Improving Understanding and Treatment of Rare Diseases through Real-World Data
Abstract
The
paper discusses the challenges in developing treatments for rare diseases, such
as limited understanding, small patient populations, diagnostic delays, and
disease heterogeneity. It emphasizes the role of real-world data in enhancing
the understanding of rare diseases by providing insights into natural history,
patient populations, treatment effects, biomarker identification, data sharing,
and regulatory decision-making. The paper highlights recommendations to improve
rare disease research, including promoting data sharing, utilizing advanced
technologies, enhancing patient engagement, establishing rare disease
registries, and fostering translational research initiatives. By addressing
these challenges and implementing these recommendations, the rare disease
research community can accelerate scientific discoveries, improve patient
outcomes, and transform healthcare for individuals affected by rare diseases.
Keywords: Treatment
outcomes, patient engagement, diagnostic delays, data harmonization
1. Introduction
and Background
A rare disease, also known as an
orphan disease, is a medical condition that has a low prevalence in the
population. The exact definition of a rare disease can vary by region, but it
is generally characterized by affecting a small number of individuals within a
given population. In the United States, a disease is considered rare if it
affects fewer than 200,000 individuals. Rare diseases are often complex and
heterogeneous, with diverse manifestations and underlying causes. Many rare
diseases have a genetic basis, and they can present significant challenges in
terms of diagnosis, treatment, and research due to their limited prevalence and
the lack of understanding surrounding them1,2.
Patients with rare diseases often face
diagnostic delays, as healthcare providers may not be familiar with these
conditions, leading to a prolonged period between the onset of symptoms and
receiving a correct diagnosis. Additionally, the development of effective
treatments for rare diseases can be challenging due to the small patient
populations, limited research funding, and difficulties in conducting clinical
trials3.
Efforts to address the challenges
associated with rare diseases include initiatives to increase awareness,
improve access to information, enhance research collaboration, and develop
targeted therapies. Regulatory frameworks such as the Orphan Drug Act in the
United States and the Orphan Regulation in the European Union aim to
incentivize pharmaceutical companies to develop treatments for rare diseases by
providing various benefits and incentives4,5.
The purpose of this paper is to
evaluate the role of real-world datasets in improving the understanding of rare
diseases. The paper tries to identify gaps in rare disease research and come up
with recommendations to help evolve the rare disease research.
2. Literature
Review
2.1 Challenges in developing treatment for rare
disease
Developing
treatments for rare diseases presents several challenges due to the unique
nature of these conditions. Some of the key challenges in developing treatments
for rare diseases include:
- Limited
understanding: Rare diseases are often poorly understood, with
limited knowledge about their underlying causes, disease mechanisms, and
natural history. This lack of understanding can hinder the development of
targeted therapies and effective treatment strategies.
- Small patient
populations: Rare diseases by definition affect a small number of
individuals, making it challenging to conduct clinical trials with an adequate
sample size. The small patient populations can also limit the availability of
data needed for research and drug development.
- Diagnostic delays:
Patients with rare diseases often experience delays in diagnosis due to the
rarity of their condition and the lack of awareness among healthcare providers.
Diagnostic delays can lead to disease progression and impact the timely
initiation of treatment.
- Heterogeneity: Rare
diseases encompass a wide range of conditions with diverse manifestations and
underlying genetic causes. The heterogeneity of rare diseases can complicate
treatment development, as therapies may need to be tailored to specific
subtypes or genetic mutations.
- Lack of biomarkers: Many
rare diseases lack well-defined biomarkers that can be used to measure disease
activity, predict treatment response, or monitor disease progression. The
absence of reliable biomarkers can make it challenging to assess the efficacy
of potential treatments.
- Limited research
funding:
Research funding for rare diseases is often limited compared to more common
diseases. The scarcity of funding can impede research efforts, hinder the
translation of scientific discoveries into clinical applications, and slow down
the development of new treatments.
- Recruitment and retention
of patients: Recruiting and retaining patients for clinical trials
for rare diseases can be difficult due to the small patient populations,
geographic dispersion of patients, and the need for specialized expertise in
managing these conditions. This can prolong the timeline for drug development
and regulatory approval.
- Addressing
these challenges requires a collaborative effort among researchers, healthcare
providers, patient advocacy groups, policymakers, and pharmaceutical companies.
Initiatives to improve data sharing, increase research funding, enhance patient
engagement, and streamline regulatory processes can help overcome the obstacles
in developing treatments for rare diseases and improve outcomes for patients
with these conditions1-3,6-8.
2.2. Role of real-world data in rare disease
understanding
Real-world
datasets play a crucial role in enhancing the understanding of rare diseases by
providing valuable insights into various aspects of these conditions. Some key
roles of real-world datasets in rare disease understanding include:
- Natural history
data:
Real-world data (RWD) can provide insights into the natural history of rare
diseases by capturing information on disease progression, clinical
manifestations, treatment patterns, and outcomes in routine clinical practice.
Understanding the natural history of rare diseases is essential for developing
targeted therapies and improving patient care.
- Patient populations:
Real-world datasets can help identify and characterize patient populations
affected by rare diseases, including demographic information, comorbidities,
genetic profiles, and treatment histories. This information can aid in
stratifying patients based on disease subtypes, severity, and response to
treatment.
- Treatment effects:
Real-world evidence can be used to assess the effectiveness and safety of
treatments for rare diseases in real-world clinical settings. By analyzing
treatment outcomes, adverse events, and long-term follow-up data, researchers
can evaluate the real-world impact of interventions and inform clinical
decision-making.
- Biomarker identification:
Real-world datasets can facilitate the identification of biomarkers associated
with rare diseases by analyzing genetic, clinical, and laboratory data from
diverse patient populations. Biomarkers play a critical role in disease
diagnosis, prognosis, and treatment response, and real-world data can help
validate and refine biomarker candidates.
- Data sharing and
collaboration: Real-world datasets enable data sharing and
collaboration among researchers, healthcare providers, patient advocacy groups,
and pharmaceutical companies. By pooling data from multiple sources,
researchers can enhance the statistical power of analyses, validate findings,
and generate new hypotheses to advance rare disease research.
- Regulatory decision-making:
Real-world evidence can support regulatory decision-making processes by
providing additional insights into the safety, effectiveness, and real-world
impact of treatments for rare diseases. Regulatory agencies may use real-world
data to supplement traditional clinical trial data and inform drug approval and
post-market surveillance.
- Real-world
datasets play a vital role in advancing the understanding of rare diseases by
providing valuable insights into disease progression, patient populations,
treatment effects, biomarkers, and real-world outcomes. Leveraging real-world
evidence can help address the challenges associated with rare diseases,
accelerate research efforts, and improve patient outcomes in this critical area
of healthcare9,10.
2.3. Patient identification in real-world data
Patient
identification in real-world datasets for rare diseases is a critical aspect of
research and healthcare delivery. Several approaches and considerations are
essential for effectively identifying and characterizing patients in these
datasets:
- Disease coding and
classification: Utilizing standardized disease coding systems such as
ICD-10 (International Classification of Diseases, 10th Revision) and Orphanet
classification to accurately identify rare diseases in electronic health
records (EHRs) and administrative databases. Ensuring that rare disease codes,
synonyms, and specific phenotypic features are captured to improve patient
identification and data quality.
- Genetic testing
and molecular diagnostics: Incorporating genetic testing results,
variant data, and molecular diagnostic information to identify patients with
rare genetic disorders based on specific gene mutations or variants. Linking
genetic data with clinical phenotypes to enhance patient stratification and
subgroup identification in real-world datasets.
- Patient registries
and disease-specific databases: Leveraging disease-specific
patient registries, research databases, and collaborative networks to access
detailed clinical information, treatment outcomes, and longitudinal data for
rare disease patients. Integrating data from multiple sources to create a
comprehensive view of patient characteristics, disease progression, and
response to therapies.
- Natural Language
Processing (NLP) and Text mining: Applying NLP algorithms and text
mining techniques to extract relevant information from unstructured clinical
notes, physician narratives, and medical reports in EHRs. Identifying key
clinical features, diagnostic criteria, and disease-specific keywords to
improve patient identification and phenotype characterization.
- Data linkage and
Cross-Referencing: Linking patient data across different
datasets, healthcare systems, and research repositories to create a
longitudinal patient profile and track healthcare utilization, treatment
patterns, and outcomes over time. Cross-referencing patient identifiers, genetic
data, and clinical variables to ensure data accuracy, privacy protection, and
data harmonization across diverse sources.
- Collaborative data
sharing and interoperability: Promoting data sharing initiatives, data
harmonization standards, and interoperable platforms to facilitate the exchange
of patient data and research findings among institutions, researchers, and
healthcare providers. Establishing data sharing agreements, governance
frameworks, and ethical guidelines to protect patient privacy, confidentiality,
and data security in real-world datasets.
By
employing these strategies and technologies, researchers and healthcare
professionals can enhance patient identification, data quality, and research
insights in real-world datasets for rare diseases. This comprehensive approach
enables the effective study of rare diseases, personalized medicine
initiatives, and the development of innovative therapies tailored to individual
patient needs9,11,12.
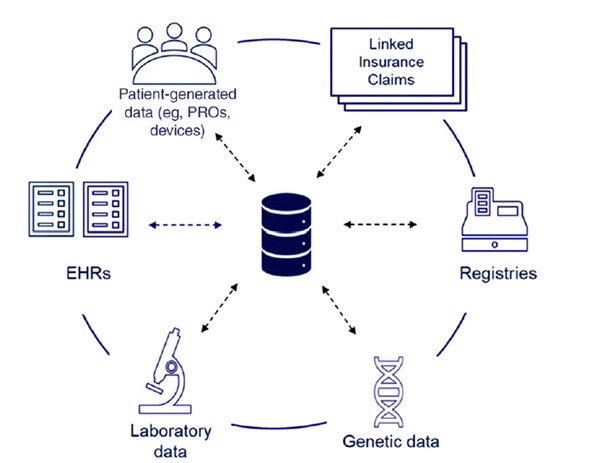
Figure
1:
Harmonization of Real-World Datasets9
2.4. Recommendations to improve rare disease
research
Improving
rare disease research requires a multidimensional approach that addresses
various challenges in data collection, patient identification, research
collaboration, and therapeutic development. Here are some recommendations to
enhance rare disease research:
- Enhance
Data Sharing and Collaboration: Encourage data sharing initiatives,
collaborative research networks, and multi-stakeholder partnerships to
facilitate the exchange of data, resources, and expertise in rare disease
research. Establish standardized data sharing protocols, interoperable
platforms, and governance frameworks to promote transparency, reproducibility,
and data harmonization across different datasets.
- Utilize
Advanced Technologies: Embrace innovative technologies such as artificial
intelligence (AI), machine learning, and data analytics to analyze large-scale
datasets, identify disease patterns, and predict treatment outcomes in rare
diseases. Implement bioinformatics tools, computational algorithms, and
high-throughput sequencing platforms to accelerate genetic discovery, biomarker
identification, and drug repurposing efforts.
- Improve
Patient Engagement and Advocacy: Involve patients, caregivers, and patient
advocacy organizations in research design, clinical trials, and healthcare
decision-making to ensure that research priorities align with patient needs and
preferences. Empower patients with access to educational resources, support
networks, and personalized health information to enhance their participation in
research studies and treatment decision-making.
- Establish
Rare Disease Registries: Develop comprehensive rare disease registries that
capture longitudinal patient data, genetic information, clinical outcomes, and
treatment responses to support epidemiological studies, natural history
research, and therapeutic development. Harmonize data collection standards,
data elements, and data sharing agreements across national and international
registries to enable cross-border research collaborations and data exchange.
- Promote
Translational Research: Foster translational research initiatives that bridge
basic science discoveries with clinical applications to accelerate the
development of novel diagnostics, therapies, and precision medicine approaches
for rare diseases. Support interdisciplinary research teams, translational
research centers, and public-private partnerships to translate scientific
insights into tangible benefits for patients and healthcare providers.
- Enhance
Regulatory Support: Advocate for regulatory incentives, orphan drug
designations, and expedited review pathways to incentivize pharmaceutical
companies, biotech firms, and academic researchers to invest in rare disease
drug development. Collaborate with regulatory agencies, patient advocacy
groups, and industry stakeholders to streamline regulatory processes, improve
access to orphan drugs, and promote patient-centered drug development
strategies.
By
implementing these recommendations, the rare disease research community can
overcome existing challenges, accelerate scientific discoveries, and improve
the diagnosis, treatment, and quality of life for individuals affected by rare
diseases. Collaboration, innovation, patient engagement, and regulatory support
are key drivers for advancing rare disease research and transforming healthcare
outcomes for rare disease patients13-15.
3.
Conclusion
The
paper emphasizes the importance of utilizing real-world datasets to enhance the
understanding and treatment of rare diseases. By addressing the challenges
faced by patients with rare diseases, such as diagnostic delays and limited
research funding, and by implementing recommendations to improve data sharing,
collaboration, and the use of advanced technologies, the rare disease research
community can accelerate scientific discoveries and improve healthcare outcomes
for individuals affected by rare diseases. Collaboration, innovation, patient
engagement, and regulatory support are highlighted as key drivers for advancing
rare disease research and transforming healthcare outcomes for rare disease
patients.
4. References
- Valdez R, Ouyang
L, Bolen J. Public health and rare diseases: Oxymoron no more. Prev Chronic Dis
2016;13: 05.
- Eurordis. Rare
diseases: understanding this public health priority. Eurordis 2005
- Richter T,
Nestler-Parr S, Babela R, et al. Rare disease terminology and definitions-A
systematic global review: report of the ISPOR rare disease special interest
group. Value Health 2015;18: 906-914.
- Council of the
European Union. Council recommendation on action in the field of rare diseases-2947th
employment, social policy, health and consumer affairs-council meeting. Council
of the European Union 2009.
- National Institute
of Health. Public Law 97-414 97th Congress. National Institute of Health 1983.
- Ji X,
Liang J, Ji S. Research status in treatment of rare diseases. Chinese Journal
of Clinical Pharmacy 2019;35: 115-118.
- Jiang J, Jing L,
Li W. Current views on rare diseases research and orphan drugs development.
Journal Bioengineering, 2011;27: 724-729.
- Kulagin A,
Ptushkin V, Lukina E, et al. Phase III clinical trial of Elizaria® and Soliris®
in adult patients with paroxysmal nocturnal hemoglobinuria: Results of
comparative analysis of efficacy, safety, and pharmacological data. Blood 2019;134:
3748.
- Liu S, Barrett JS,
Leonardi ET, et al. Natural History and Real-World Data in Rare Diseases
Applications. J Clin Pharmacol 2022;62: 38-55.
- Shen F, Zhao Y,
Wang L, et al. Rare disease knowledge enrichment through a data-driven
approach. BMC Medical Informatics and Decision Making 2019;19.
- Girman CJ, Ritchey
ME, Zhou W, Dreyer NA. Considerations in characterizing real-world data
relevance and quality for regulatory purposes: a commentary. Pharmacoepidemiol
Drug Saf 2019;28: 439-442.
- Rubinstein Y. The
GRDR-GUID: a model for global sharing of patients de-identified data. Academia 2014.
- EUCERD. EUCERD
core recommendations on rare disease patient registration and data collection.
EUCERD 2013.
- Commission Expert
Group on Rare Diseases. Recommendation on ways to improve codification for rare
diseases in health information systems. 2014.
- RD-Action. Overview report on the state of the art of rare disease activities in Europe. RD-Action, 2018.