Synergistic Machine Learning: Fusing Genomic Insights with Clinical Applications in Healthcare
Abstract
In the medical field, genomic data are poised to
unlock the potential of personalized diagnosis and treatment, with machine
learning as a critical enabler. This paper proposes the integration of Machine
Learning with genomics and clinical data, a solution that can navigate the
complexities and heterogeneity of medical data. As machine learning algorithms
realize their potential, the medical trend is shifting towards individualized
diagnosis and treatment, offering a promising future for healthcare.
Keywords: Machine Learning, Healthcare, Genomics,
Clinical Applications, Personalized therapy
1.
Introduction
The
emergence of advanced genomic high-throughput technologies has transformed our
comprehension of disease mechanisms and propelled the progress of precision
medicine. However, the challenge of transforming genomic data into actionable
intelligence that can inform clinical decisions promptly remains significant.
The integration of genomic and clinical data poses massive computing and
analytical challenges. With its ability to transform genomic data into a wealth
of information, machine learning offers a potential solution. This article
proposes using collaborative techniques that consider genetic parameters and
clinical practice to enhance diagnostic accuracy and issue personalized
treatment plans that meet individual needs.
2.
Problem Statement
The
healthcare system evolved from the traditional concept of a
"one-size-fits-all" approach to individual changes based on the
population averages of treatment decisions, as visualized in the figure below (Figure
1);
This
approach undermines the complexity of the relationship between genetic privacy,
genetics, environmental factors, and disease manifestations1. Consequently, the care could be of better
quality and more priced. From
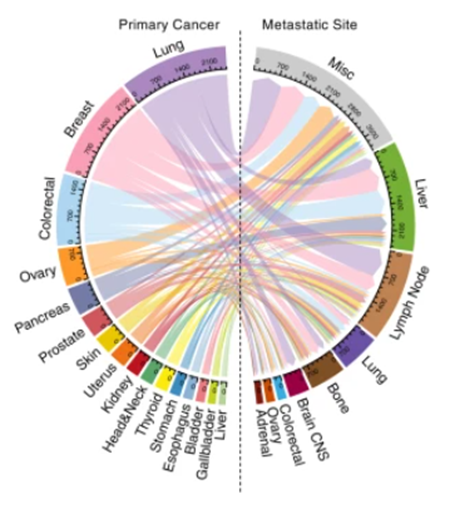
Figure
1.
Visualization of genomic data1
this
standpoint, the rise and complexity of genomic data will lead to its
processing, interpretation, and medication integration in healthcare settings.
Adequate data management tools and analytical principles are a requirement for
harnessing the hidden power of genomics.
3.
Solution
The
architecture of the framework relies on the latest ML algorithm. It carries out
complex algorithms and data analytics through the patients' genetic data and
clinical information. Such techniques include the oldest to the novel models of
supervised learning, including the most recent deep learning architectures,
which are capable of detecting the hidden patterns in genomics data of high
dimensions and making accurate predictions and decisions, as illustrated in the figure below2. One of the formulas utilized in these
calculations for dimensionality reduction techniques for genomic data analysis
includes the formula for Principal
Component Analysis (PCA) (Figure 2 and 3)
Figure 2. Patients' genetic data and clinical information2
Y=XW
Y: Output or predicted values.
?: Input matrix with predictor variables.
?: Weight matrix or coefficients for features.
Figure
3.
Gene expression3
Figure 4. The architecture of
the framework5
The
algorithm, operating via machine learning models on all datasets, including
genotype data, clinical phenotypes, and treatment outcomes, can detect
biomarkers, genetic variations, expressions illustrated below, and molecular
signatures associated with disease onset, progression, and treatment response3.
Furthermore,
the framework adopts feature selection, dimensionality reduction, and ensemble
learning to break the curse of dimensionality and improve model
interpretability. ML-based decision support tools can be adopted into the
clinical workflow to help doctors face the challenge of genomic data coding and
make better, more personalized treatment decisions4.
Genetic coding is illustrated below (Figure 4);
Therefore,
this becomes an ideal route for extracting and processing genomic information
to translate to personalized medicine.
4.
Uses
The
synergistic machine learning in medicine encompasses many applications targeted
at changing medical practice forever. The application that uses ML algorithms
to identify people at a higher risk of being affected by various diseases via
the analysis of both genomic and clinical data is one of the fantastic
achievements5. The following formula
is employed in classification algorithms for this disease risk prediction;
The formula for
Logistic Regression
P (Y=1∣ X) = 1/
1+e^[−(β0+β1X1+…+βnXn)]
·P(Y=1∣X): Probability of the outcome ?Y being 1 (success) given the input variables ?X.
·?e:
Euler's number, approximately equal to 2.71828, raised to the power of the
linear combination of input variables and their coefficients.
·?0,?1,…,??β0,β1,…,βn: Coefficients determining the impact of input variables ?1,?2,…,??X1,X2,…,Xn on the probability of the outcome being 1. ?0β0
is the intercept term.
·?1,2,…,??X1, X2,…, Xn: Input variables or features influencing the outcome ?Y.
The
initial diagnosis will help in the provision of timely interventions as well as
preventive measures that may eliminate the risk of the disease or at least
reduce its progression. In addition, the patients are clustered or grouped
based on their molecular profile to prescribe targeted therapy, which improves
patient outcomes6 (Figure 5).
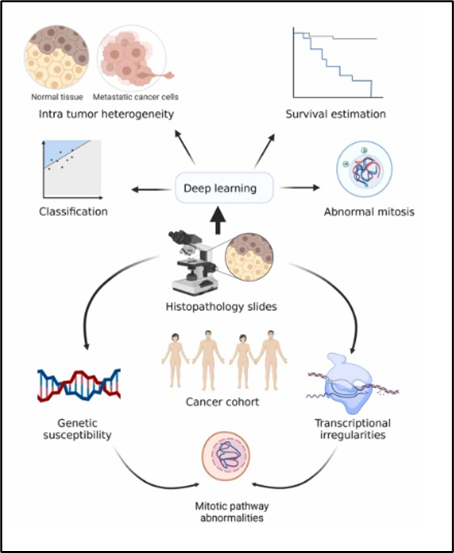
Figure
5. Integration of histological slides and
genetic susceptibility data6
Moreover,
cooperative machine learning transcends diagnosis and risk prediction, using
patients' genomic information to design individualized treatment timetables
that consider their unique genomic profiles. Synthesis of genetic diversity and
molecular processes will make personalized therapies more powerful with minimum
adverse effects (Figure 6).
Figure
6.
Synthesis of genetic diversity and molecular processes
Firstly,
integrating genomics into the clinical decision support systems helps
physicians have conclusive evidence and the opportunities to apply methods of
treatment that follow the patient's condition changes and disease progression.
All the phases of patient care, such as disease prevention and diagnostics,
treatment optimization, and care management, fall under the domain of
collaborative machine learning in healthcare (Figure 7).
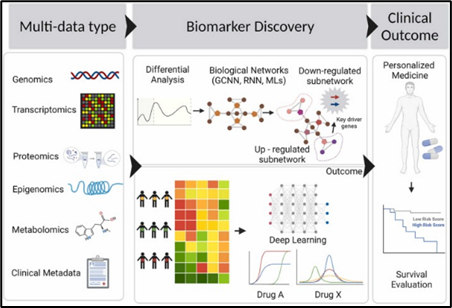
Figure 7. Deep learning ensemble modeling7
5.
Impact
This
integration of genomics with clinical care via MLissynonym for new and upcoming
healthcare delivery practices is primarily because GBL is competent in
decreasing the number of wrong diagnoses, even treatment, and, for that matter,
better results among the patients7.
In such a scenario, an outcome can be achieved through a personalized therapy
package that includes a maximized positive impact and minimized negative effect
(Figure 8).
Moreover,
these revelations are turning out to be a catalyst for developing new drugs and
personalized treatments that only fit some gene variations8.
Figure
8.
Basics of deep learning models8
Synergistic
machine learning in healthcare affects individual patients and benefits
society: it provides healthcare care at a lower cost, more effective population
health, and an equitable health system.
6.
Scope
Genomic
data integrated with machine learning has multifaceted clinical applications
consisting of technical, ethical, legal, and interdisciplinary factors (Figure
9).
Figure
9.
Genomic data integrated with machine learning
Designing
machine learning algorithms, building an exemplary data structure, and
implementing interoperable software solutions will ease the technical issues.
Moreover, a robust ethical and legal framework for patient consent, data
privacy, and security needs to be developed while considering ways to avoid
algorithm bias and ensure transparency and auditability of the decision-making
process9.
Allele
frequency, the ratio of copies of a particular allele to the total number of
alleles in a population, is one of the most essential tools for studying
genetic variability and population change.
The formula for Allele
Frequency Calculation
Allele frequency
= (# of copies of the allele) / (Total # of alleles in the population)
Appreciation
of allele frequencies is the framework for evolutionary processes, population
genetics, and the inheritance of genetic traits in different populations (Figure
10).
Figure
10.
Structure-based on generative deep learning models10
To
a lesser extent, we still have to work together, more importantly, to
streamline the regulations as well so that all of us, including the regulators,
policymakers, and industry members, can come up with clear guidelines and
standards to be put in place to be made10.
On the other hand, creating an interdisciplinary team bringing together
clinicians, data scientists, bioinformaticians, and ethicists will stimulate
innovation and knowledge sharing and allow the field to be viewed in a new
light. Furthermore, the significance of machine learning in healthcare in
saving lives, improving treatment delivery, and creating equal access to
genomics needs to be referenced11.
7.
Conclusion
In conclusion, applying genomic aspects to therapeutic applications or machine learning is an invention comparable to redefining diagnosis, treatment, and prognosis in medicine. ML can evaluate, interpret, and comprehend the genome's intricate knowledge. Thus, this method allows for proper diagnosis, personalized treatment, and healthcare supplies. Besides all the technical, ethical, and legal obstacles still to be addressed, there is a pressing need for widespread involvement and knowledge dissemination. Collaborative ML could evolve to a point where an individual's healthcare could be personalized to their genetic makeup and healthcare requirements instead of just general.
8. References
- Yu K. A Study on the Future Economic Trends in the
World. BCP business & management
2022.
- Sahu M, Gupta R, Ambasta RK, Kumar P. Artificial
intelligence and machine learning in precision medicine: A paradigm shift in
big data analysis. Progress in
molecular biology and translational science 2022;190: 57-100.
- Abdelhalim H, Berber A, Lodi M, et al. Artificial Intelligence,
Healthcare, Clinical Genomics, and Pharmacogenomics Approaches in Precision Medicine.
Front Genetics 2022;13.
- Hulsen T, Jamuar SS, Moody AR, et al. From Big Data to Precision Medicine. Front medicine 2019;6.
- Alowais SA, Alghamdi SS, Alsuhebany N, et al. Revolutionizing healthcare:
the role of artificial intelligence in clinical practice,” BMC Medical Education 2023;23.
- Yang S, Zhu F, Ling X, Liu Q, Zhao PW. Intelligent
Health Care: Applications of deep learning in computational medicine. Front Genetics 2021;12.
- Joshi
M, Pal A, Sankarasubbu M. Federated Learning for Healthcare Domain-Pipeline,
Applications, and Challenges. ACM
transactions on computing for healthcare 2022;3: 1-36.
- SA Alowais, Alghamdi SS, Alsuhebany N, et al. Revolutionizing healthcare:
the role of artificial intelligence in clinical practice. BMC Medical Education 2023.
- Mohammed
MA, Abdulkareem KH, Dinar AM, Garcia-Zapirain B. Rise of deep learning clinical
applications and challenges in omics data: A systematic review. Diagnostics 2023;13.
- Krishnan G, Singh S, Pathania M, et al. Artificial intelligence in
clinical medicine: catalyzing a sustainable global healthcare paradigm. Front artificial intelligence 2023;6.
- Dharmaratne
S. Studies on self-incompatibility in 'Brassica napus’. University of St
Andrews 2018.