methanol extracts of A. annua recorded an LC50 value of 0.602 ml (Table 1)
exhibiting larvicidal activity against Ae.
aegypti larvae with a 24 hours LC50 value of 0.602 ml.
Table 1: Larvicidal activity of extracts of A. annua against Ae. Aegypti.
|
Solvent |
LC50 (ml) |
Slope ± SD Chi-square |
χ2 |
Df |
P value |
|
Ethanol |
0.131 |
0.65 ±
0.620 |
6 |
2 |
0.0497 |
|
Methanol |
0.602 |
0.51 ±
0.351 |
4 |
2 |
0.0345 |
|
Water |
0.251 |
0.42 ±
0.247 |
4 |
2 |
0.1353 |
Between
solvents
χ2= 6.592, df =4, P =0.159
4.
Discussion
4.1. Determination of acute toxicity of
ethanol, methanol and water extracts of Artemisia
annua leaves against larvae of Aedes
aeqypti
In this
study, the significant difference in the acute toxicity of different
concentrations of ethanol and methanol extracts of A. annua against larvae of Ae.
aegypti (Figures 1 and 2) could be attributed to the analytical grade of
the solvent used which is in agreement with the report of Ngwamah et al.10 who worked on
Comparative insecticidal activity of five Nigerian plant species against
mosquito vectors in Yola, Adamawa state, Nigeria and reported a significant
difference among methanol and petroleum ether extracts. The high mortality rate
observed at 0.3 ml for ethanol and methanol after 24 hours of exposure is due
to high concentration of the extract and this is consistent with the findings
of Naimah11 who worked on the larvicidal effect of Artemisia
annua (Asterales: asteraceae) against the dengue fever mosquito vector Aedes
aegypti (Diptera: Culicidae) and reported a high mortality rate of the
larvae of Ae. aegpti at low
concentrations of ethanol after 24 hours of exposure. Similar findings were
also by Musa
et al.12
who studied the effect of aqueous and methanolic leaf extracts of A. conyzoides L and Guiera senegalensis L. against mosquito larvae in Zaria using
different concentrations of 50,100,200 and 400 ppm, and reported 100%
mortalities after 24 hours post treatment. The non-significant difference in
the mortality rate of larvae of Ae.
aegypti in relation to concentrations of water extracts of A. annua (Figure 3) in this study was
consistent with the findings of Njila et
al.13 who worked on the potency
of goat weed (Ageratum conyzoides L.)
to Culex quiquefasciatus larvae and
adults and revealed that water does not contain any chemical, it is mild and
inactive and polar in nature. However, the high mortality rate at 0.3 ml
concentration after 24 hours exposure (Figure 3) is similar to the findings of
Paulo et al.14 working on larvicidal activity of water
extract of Moringa oleifera seeds
against Aedes aegypti and its
toxicity upon laboratory animals reported 99.2% larvae mortality within 24
hours at 5200 microg/mL. Hazrat et al15.
who work on larvicidal activity of different plant extracts at different
concentrations against 3rd instar larvae of Aedes
albopictus, also reported high larvae mortality of 82% in Clonorchis sinensis, 58% in Myristica fragrans, 70% in Matricaria chamomilla, 62% in Mentha spicata and 67% in Zingiber officinale at 800ppm after 24
hours of exposure.
4.2. Determination of Lethal Concentration (Lc50)
of the leaves extract of Artemisia annua
on larvae of Aedes aegypti
Ethanol, methanol and water extract of Artemisia annua were tested for
larvicidal activity against larvae of Aedes
aegypti mosquito. The larvicidal activites of ethanol extract of A. annua against larvae of Ae. aegypti shows larvicidal activity at
LC50 value of 0.131 ml. Modise and Ashafa16 reported similar larvicidal activity of Foeniculum vulgare against Culex quinquefasciatus mosquitoes at LC50 value of 0.10mg/mL
after 24 hours of exposure. Naimah11
also reported that ethanol extracts of A.
annua exhibit toxicity against larvae of Ae. aegypti at LC50 value of 120.37 ppm after 24 hours
of exposure. The larvicidal activities of methanol extract of A.annua shows larvicidal activity
against larvae of Ae. aegypti at LC50
value of 0.60ml. Njila et al.13 recorded similar larvicidal activity of methanolic
extracts of Ageratum conyzoides at LC50
value of 1.09 ml after 24 hours of exposure. Extract of
Cymbopogon citrates (lemon grass)
exhibit the same level of or stronger larvicidal activity against Cx. quinquefasciatus at LC50
value of 3.495 g/l and 2.852 g/l17.
The water extract gave the LC50 value of 0.25 ml indicating that the
water extract is potent and exhibit larvicidal activity against larvae of Ae. aegypti at LC50 value of
0.25 ml. Japheth et al.18
reported similar larvicidal activity of Zanthoxylum
gilletii essential oil extracted by hydro-distillation against Anopheles gambiae at LC50 of
0.005773 mg/ml after 24 hours of exposure. The water extracts of Moringa oleifera seeds was tested
against Aedes aegypti larvae with LC50
value of 1260 uglml14. It was
observed that the water extract of Moringa
oleifera seeds were relatively toxic to larvae of Ae. aegypti. Similar finding was made by Njila et al13. who recorded LC50 value of
1.38 ml of the water extract of Ageratum
conyzoides on larvae of Culex
quinquefascatus.
5.
Conclusion
Plants are rich source of bioactive organic
chemicals with an advantage over synthetic pesticides since they are less
toxic, less prone to development of resistance and with easy biodegradability.
The percentage mortalities observed in this study for ethanol and methanol leaf
extracts of Artemisia annua shows significant difference and have the
highest percentage mortality against Ae. aegypti larvae than water which
shows no significant difference. Therefore different plants exhibit different
level of potency against different mosquito species when extracted with
different types of solvents. Lethal concentration (LC50) values
obtained in this study depicted that ethanol extract shows the strongest effect
with an LC50 of 0.13 ml, water extract with the LC50 of
0.25 ml and methanol extract with the LC50 of 0.60 ml. This reveals
that the leaves of Artemisia annua are very good, highly effective and a
larvicide that is less harmful to the environment. The result of this study
also reveals that the ethanol extracts of A. annua is considered the
best in terms of LC50 as well as percentage mortality compared to
methanol and water in controlling the larvae of Ae. aegypti.
6.
Acknowledgements
We would like to thank Dr. Angela Ogbonna of the
Department of Plant Science and Biotechnology, University of Jos, Nigeria for
providing the Artemisia annua used for the research.
7.
References
- Bora KS, Sharma A. The genus Artemisia: a comprehensive review. Pharmaceutical Biology, 2011; 49: 101-109.
- https://www.who.int/mediacentre/news/notes/2007/np10/en/
- Zhang Y, Ding W, Zhao Z, et al.
Studies on Acaricidal Bioactivities of Artemisia
annua L. Extracts against Tetranychus
cinnabarinus Bois. (Acari: Tetranychidae). Agricultural Sciences in China, 2008;7: 577-584
- Wang Z, Kim
JR, Wang R, et al. Larvicidal activity of Cnidium
monnieri fruit coumarins and
structurally related compounds against insecticide-susceptible and insecticide
resistant Culexpipienspallens and Aedes aegypti.Pest Management Science, 2011;
68: 1041-1047.
- Adanan CR,
Zairi J, Ng KH. Efficacy and sub lethal effects of mosquito mats on Aedes aegypti and Culex quinquefasciatus (Diptera:
Culicidae). In Proceedings of the 5th
international conference on urban pests, 2005; 12: 55-88.
- Govindarajan
M, Mathivanan T, Elumalai K, et al. Mosquito larvicidal, ovicidal, and
repellent properties of botanical extracts against Anopheles
stephensi, Aedes aegypti, and Culex
quinquefasciatus (Diptera: Culicidae). Journal
of Parasitology Research, 2011; 109: 353-367.
- Lee YW, Zairi
J. Laboratory evaluation of Bacillus
thuringiensis H-14 against Aedes
aegypti. Tropical Biomedicine, 2005;
22: 5-10.
- https://www.who.int/neglected_diseases/vector_ecology/mosquito-borne-
/en/
- WHO. Instruction for determining the susceptibility
or resistance of mosquito larvae to insecticide. World Health Organization,
Geneva, Switzerland, 1981.
- Ngwamah JS,
Naphtali RS, Pukuma MS, et al. Comparative Insecticidal Activity of Five Nigerian Plant
Species against Mosquito Vectors in Yola, Adamawa State, Nigeria. International
Journal of Mosquito Research, 2018; 5: 131-141.
- Naimah AA.
Larvicidal effect of Artemisia annua (Asterales:
asteraceae) against the dengue fever mosquito vector Aedes aegypti (Diptera: Culicidae). International Journal of Mosquito Research,
2018; 5: 35-38.
- Musa AO, Sow
GJ, Nasir IF, et al. Effects of aqueous and methanolic leaf extracts of A. conyzoides L. and Guiera senegalensis L. against mosquito
larva in Zaira. Department of Biological Sciences & NBSP. Ahamadu Bello
University, Zaira. Journal of tropical
Biosciences, 2015; 10: 0795-3089.
- Njila HL,
Edozie B, Gushit JS, et al. The Potency of Goat Weed (Ageratum
conyzoides L.) to Culex
quinquefasciatus Larvae and Adults. Journal
of Laboratory Science 2018; 5: 61-71.
- Paulo MPF,
Ana FUC, Davi FF, et al. Larvicidal activity of the water extract of Moringa oleifera seeds against Aedes aegypti and its toxicity upon
laboratory animals. Anais da Academia
Brasileira de ciencias, 2009; 81: 207-216.
- Hazrat B,
Sumrin S, Sadrud D. Bio-Pesticides: New tool for Control of Aedes (Stegomyia)
albopictus (culicidae:Diptera) in Pakistan. Journal
of Arthropod Borne Diseases, 2017; 11: 278-285.
- Modise SA,
Ashafa AO. Larvicidal, Pupicidal and Insecticidal activities of Cosmos bipinnatus, Foeniculum vulgare and Tagetes minuta against Culex quinquefasciatus Mosquitoes. Tropical Journal of pharmaceutical Research
2016; 15: 965-972.
- Adakole JA, Adeyemi AFF. Larvicidal Effects of Cymbopogocitratus (Lemom grass) extract against Cx.quinquefasciatuslarvae (Diptera: Culicidae). International Journal of Applied Environmental sciences, 2012; 7: 187.
- Japheth OO, Josphat MC, John VM. Chemical composition
and larvicidal activity of Zanthoxylum
gilletii essential oil against Anopheles
gambiae. African Journal of
Biotechnology, 2014; 13: 2175-2180.