Explainable AI Frameworks for Patient-Level Claims Data Analytics
Abstract
Artificial intelligence-driven
analytics applied to patient‑level healthcare claims data have become central
to pharmaceutical decision‑making, enabling prediction of treatment adherence,
therapy switching and longitudinal patient behavior. However, the increasing
reliance on complex machine learning models has introduced significant concerns
related to transparency, interpretability and trust-particularly in regulated
environments where decisions influence patient access, real‑world evidence
generation and strategic planning.
This study
proposes a comprehensive explainable
artificial intelligence (XAI) framework tailored for patient‑level
claims data analytics in pharmaceutical contexts. The framework integrates high‑performance
predictive modeling with post‑hoc interpretability techniques, including
feature attribution and local explanation methods, to ensure model outputs
remain transparent, clinically meaningful and decision‑relevant. In addition, a
dedicated governance and auditability layer is embedded to support regulatory
compliance, traceability and responsible AI deployment across the analytics
lifecycle.
By structuring
explainability as a first‑class system capability-rather than an afterthought-the
proposed framework enables stakeholders to understand not only what predictions are generated, but why they are produced and how they can be acted upon. This work
contributes to the growing body of trustworthy AI research by offering a
scalable, enterprise‑ready approach that balances predictive performance with
interpretability, supporting real‑world evidence development and patient‑centric
pharmaceutical analytics.
Keywords: Explainable AI, Healthcare Claims
Data, Patient Behavior Analytics, Model Interpretability, Pharmaceutical
Analytics, Treatment Adherence, Real World Evidence
1.
Introduction
Healthcare claims
data have emerged as one of the most valuable sources of real‑world data for
understanding patient‑level healthcare utilization, treatment pathways and cost
dynamics across large populations. Generated through insurance billing and
administrative processes, claims datasets capture detailed longitudinal records
of diagnoses, procedures, prescription activity and provider interactions,
enabling sustained observation of patient behavior over time1. As a result, claims data play a
foundational role in pharmaceutical analytics, health economics, outcomes
research and real‑world evidence (RWE) generation.
The growing scale and
availability of patient‑level claims data have accelerated the adoption of
advanced analytical methods aimed at predicting clinically and commercially
relevant outcomes such as medication adherence, treatment switching,
hospitalization risk and disease progression2.
Machine learning models have become central to these efforts, allowing
pharmaceutical organizations to identify complex patterns within high‑dimensional
claims datasets that are difficult to capture using traditional statistical
techniques3. In practice, such
models support critical decision‑making processes, including patient
segmentation, intervention targeting, market access planning and post‑market
effectiveness assessment.
Despite these
advances, the increasing use of sophisticated machine learning techniques has
introduced a major challenge: the
opacity of black‑box models. Many high‑performing approaches, including
ensemble methods and neural networks, provide limited insight into the
reasoning behind their predictions4,5.
In pharmaceutical and healthcare settings-where analytical outputs directly
influence patient care strategies, regulatory submissions and payer engagement-this
lack of transparency can undermine trust, hinder adoption and complicate
compliance requirements.
Explainable
artificial intelligence (XAI) has emerged as a critical response to these
concerns, aiming to make complex model behavior understandable to human
stakeholders. Techniques such as feature attribution, surrogate models and
local explanations offer mechanisms to interpret predictions without
sacrificing model performance6.
However, in many applied settings, explainability is implemented in an ad hoc
or fragmented manner, detached from broader analytics workflows, governance
standards and operational decision‑making needs.
In pharmaceutical
claims analytics, this fragmentation is particularly problematic. Existing
approaches often focus narrowly on model performance or explanation techniques
in isolation, failing to address the full lifecycle of AI deployment-from data
preprocessing and modeling to explanation generation, auditability and
regulatory defensibility. Moreover, current research rarely frames
explainability as a decision‑enabling
capability, aligned with real‑world pharmaceutical use cases such as
adherence intervention design, treatment optimization and evidence‑based
strategy development.
To address these
gaps, this study proposes a structured
explainable AI framework specifically designed for patient‑level claims data analytics
in pharmaceutical environments. The framework integrates predictive
modeling with interpretable explanation techniques and embeds governance
mechanisms that ensure transparency, traceability and compliance across the
analytics pipeline7. The contributions
of this work are threefold:
·
It introduces a unified architectural
framework combining data processing, modeling, explainability and governance;
·
It demonstrates how explainability
enhances the usability and trustworthiness of AI‑driven insights in pharmaceutical
decision‑making; and
·
It provides a scalable foundation for
responsible AI adoption in real‑world evidence and patient‑centric analytics.
2. Literature Review
2.1. Overview of explainable
AI techniques
Explainable
artificial intelligence (XAI) has emerged as a critical area of research in
response to the growing adoption of complex machine learning models whose
internal decision processes are difficult to interpret. High‑performing
algorithms-such as ensemble methods and deep neural networks-often operate as
black boxes, creating challenges in domains where decision transparency is
essential4. To address this gap,
a range of explainability techniques has been developed to provide insight into
model behavior without sacrificing predictive performance.
Among the most widely
adopted methods are Local Interpretable
Model‑Agnostic Explanations (LIME) and Shapley Additive Explanations (SHAP). LIME generates local,
instance‑level explanations by approximating the behavior of complex models
with simpler surrogate models in the vicinity of a specific prediction2. SHAP, grounded in cooperative game
theory, assigns contribution values to individual features in a manner that is
both consistent and theoretically sound6.
Other commonly used techniques include partial dependence plots (PDPs),
counterfactual explanations and rule‑based surrogate models, each offering
varying levels of interpretability, scope (local vs. global) and computational
complexity.
Collectively, these approaches
aim to bridge the gap between predictive accuracy and human‑understandable
reasoning. However, their practical value depends not only on theoretical
soundness but also on their ability to support real decision workflows-particularly
in regulated and high‑impact environments such as healthcare and
pharmaceuticals.
2.2. Explainable AI
in healthcare applications
The application of
explainable AI within healthcare has expanded rapidly across domains such as
clinical risk prediction, diagnostic support, disease prognosis and treatment
planning. In these contexts, interpretability is essential for validating model
outputs, ensuring clinical relevance and supporting trust among practitioners
and patients alike8. XAI
techniques enable stakeholders to identify the key drivers behind predictions-such
as risk factors, treatment indicators or utilization patterns-thereby
supporting more transparent and accountable decision‑making.
Recent studies
demonstrate that embedding explainability within healthcare AI systems improves
model validation, facilitates bias detection and enhances adoption among
clinicians and healthcare administrators9.
Importantly, explainability also plays a critical role in aligning AI outputs
with established clinical knowledge, helping domain experts assess whether
predictions are plausible and actionable rather than statistically opaque.
Despite this
progress, much of the existing healthcare XAI literature focuses on clinical or
imaging‑centric applications. Comparatively less attention has been given to real‑world administrative data, such
as claims, where interpretability must support not only clinicians but also
pharmaceutical decision‑makers, regulators and policy stakeholders.
2.3. Claims data
analytics and patient-level prediction
Healthcare claims
data analytics centers on extracting insight from large‑scale administrative
records that capture patient interactions with the healthcare system over time.
Claims‑based machine learning models are widely used to predict outcomes such
as medication adherence, treatment switching, hospitalization likelihood and
cost trajectories10. These
predictions are particularly valuable in pharmaceutical contexts, where
understanding patient‑level dynamics informs intervention strategies, post‑market
evaluation and real‑world evidence generation.
The high
dimensionality and longitudinal structure of claims data often necessitate the
use of advanced machine learning models capable of capturing non‑linear
relationships and temporal dependencies. While such models deliver strong
predictive performance, they frequently reduce interpretability-limiting
visibility into how patient attributes, utilization patterns and treatment
histories interact to produce specific outcomes. This trade‑off becomes
especially problematic in pharmaceutical decision‑making, where predictions
must be explainable to support regulatory submissions, payer engagement and
patient‑centric program design.
2.4. Limitations of existing
approaches
Despite the maturation
of XAI techniques, several limitations persist in their application to
healthcare claims analytics. First, many interpretability methods are applied post hoc and operate independently of
the broader analytics lifecycle. As a result, explanations may not fully
capture the internal logic of predictive models or may vary significantly
across different interpretation techniques, leading to ambiguity in decision
support5.
Second, existing
approaches often lack integration with governance and compliance mechanisms. In
regulated environments, explainability must extend beyond feature attribution
to include traceability of data inputs, model versions, assumptions and outputs
over time. Current studies frequently emphasize model accuracy and
interpretability in isolation, without addressing auditability, reproducibility
or alignment with real‑world evidence standards7.
Finally, many XAI
implementations are not designed with enterprise scalability in mind.
Computational cost, explanation instability and limited support for
longitudinal analysis can hinder adoption in real‑world pharmaceutical
analytics platforms that operate at population scale.
2.5. Research gap
synthesis
The literature
reveals a clear gap in the development of integrated explainable AI frameworks tailored to patient‑level
healthcare claims data analytics. While individual explainability techniques
such as SHAP and LIME are well established, their application remains
fragmented and insufficiently connected to governance, auditability and
decision workflows in pharmaceutical environments.
Specifically, there
is a lack of end‑to‑end frameworks that unify data preprocessing, predictive
modeling, explainability, continuous monitoring and regulatory compliance
within a single operational architecture. Addressing this gap requires moving
beyond ad hoc interpretation of black‑box models toward system‑level explainability, where transparency is embedded
throughout the analytics lifecycle and aligned with real‑world pharmaceutical
decision‑making.
This research
responds to that need by proposing a comprehensive explainable AI framework
that balances predictive performance with interpretability and governance,
supporting trustworthy, patient‑centric and enterprise‑ready claims data
analytics (Table 1).
Table 1:
Summary of Existing XAI Methods in
Healthcare.
|
Method |
Scope |
Primary Output |
Best For (Decision Use) |
Strengths |
Limitations / Cautions |
|
LIME |
Local |
Local
surrogate explanation |
Why this patient was flagged
(case review, outreach decisions) |
Model‑agnostic;
intuitive |
Can
be unstable across runs |
|
SHAP |
Global + Local |
Feature
contributions (Shapley values) |
Global drivers + patient‑level rationale
(governed insights, stakeholder trust) |
Consistent;
theoretically grounded |
Computationally
intensive |
|
PDP |
Global |
Marginal
feature effect curve |
Feature effect intuition
(policy/threshold discussions) |
Easy
to visualize |
Assumes
feature independence |
|
Counterfactuals |
Local |
Minimal changes for
different outcome |
Actionability (what changes could reduce switching
/ non‑adherence) |
Actionable;
user-friendly |
Hard for complex
constraints |
|
Rule‑based models |
Global |
Interpretable
rules |
High transparency requirements |
Highly
transparent |
Lower
predictive accuracy |
3. Theoretical
Foundation
3.1. Interpretability
vs Explainability
Interpretability and
explainability are closely related but conceptually distinct constructs in
artificial intelligence. Interpretability
refers to the inherent transparency of a model’s internal mechanics, allowing
users to directly understand how inputs are transformed into outputs. Models
such as linear regression or decision trees are often considered interpretable
because their structure and decision logic can be examined directly10. However, these models may lack the
expressive power required to capture complex, non‑linear relationships common
in high‑dimensional healthcare claims data.
Explainability, by contrast, addresses the challenge
of understanding complex or opaque models by applying external techniques that
make their behavior intelligible without exposing internal structure11. In practice, explainability enables
stakeholders to reason about predictions from high‑performing models-such as
gradient boosting machines and neural networks-using post hoc interpretation
methods. This distinction is particularly relevant in pharmaceutical analytics,
where predictive accuracy alone is insufficient unless outputs can be
explained, validated and defended across medical, regulatory and business
contexts.
For patient‑level
claims analytics, explainability therefore represents a strategic capability rather than a technical enhancement. It
enables organizations to retain advanced modeling performance while meeting the
transparency and accountability expectations required for real‑world evidence
(RWE) generation and decision support.
3.2. Trust, Fairness
and Accountability in healthcare AI
Trust is a foundational
requirement for the adoption of AI systems in healthcare and pharmaceutical
decision‑making. Unlike consumer analytics, healthcare AI influences decisions
that affect patient access, treatment pathways and population‑level outcomes.
Explainability contributes directly to trust by enabling stakeholders to assess
whether model predictions are plausible, consistent with domain knowledge and
supported by meaningful data signals8.
Fairness is a critical related dimension, particularly
in patient‑level analytics where biases embedded in historical claims data may
lead to unequal outcomes across demographic or clinical subgroups. Explainable
AI techniques support fairness assessment by revealing feature contributions
and highlighting whether sensitive attributes or proxies disproportionately
influence predictions9. Without
explainability, such biases may remain hidden, undermining both ethical
responsibility and regulatory credibility.
Accountability refers to the ability to trace, justify
and audit decisions generated by AI systems. In pharmaceutical environments,
accountability extends beyond model development to include how predictions are
used in downstream processes such as patient targeting, adherence interventions
and evidence planning. Explainability mechanisms provide the transparency
required to document why decisions were made and to support internal
governance, external review and regulatory scrutiny.
Together, trust,
fairness and accountability form a triad
of responsible AI principles that are especially relevant in claims‑based
analytics, where decisions must be both scalable and defensible.
3.3. Regulatory and governance
perspectives
The application of AI
to healthcare and pharmaceutical analytics operates within an increasingly
stringent regulatory and governance landscape. Regulatory bodies and payers
place growing emphasis on the credibility, reproducibility and transparency of
real‑world evidence used to support decision‑making. As AI models become more
deeply embedded in evidence generation and strategy formulation, the ability to
explain and audit their outputs becomes essential. Governance frameworks play a
central role in operationalizing explainability within this context. Effective
governance encompasses model documentation, version control, performance
monitoring, bias detection and traceability of data inputs and analytical
assumptions7. In patient‑level
claims analytics, governance ensures that predictions and explanations can be
reviewed, reproduced and validated over time—rather than existing as one‑off
analytical artifacts.
Importantly,
explainability strengthens governance by linking technical model behavior to documented decision logic. It
enables organizations to demonstrate not only what a model predicts, but how
those predictions were derived and whether they align with established clinical
and business reasoning. This alignment is critical for supporting regulatory
submissions, payer engagement and responsible use of AI in population‑scale
analytics.
Within this
theoretical framework, explainability is therefore not treated as an optional
model add‑on, but as a core pillar of trustworthy, enterprise‑ready AI systems
in pharmaceutical claims analytics.
4. Proposed explainable
AI framework
4.1. Framework overview
(Architecture
and Design Principles)
The proposed
explainable AI framework is designed as a modular, layered enterprise architecture that integrates patient‑level
healthcare claims data, predictive modeling, explainability mechanisms and
governance controls into a single, cohesive analytics system. The primary
objective of the framework is to ensure that predictions related to patient
behavior-such as treatment switching, medication adherence and healthcare
utilization-are not only accurate but also transparent, auditable and decision‑ready5.
Unlike ad hoc XAI
implementations that focus narrowly on model interpretation, this framework
adopts a governance‑by‑design approach,
where explainability, traceability and accountability are embedded across the
full analytics lifecycle. The architecture supports end‑to‑end workflows,
beginning with claims data ingestion and extending through prediction,
explanation, monitoring and regulatory defensibility. Each layer performs a
well‑defined function while maintaining interoperability through standardized data
flows, metadata capture and feedback mechanisms.
From an enterprise
deployment perspective, the framework is designed to operate at population scale, support longitudinal
analytics and integrate seamlessly into pharmaceutical decision‑making
environments that require reproducibility, role‑based access and auditability.
The layered design enables organizations to evolve individual components-such
as models or explanation techniques-without destabilizing the overall system.
4.2. Framework components
4.2.1. Data layer: Claims
data ingestion and preprocessing
The data layer forms the foundation of the
framework and is responsible for ingesting, standardizing and preparing patient‑level
healthcare claims data from multiple sources, including medical claims,
pharmacy claims and provider billing records3.
Given the longitudinal and heterogeneous nature of claims data, this layer
performs critical preprocessing tasks such as data cleaning, normalization,
feature extraction and temporal alignment.
Key data elements
include diagnosis codes, procedure codes, prescription histories, refill
timing, healthcare utilization metrics and demographic attributes. To support
downstream explainability and governance, the data layer also captures metadata and lineage information,
enabling traceability of how raw records are transformed into analytical
features. This foundation is essential for ensuring consistency across model
training, prediction generation and explanation outputs.
4.2.2. AI modeling layer: Predictive analytics
at patient scale
The AI modeling layer applies machine
learning algorithms to generate patient‑level predictions aligned with
pharmaceutical analytics use cases, such as medication adherence risk,
likelihood of treatment switching and hospitalization propensity. Models
including gradient boosting methods (e.g., XGBoost), random forests and neural
networks are used to capture complex, non‑linear relationships inherent in high‑dimensional
claims data12.
Model selection
within this layer balances predictive performance with explainability
compatibility and operational robustness. Importantly, models are not treated
as isolated artifacts; they are deployed within a controlled lifecycle that
includes versioning, performance monitoring and validation checkpoints. This
design supports repeatability and ensures that model outputs remain consistent
and defensible over time, especially when used in real‑world evidence
generation or strategic decision‑making.
4.2.3. Explainability layer: Structured transparency
and model insight
The explainability layer operationalizes
transparency by integrating post hoc interpretability techniques as a native
system capability rather than an external analytic step. Techniques such as
SHAP and LIME are used to generate both global
explanations (e.g., feature importance trends across populations) and local explanations (e.g., drivers of
individual patient predictions).
SHAP values quantify
the contribution of each feature to a prediction in a consistent and theoretically
grounded manner, enabling population‑level insight and cross‑model comparison6. LIME complements this by providing
localized explanations for individual predictions, supporting case‑level
reasoning and validation. Together, these methods enable stakeholders to
understand why predictions are
produced and assess whether they align with clinical knowledge, business logic
and ethical expectations.
Crucially,
explanation artifacts are treated as first‑class
outputs, stored, versioned and linked to their corresponding data inputs
and model versions. This design ensures explanations can be audited, reproduced
and reviewed—an essential requirement for enterprise adoption and regulatory
confidence.
4.2.4. Governance layer: Accountability,
Compliance and monitoring
The governance layer enforces
accountability and regulatory alignment across the analytics lifecycle. It
establishes mechanisms for audit trails, bias detection, performance tracking
and documentation of analytical assumptions. This layer ensures that every
prediction and explanation can be traced back to its underlying data, model
configuration and execution context7.
Governance functions
include continuous monitoring of model drift, validation of explanation
stability and enforcement of compliance standards related to data privacy and
real‑world evidence usage. Role‑based controls enable appropriate access to
predictions and explanations, supporting both internal governance and external
review. By embedding governance directly into the framework, explainability
becomes operational rather than aspirational-supporting trust at scale.
4.3. Feedback loop
for continuous learning and control
A key strength of the
proposed framework is its closed
feedback loop, which connects governance and performance monitoring back
to the data, modeling and explainability layers. Observed outcomes-such as
changes in patient behavior, intervention effectiveness or shifts in data
distributions-are fed back into the system to recalibrate models, refine
features and validate explanation relevance13.
This continuous
learning mechanism enables the framework to adapt to evolving healthcare
dynamics while maintaining analytical consistency and control. Importantly,
updates are governed rather than automatic: changes are logged, reviewed and
validated to ensure that adaptability does not compromise transparency or
trust. This balance between agility and governance is especially critical in
pharmaceutical environments, where both innovation and accountability are
required.
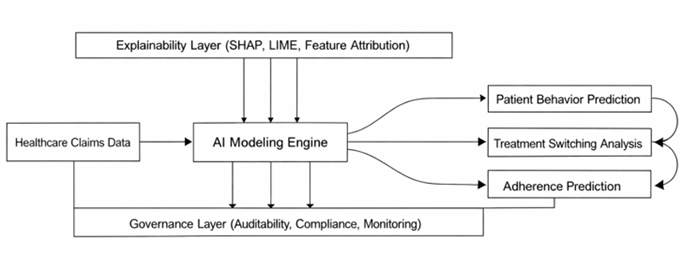
Figure 1:
Conceptual framework illustrating the
integration of healthcare claims data, AI modeling, explainability mechanisms
and governance layers for transparent patient-level analytics. The framework
highlights the flow from data ingestion to predictive outputs, including
patient behavior, treatment switching and adherence, with embedded
explainability and feedback loops for continuous model improvement.
Figure 2:
Layered architecture of the proposed
explainable AI system for healthcare claims analytics. The diagram presents the
hierarchical structure consisting of the data layer, AI modeling layer,
explainability layer and governance layer, with bidirectional data flow and
feedback mechanisms to support continuous learning, auditability and regulatory
compliance.
5. Research
Methodology
This study adopts a structured, decision‑oriented research
methodology to design and evaluate an explainable artificial
intelligence framework for patient‑level healthcare claims data analytics.
Rather than focusing exclusively on predictive accuracy, the methodology
emphasizes decision validity,
explainability quality and deployment readiness-criteria that are
essential for real‑world adoption in pharmaceutical analytics environments8.
5.1.
Data foundation and analytical context
The analysis is based
on longitudinal healthcare claims data,
integrating medical and pharmacy records that capture patient interactions
across diagnoses, prescriptions and healthcare utilization over time9. The dataset includes structured
attributes such as diagnosis codes, procedure codes, prescription histories,
refill timing and demographic characteristics, enabling end‑to‑end tracking of
patient treatment trajectories and behavioral patterns14.
From a pharmaceutical
analytics perspective, the longitudinal nature of claims data is particularly
critical because it allows for observation of temporal dynamics, including changes in adherence behavior,
treatment switching events and utilization patterns. These longitudinal
properties directly support real‑world evidence generation and patient‑centric
decision‑making rather than static, cross‑sectional analysis11.
5.2.
Feature engineering for explainability and decision relevance
Feature engineering
is performed with explicit consideration of interpretability and decision relevance, not solely predictive
power. Derived variables are constructed to represent clinically and
operationally meaningful dimensions of patient behavior and health status.
Medication adherence
is quantified using established measures such as proportion of days covered (PDC), providing interpretable
indicators of therapy compliance. Treatment switching is captured through
features reflecting the frequency, timing and sequence of regimen changes12. Clinical complexity is incorporated
using comorbidity indices derived from diagnosis codes, while healthcare
utilization is represented through inpatient admissions, outpatient visits and
pharmacy activity.
Temporal features are
engineered to capture trends and transitions in patient behavior over time,
allowing models to reflect evolving risk rather than static snapshots5. This feature design ensures that
downstream explanations can be meaningfully interpreted by domain experts and
aligned with pharmaceutical decision workflows.
5.3.
Predictive modeling with explainability compatibility
The predictive
modeling process involves the application and comparison of multiple machine learning algorithms
capable of handling high‑dimensional, structured claims data. Models evaluated
include gradient boosting (e.g., XGBoost), random forest classifiers and neural
network architectures, each selected for their ability to capture complex non‑linear
relationships12.
Model selection
criteria extend beyond performance metrics to include stability, explainability compatibility and operational robustness.
Gradient boosting models, in particular, are emphasized due to their strong
performance in structured data settings and compatibility with post‑hoc
explanation techniques. Models are trained, validated and versioned in a
controlled manner to support reproducibility and lifecycle management-both
critical for enterprise deployment.
5.4.
Explainability integration and evaluation
Explainability is
embedded directly into the analytical pipeline using post‑hoc interpretability techniques rather than being applied as
an external or ad hoc step. SHAP is employed to generate both global and local
explanations by quantifying the contribution of individual features to model
predictions in a consistent and theoretically grounded manner6,7.
Complementarily, LIME
is applied to produce localized, instance‑level explanations by approximating
model behavior in the vicinity of individual predictions2. Together, these techniques support
explanation at both population and patient levels-an essential requirement for
pharmaceutical analytics where decisions must be justified across cohorts as
well as individual cases.
Explainability
outputs are evaluated not only for interpretability, but also for:
·
Stability
across model runs and datasets.
·
Consistency
between explanation methods.
·
Plausibility
relative to clinical and domain knowledge.
This evaluation
ensures that explanations are reliable and actionable rather than purely
descriptive.
5.5.
Evaluation framework: Beyond model accuracy
The framework is
evaluated using a multi‑dimensional
assessment strategy that reflects real‑world pharmaceutical decision
requirements. Predictive performance is measured using standard classification
metrics, including accuracy, precision, recall and area under the receiver
operating characteristic curve. However, these metrics are treated as necessary
but insufficient conditions for deployment.
To assess decision validity, interpretability
evaluation focuses on whether explanations:
·
Identify clinically meaningful drivers
of behavior.
·
Remain consistent across patient
subgroups.
·
Support defensible intervention planning.
Trustworthiness is
evaluated through transparency, reproducibility and alignment with established
medical and pharmaceutical knowledge1.
This approach ensures that model outputs and explanations are suitable for
consumption by non‑technical stakeholders involved in patient programs,
evidence planning and strategic decision‑making.
5.6.
Enterprise deployment readiness
Beyond analytical
performance, the methodology explicitly considers enterprise deployment readiness. This includes assessing
computational scalability of explanation techniques, reproducibility of results
and compatibility with governance and compliance requirements.
Explainability
artifacts are treated as first‑class outputs-stored, versioned and linked to
specific data inputs and model configurations. Continuous monitoring mechanisms
are assumed to track model performance, explanation stability and drift over
time, supporting responsible deployment in dynamic healthcare environments
(Ahmad et al., 2018).
By aligning technical
evaluation with governance, trust and operational scalability, the methodology
ensures that the proposed framework is suitable not only for experimental
validation but also for real‑world pharmaceutical analytics platforms (Table
2).
Table 2: Dataset
Variables and Features.
|
Domain |
Feature Group |
Variable |
Description |
Data Type |
|
Demographic |
Patient Profile |
Age |
Patient age |
Numerical |
|
Demographic |
Patient Profile |
Gender |
Patient gender |
Categorical |
|
Clinical |
Clinical Condition |
Diagnosis Codes |
Disease classification (ICD) |
Categorical |
|
Treatment |
Therapy History |
Medication History |
Prescription records over time |
Sequential |
|
Behavioral |
Treatment Behavior |
Adherence Rate |
Proportion of days covered (PDC) |
Numerical |
|
Behavioral |
Treatment Behavior |
Switching Indicator |
Change in treatment regimen |
Binary |
|
Utilization |
Healthcare Utilization |
Hospital Visits |
Number of inpatient admissions |
Numerical |
|
Utilization |
Healthcare Utilization |
Outpatient Claims |
Frequency of outpatient services |
Numerical |
6. Experimental
Results and Analysis
6.1. Predictive performance as decision enablement
The predictive models
were evaluated using standard classification metrics, including accuracy,
precision, recall and area under the receiver operating characteristic curve
(AUC). While these metrics provide an essential baseline for assessing statistical
performance, they are interpreted here primarily in terms of decision enablement rather than
algorithmic comparison alone.
Among the evaluated
models, gradient boosting (XGBoost) demonstrated the strongest overall
performance, exhibiting high accuracy and robustness in predicting patient‑level
outcomes such as medication adherence and treatment switching. Neural network
models achieved comparable performance but required significantly higher
computational resources, while random forest models offered a balanced trade‑off
between performance stability and interpretability. Logistic regression,
although more transparent by design, exhibited lower predictive strength,
limiting its suitability for complex claims‑based decision scenarios.
From a pharmaceutical
analytics perspective, these findings indicate that high‑performing models are necessary but not sufficient. The
selected models must not only predict outcomes reliably, but also support
downstream explainability and governance requirements. Gradient boosting models
were therefore favored, as they provided strong predictive capability while
remaining compatible with explainability techniques essential for responsible
deployment.
6.2. Explainability outputs as decision
insights
Explainability analysis
played a central role in transforming model outputs into actionable decision insights. SHAP‑based
global explanations revealed a consistent set of high‑impact drivers across
models, including prior medication adherence, refill gaps, comorbidity burden
and healthcare utilization frequency. These features align closely with
established clinical and pharmaceutical understanding of patient behavior,
reinforcing confidence in the model’s reasoning.
At the population
level, explainability outputs enabled stakeholders to understand which factors systematically influence
outcomes, supporting cohort‑based planning and evidence development. At
the patient level, local explanations generated through LIME highlighted how
individual characteristics contributed to specific predictions, enabling case‑level
assessment and validation.
Rather than serving
as descriptive artifacts, explainability outputs were evaluated for decision relevance—specifically,
whether they:
·
Identified modifiable drivers suitable
for intervention.
·
Remained consistent across patient
subgroups.
·
Aligned with domain knowledge and real‑world
evidence expectations.
This evaluation
confirmed that explainability techniques meaningfully enhanced trust in model
outputs and supported their use in pharmaceutical decision workflows.
6.3. Scenario‑driven case analysis
6.3.1.
Treatment switching prediction as early warning
In treatment
switching prediction scenarios, the framework effectively identified patients
at elevated risk based on declining adherence patterns, widening refill gaps,
increasing comorbidity complexity and higher healthcare utilization.
Explainability outputs consistently showed that refill gap dynamics and recent
prescription instability were among the strongest contributors to switching
risk.
From a decision‑making
standpoint, these insights enable earlier
identification of at‑risk patients, allowing pharmaceutical teams to
intervene proactively-through patient support programs or evidence‑based
engagement strategies-rather than reacting after switching occurs. Importantly,
explanation transparency allowed stakeholders to distinguish between clinically
driven switching and behaviorally driven patterns, supporting more targeted
responses.
6.3.2.
Medication adherence risk stratification
In adherence
prediction scenarios, the model demonstrated high sensitivity in identifying
patients likely to become non‑adherent. SHAP analysis indicated that historical
adherence measures, medication complexity and demographic context were the
dominant contributors to risk.
These explainable
insights enable decision‑makers to move beyond binary risk flags toward interpretable risk stratification,
where interventions can be tailored based on why adherence risk is elevated. For example, interventions for
patients driven by regimen complexity may differ from those driven by access or
utilization instability. This differentiation is critical for designing
scalable, patient‑centric adherence strategies.
6.4.
Explainability versus black‑box performance trade‑offs
A comparison with non‑explainable
(“black‑box”) model deployments demonstrated that the integration of
explainability techniques did not
result in meaningful degradation of predictive performance. Instead,
explainability substantially improved transparency, validation capability and
stakeholder confidence without compromising accuracy6.
This finding
reinforces a key premise of the proposed framework: explainability and performance are not competing objectives in
well‑designed pharmaceutical analytics systems. When explainability is embedded
into the modeling and governance lifecycle, it strengthens decision quality
rather than constraining model capability.
6.5.
Implications for real‑world deployment
Beyond numerical
evaluation, the experimental analysis highlights how explainable AI frameworks
unlock practical value for real‑world pharmaceutical deployment. By linking
model predictions to interpretable drivers, the framework supports:
·
Auditable decision‑making aligned with
regulatory expectations
·
Cross‑functional adoption across
analytics, medical and strategy teams
·
Continuous monitoring and refinement of
models based on observed outcomes
These results
demonstrate that explainability transforms claims‑based analytics from
predictive outputs into defensible,
actionable decision intelligence, capable of operating at enterprise
scale (Tables 3 and 4).
Table 3:
Model Performance Comparison.
|
Model |
Accuracy |
Precision |
Recall |
AUC |
|
XGBoost |
0.89 |
0.87 |
0.85 |
0.92 |
|
Random Forest |
0.86 |
0.84 |
0.82 |
0.89 |
|
Neural Network |
0.88 |
0.86 |
0.84 |
0.91 |
|
Logistic Regression |
0.81 |
0.79 |
0.77 |
0.85 |
Table 4:
Explainability Output Summary.
|
Feature |
Relative Importance |
Effect on Outcome |
Decision Insight
(Why It Matters) |
|
Adherence History |
High |
Positive |
Past adherence
strongly predicts future adherence behavior and should anchor patient risk
stratification. |
|
Refill Gap |
High |
Negative |
Larger refill gaps
signal elevated non‑adherence risk and represent a modifiable intervention
point. |
|
Comorbidity Index |
Medium |
Positive |
Higher clinical
burden increases likelihood of treatment instability and switching. |
|
Age |
Medium |
Mixed |
Impact varies by
subgroup, indicating need for cohort‑specific interpretation rather than
global rules. |
|
Hospital Visits |
Low |
Positive |
Frequent inpatient
activity reflects underlying instability but is a secondary risk driver. |
Figure 3: Feature
Importance / SHAP Visualization.
(Figure 3)
SHAP based feature importance visualization illustrating the contribution of
key variables to model predictions in patient-level claims analytics. The chart
highlights the relative impact of factors such as adherence history, refill
gaps, comorbidity index, age and healthcare utilization on predictive outcomes.
Higher SHAP values indicate greater influence on the model’s decision, enabling
transparent interpretation of the drivers behind patient behavior predictions,
including treatment switching and medication adherence.
7.
Discussion
The findings of
this study highlight the practical value of integrating explainable AI into
patient-level claims data analytics11. From a pharmaceutical decision-making
perspective, the ability to interpret model predictions enhances strategic
planning, particularly in areas such as patient targeting, adherence
improvement programs and treatment optimization. Explainability enables
stakeholders to identify the underlying drivers of patient behavior, thereby
supporting more informed and evidence-based decisions2.
The incorporation
of explainability techniques significantly strengthens the role of real-world
evidence in healthcare analytics. By providing transparent insights into
predictive models, the framework improves the credibility and usability of
AI-driven outputs. This is particularly important in regulated environments,
where decision-making must be supported by interpretable and auditable
evidence.
However, the
study also highlights the inherent trade-off between predictive accuracy and
interpretability. While complex models such as gradient boosting and neural
networks offer higher accuracy, they require additional layers of explanation
to ensure transparency. Simpler models, although more interpretable, may not
capture the full complexity of claims data. The proposed framework addresses
this trade-off by combining high-performance models with robust explainability
techniques.
From a deployment
perspective, integrating explainable AI into pharmaceutical analytics systems
requires careful consideration of infrastructure, scalability and user
interface design14 Organizations must ensure that
explanations are not only accurate but also accessible and actionable for
decision-makers1.
Additionally,
continuous monitoring and updating of models are necessary to maintain
performance and relevance over time.
8.
Limitations
Despite its
contributions, this study has several limitations that merit consideration.
First, the quality and completeness of healthcare claims data can significantly
influence both predictive accuracy and explanation reliability. Coding
inconsistencies, missing data and variability across healthcare systems may
affect generalizability and require dataset‑specific adaptation.
Second, while
explainability techniques such as SHAP and LIME provide valuable insight, they
can introduce computational overhead, particularly in large‑scale, real‑time
environments. Enterprise deployments must therefore balance explanation
granularity with performance and cost considerations.
Finally organizational
readiness remains a non‑technical constraint. Effective adoption of explainable
AI requires stakeholder education, alignment across analytics and decision
teams and trust in both model outputs and explanations. These factors are
critical for translating technical capability into operational impact.
9.
Conclusion
This study presents a
comprehensive explainable artificial intelligence framework for patient‑level
healthcare claims data analytics with direct relevance to pharmaceutical
decision‑making. As machine learning models increasingly influence real‑world
evidence generation, patient targeting strategies and treatment optimization
efforts, the need for transparency, interpretability and accountability has
become critical.
The proposed
framework addresses this need by integrating high‑performance predictive
modeling with robust explainability techniques and embedded governance
mechanisms within a unified, scalable architecture. Results demonstrate that
advanced models such as gradient boosting and neural networks can be
effectively combined with post‑hoc interpretation methods, including SHAP and
LIME, without materially compromising predictive performance. More importantly,
the explainability layer enables stakeholders to understand the drivers of
model predictions, supporting informed, defensible and patient‑centric decision‑making.
By embedding
auditability, traceability and monitoring into the analytics lifecycle, the
framework aligns AI‑driven insights with regulatory expectations and real‑world
evidence standards. The inclusion of feedback loops further enables continuous
learning, allowing the system to adapt to evolving patient behavior and
healthcare dynamics while maintaining consistency and reliability.
Overall, this work
advances the practical application of explainable AI in healthcare claims
analytics by demonstrating that interpretability is not a trade‑off against
performance, but a prerequisite for responsible, enterprise‑scale deployment.
Future research may extend this framework through causal inference, privacy‑preserving
learning and federated architectures to further strengthen trust, robustness
and scalability in pharmaceutical analytics environments.
10. References
11. Molnar
C. Interpretable machine learning.
Lulu. Com, 2020.