Autoimmune Hepatitis Through the View of Personalized and Precision Healthcare Driven Approaches
By integrating interconnected datasets, multi‑OMICS technologies provide a comprehensive understanding of the continuum between health and disease and support PPM‑guided approaches to screening, prediction, monitoring and targeted therapy. In this context, PPM‑guided technologies represent a unique preventive strategy for diagnosing, monitoring, controlling and managing chronic disorders, including autoimmune diseases, with autoimmune hepatitis (AIH) as a paradigmatic example1.
AIH is an immuno‑inflammatory liver disease of unknown etiology and non‑self‑limiting clinical course, induced by environmental factors in genetically predisposed individuals, with a wide range of clinical manifestations. It is typically accompanied by increased alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels, elevated immunoglobulin G (IgG) and/or gamma globulins, the presence of autoantibodies (autoAbs) and morphological signs of active portal/lobular hepatitis, with a generally favorable response to immunosuppressive therapy (IST) but progression to liver cirrhosis (LC) and end‑stage liver failure in the absence of treatment. In brief, AIH is characterized by hypergammaglobulinemia, elevation of serum autoAbs and transaminases, and interface hepatitis, requiring liver biopsy and serologic testing, as well as close monitoring of liver function tests and serum immunoglobulin levels2-4.
The etiology of AIH is not entirely unraveled, but accumulating evidence supports an intricate interaction among genetic variants, environmental factors and epigenetic modifications, including gene–gene and gene–environment interactions with drugs, viral infections and the complex microbiome. Epigenomic alterations such as DNA methylation, histone modifications and non‑coding RNA transcription contribute to dysregulated immune tolerance. In this context, PPM‑guided approaches are particularly attractive in AIH because of its clinical and biological heterogeneity, the frequent subclinical or atypical presentation and the difficulty in predicting disease progression and treatment response. PPM offers the potential to identify at‑risk individuals earlier and to stratify patients more accurately for effective and individually targeted management1.
2. Classical view on autoimmune
hepatitis
2.1. Epidemiology
The incidence of AIH varies from 0.67 to 2.0 cases per 100,000
population per year, and the prevalence ranges from 4.0 to 42.9 cases per
100,000 population, depending on the geographical region. The ratio of men to
women ranges from approximately 1:4 to 1:6. A significant increase in the
incidence of AH in the first two decades of the 21st century was noted in
Spain, Denmark and the Netherlands, while in the Asia-Pacific region there was
a stable, albeit consistently high level. This geographical differentiation can
partly be explained by the "hygiene hypothesis", which suggests high
sanitary standards in advanced economies, resulting in a significant reduction
in contact with the external microflora. This, in turn, causes changes in the
composition of the microbiome, which leads to the induction of systemic
autoimmune reactions3,4.
2.2. Etiology and pathogenesis
The etiology of AIH remains unknown, but it is believed that the
interaction of genetic and epigenetic factors determines the occurrence and
outcome of the disease.
Meanwhile, the pathogenesis of AIH is complex, involving T cells,
macrophages and plasma cells that invade the periportal parenchyma and trigger
an inflammatory cascade that can result in liver damage. Numerical and
functional defects of regulatory T cells have a permissive role that enables
autoimmune liver injury to occur and persist4,5.
The dominant hypothesis suggests that AIH develops in genetically
predisposed individuals after exposure to one or more environmental factors,
leading to loss of immune tolerance. Autoimmune mechanisms are then sustained
through the following processes:
4) T-regulatory (Tregs) cell dysfunction.s a result, all these complex mechanisms of an aberrant immune response with an imbalance between pro-inflammatory and
The predominance of women is explained by the location of some genes involved in the development of immunological tolerance on the X chromosome. The modulating effect of the X chromosome on innate and adaptive immune pathways involved in autoimmune processes has been revealed. AIH is often combined with other immune-mediated diseases, primarily autoimmune, but also allergic and polygenic autoinflammatory, which suggests a common pathogenesis.
AIH is a polygenic disease, the main risk-bearing loci of certain HLA
alleles are present in about 50% of AIH patients, but they alone do not explain
the observed risks of the disease. Studies of candidate gene associations and
large-scale genome-wide association studies (GWAS) have indicated a strong
genetic predisposition to this disease. This predisposition to AIH in adults is
associated with the presence of certain HLA alleles encoded by highly
polymorphic genes associated with the MHC on the short arm of chromosome 6,
class I (A*01, B*08) and class II (DRB1*03, -04, -07 or -13) depending on
geographical regions and ethnicity. Among the risk alleles, HLA-DRB1*03:01 and
HLA-DRB1*04:01 (the two most frequently described) account for the vast
majority of associations between HLA and AIH in European and American
populations.
Epigenetic mechanisms directly affect gene expression without altering
nucleotide sequences and are reversible, cell‑type specific and influenced by
age, sex and environmental factors. Limited studies suggest that
hypomethylation of regulatory genes and dysregulated microRNA expression
contribute to loss of immune tolerance in AIH6-8.
2.3. Classification
1) Type 1 AIH (about 90% of all cases) — characterized by the presence of antinuclear (ANA) and (or) anti-smooth muscle (ASMA) antibodies;
2) Type 2 AIH is characterized by the presence of antibodies to type 1 liver/kidney microsomes (anti-LKM-1) in 100% of cases, as well as antibodies to type 1 liver cytosolic antigen (anti-LC-1) and anti-LKM-3;
3) Type 3 AIH is characterized by the presence of an antibody to soluble liver antigen / hepatic pancreatic antigen (anti-SLA/LP) in the absence of other autoantibodies.
It is believed that the type of AIH is important in the management of children (it helps in predicting treatment outcomes), but it is not informative for adult patients, since the latter have a similar course of AIH of various types2.
2.4. Clinical course and diagnostics
2.4.1. Clinical presentation and acute severe AIH
The clinical manifestations of AIH are diverse and range widely from asymptomatic and low-symptomatic over several years to acute forms with liver failure. Approximately one third of people are diagnosed at the LC stage. Early diagnosis and consistent, adequate IST are crucial for a prognosis that is favorable with proper treatment. AIH is relatively rare in the general population and can be easily overlooked due to its variable clinical presentation and sometimes difficult diagnosis. The diagnosis of AIH should be considered for any unclear acute or chronic hepatopathy.
Acute severe AIH is defined as a condition that develops within 26 weeks from the first clinical symptoms to the moment of hospitalization with characteristic indicators: jaundice and an increase in the international normalized ratio (INR ≥1.5) in the absence of histological signs of LC. It is important to note that patients with acute severe AIH very often have symptoms that are not typical for the disease, including normal IgG levels and the absence of serological markers2,3.
2.4.2. Serological markers and scoring systems
An international AIH study group has developed a scoring system for the diagnosis of this disease, which has undergone minor modifications in subsequent years (Table 1); along with it, a simplified system of diagnostic signs has been proposed (Table 2). Indirect immunofluorescence is the preferred and main method of routine testing for all Abs (ANA, ASMA, anti-LKM-1), except for Abs against SLA/LP – they should be performed using an immunoblot.
Table 1.
Comprehensive Diagnostic Scoring System of the International Autoimmune Hepatitis Group
|
Clinical features |
Points |
Clinical features |
Points |
|
Female |
+2 |
Average alcohol intake (g/day) | |
|
<25 |
+2 | ||
|
>60 |
−2 | ||
|
AP:AST (or ALT) ratio |
Histologic findings | ||
|
<1.5 |
+2 |
Interface hepatitis |
+3 |
|
1.5–3.0 |
0 |
Lymphoplasmacytic infiltrate |
+1 |
|
>3.0 |
−2 |
Rosette formation |
+1 |
|
Biliary changes |
−3 | ||
|
Other atypical changes |
−3 | ||
|
None of above |
−5 | ||
|
Serum globulin or IgG level above ULN |
Concurrent immune disease, including celiac
disease |
+2 | |
|
>2.0 |
+3 |
Other autoantibodies |
+2 |
|
1.5–2.0 |
+2 |
HLA DRB1*03 or DRB1*04 |
+1 |
|
1.0–1.5 |
+1 | ||
|
<1.0 |
0 | ||
|
ANA, SMA, or anti-LKM1 |
Response to corticosteroids | ||
|
>1:80 |
+3 |
Complete |
+2 |
|
1:80 |
+2 |
Relapse after drug withdrawal |
+3 |
|
1:40 |
+1 | ||
|
<1:40 |
0 | ||
|
AMA positive |
−4 | ||
|
Viral hepatitis markers |
Aggregate score pretreatment | ||
|
Positive |
−3 |
Definite autoimmune hepatitis |
>15 |
|
Negative |
+3 |
Probable autoimmune hepatitis |
10–15 |
|
Hepatotoxic drug exposure |
Aggregate score posttreatment | ||
|
Positive |
−4 |
Definite autoimmune hepatitis |
>17 |
|
Negative |
+1 |
Probable autoimmune hepatitis |
12–17 |
AP - alkaline phosphatase; AST - aspartate aminotransferase; ALT - alanine aminotransferase; IgG - immunoglobulin G; ULN - upper limit of the normal; HLA - human leukocyte antigen; ANA - antinuclear antibodies; SMA - smooth muscle antibodies; anti-LKM1 - antibodies to liver kidney microsome type 1; AMA - antimitochondrial antibodies (Table 2).
Table 2: Simplified Diagnostic Criteria for AIH.
|
Variable |
Cutoff |
Point |
|
Autoantibodies |
ANA or SMA 1:40 |
1 |
|
ANA or SMA ≥1:80 |
2 |
|
|
LKM (≥1:40) or SLA positive |
2 |
|
|
IgG |
>ULN |
1 |
|
>1.1×ULN |
2 |
|
|
Liver histology |
Compatible |
1 |
|
Typical |
2 |
|
|
Absence of viral hepatitis |
Yes |
2 |
≥6 – AIH; ≥7 –
definite AIH.
ANA – anti-nuclear antibodies; SMA – anti-smooth muscle antibodies; LKM – liver kidney/microsome; SLA – soluble liver antigen; IgG – immunoglobulin G; ULN – upper limit of normal.
The absence of autoAbs with confirmed AIH makes it possible to diagnose a seronegative variant of the disease; the frequency of its detection, according to various sources, varies from 1-5% to 19-34%. Seronegativity often accompanies acute severe AIH at the initial stage, as autoAb secretion is delayed in these patients. This determines the expediency of repeated serological examination after 2-4 weeks. In some cases, the diagnosis can confirm the detection of atypical autoAbs, for example, perinuclear antineutrophil cytoplasmic Abs (pANCA)3,4,9.
Patients with AIH are not characterized by an increase in the activity of alkaline phosphatase (ALP), as well as a significant increase in the value of gamma-glutamyltranspeptidase (GGT). Thus, the activity of ALP ≥ 2ULN and/or GGT ≥5 ULN may indicate the development of overlap syndrome with cholestatic liver disease – primary biliary cirrhosis (PBC) or primary sclerosing cholangitis (PSC). Such patients need to undergo additional examination, including the determination of antibodies typical of PBC (antimitochondrial antibodies – AMA-M2, anti-gp-210, anti-sp-100) and magnetic resonance cholangiopancreatography (MRCP)2.
2.4.3. Histology: The histological picture of AIH is characterized by periportal inflammation with piecemeal or bridged necrosis, lymphomacrophagal infiltration in the portal and periportal zones, often with the presence of a significant number of plasma cells. Cirrhosis usually has macronodular features and is formed against the background of the ongoing activity of the pathological process. There are no pathognomonic histological features of AIH2,3,10.
2.4.4. Overlap
syndromes and differential diagnosis: In the differential
diagnosis of cholestatic liver lesions or their combination with AIH, it is
desirable to conduct an immunohistochemical examination of tissue sections with
cytokeratin 7 (CK7) to objectify the nature of hepatocyte damage and the degree
of involvement of lobular and portal bile ducts in the process. CK7-positive
periportal hepatocytes are reliable signs of chronic cholestasis, and their
presence suggests considering an alternative diagnosis or overlap syndromes. It
should be noted that this sign of chronic cholestasis loses its diagnostic
value more in the presence of severe fibrosis or LC, when a nonspecific
accumulation of copper-associated protein and CK7-positivity can be observed,
regardless of the disease ethiology.
Detection of periductular fibrosis and other histological signs of PSC in patients with AIH is a mandatory criterion for the diagnosis of AIH/PSC of small ducts. At the same time, up to 10% of adult patients with AIH have signs of bile duct damage. During differential diagnostics, it is important to remember that the picture of periductular fibrosis is not highly specific for PSC and can be observed in a number of diseases.
The Paris AIH/PBC criteria are the most reliable tool
for the diagnosis of this overlap syndrome. According to them, the diagnosis
can be established if two of the three criteria of PBC and two of the three
criteria of AIH are present.
PBC criteria for AIH/PBC:
· presence of АМА-M2;
· typical histological lesions of the bile ducts.
· AIH criteria for AIH/PBC:
· serum ALT activity ≥ 5 ULN;
· serum IgG level ≥ 2 ULN or presence of ASMA;
· histologically – periportal or periseptal lymphocytic piecemeal necroses.
There are no generally accepted diagnostic criteria for PSC/AIH. Diagnosis is based on the identification of clinical, biochemical and histological signs of AIH and cholangiographic changes or histological features typical of PSC.
AIH criteria for AIH/PSC:
· The presence of ANA and (or) ASMA in a titer of at least 1:40; 2) periportal or periseptal lymphocytic piecemeal necroses.
PSC criteria for AIH/PSC:
· Or histological signs of PSC of small ducts (periductular fibrosis, ductopenia, etc.).
MRCPG is the main diagnostic method for PSC of large ducts. It should be considered that cholangiographic imaging does not reveal early changes in the ducts, therefore, patients with a high degree of suspicion of PSC / AIH require MRCPG over several years.
Special attention should be paid to patients who developed the disease in childhood and at a young age, since up to half of children with AIH have clinical, laboratory, and cholangiographic signs of PSC. The term "autoimmune sclerosing cholangitis" is often used to refer to AIH/PSC in children2,10.
2.4.5. Drug‑induced autoimmune‑like hepatitis: Drug-induced autoimmune-like hepatitis (DI-ALH) is one of the phenotypes of drug-induced liver injury, occupying 2.3-9.2% of its structure. More than 40 different drugs that can cause DI-ALH have been described, including nitrofurantoin, methyldopa, hydralazine, minocycline, interferons, statins, methylprednisolone, diclofenac, antitumor protein kinase inhibitors, monoclonal Abs (MAbs) and conjugates of Abs, TNF-α inhibitors, phytopreparations and biologically active additives. However, some drugs and vaccines may trigger the development of true AIH2,3.
2.5. Standard
treatment approach
The mainstay of treatment of AIH is non-specific
immunosuppression, consisting of steroids. Prompt response to treatment with
steroids and other immunomodulatory drugs is almost universal and supports the
diagnosis. The aims of canonical treatment are to induce and maintain long-term
remission of liver inflammation. Treatment can often even reverse liver
fibrosis, thus preventing progression to advanced cirrhosis and its
complications2-4.
The aim of AIH treatment is to achieve complete remission of the disease (as defined by the normalization of liver enzymes, normalization of serum gamma globulin levels, resolution of clinical symptoms, and improvement in histological inflammation on liver biopsy) and prevent the progression of liver damage4.
In this context, PPM-guided therapy is necessary in AIH because of its heterogeneity in clinical manifestations. Moreover, pre-early (subclinical) recognition is ideal, but initial stages may be asymptomatic or mild, disease presentation heterogeneous, and both progression and severity can be hard to predict.
IST is the basic method of AIH treatment. It helps to
stop the inflammatory process, achieve remission of the disease, helps reduce
fibrosis, and also increases the life expectancy of patients. There are several
periods in the treatment of AIH:
· Maintenance;
· Discontinuation of therapy;
· Treatment of relapse.
3. PPM‑guided stratification and biomarkers in AIH
3.1. Limitations of
current diagnostic approach
At this point, unfortunately, there is no single
specific biomarker for AIH diagnosis. Traditional serum biomarkers of liver
injury and treatment response in AIH include AST and ALT, IgG, and, less
frequently, 6‑thioguanine (6‑TG). While liver biopsies are specific to active
disease at initial presentation, their impracticality for serial use,
particularly in decisions regarding immunosuppressive therapy (IST)
optimization, poses challenges. Consequently, there is a pressing demand for
non‑invasive blood‑based biomarkers that can accurately identify patients at
heightened risk of relapse or function as early indicators of relapse10-12.
As AIH is a disease without signature diagnostic
features, composite diagnostic criteria have been established, consisting of:
· Characteristic laboratory findings (elevated serum hepatic enzymes and increased serum IgG), and
· Positive disease‑defining autoantibodies (autoAbs), with exclusion of other liver diseases that may resemble AIH.
The lack of diagnostic accuracy is particularly evident in atypical cases, such as autoAb‑negative hepatitis, acute‑onset AIH with normal IgG values, and AIH overlapping with primary biliary cholangitis, primary sclerosing cholangitis or non‑alcoholic steatohepatitis (NASH).
3.2. Novel candidate
biomarkers
Novel candidate noninvasive biomarkers are emerging
from gene expression profiles, proteins, metabolites, and immune cell
phenotypes observed at various stages of the disease. For instance, growth
differentiation factor 15 (GDF15) has garnered attention as a potential
diagnostic and therapeutic marker for AIH, demonstrating significantly elevated
serum levels in AIH patients, particularly those without cirrhosis, with high
diagnostic accuracy and prognostic value to differentiate atypical forms of AIH
from other liver conditions13.
The other novel biomarker, i.e., serum GP73 is a potential noninvasive biomarker to evaluate the severity of liver necroinflammation in patients with AIH14,15.
3.3. PPM model and
multi‑omics framework
In AIH, PPM‑guided
diagnostics relies on OMICS‑derived biomarkers to refine disease staging,
predict relapse and stratify patients by expected treatment response,
complementing conventional serology and histology. Multi‑omics integration and
bioinformatics support the identification, verification and validation of such
biomarkers, thereby enabling more accurate risk assessment and individualized
therapeutic decisions16,17.
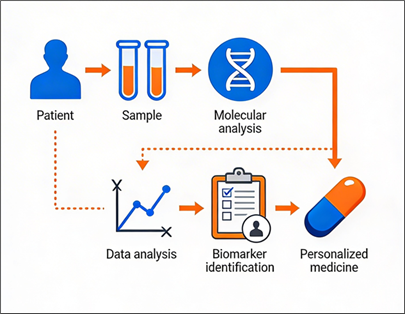
Fig. 1 The role and impact of biomarkers in daily
PPM-driven clinical practice
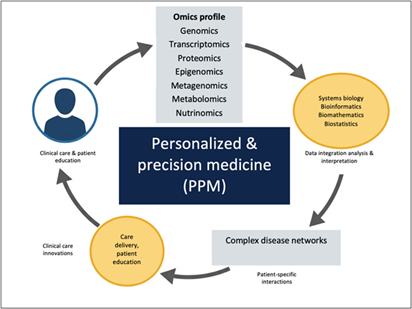
Fig. 2 Role
& Impact of OMICS Technologies and Bioinformatics for Personalized &
Precision Medicine
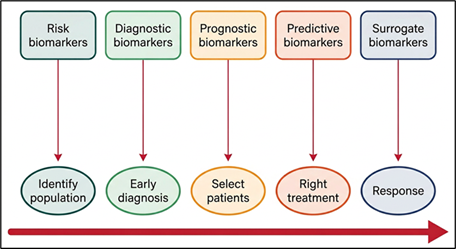
Fig. 3 PPM model as a unique strategy for diagnosing,
monitoring, controlling and managing chronic disorders: biomarkers as
diagnostic, risk, prognostic, predictive, and surrogate endpoints
Fig. 4 Application of biomarkers in relation to the
course of chronic disease
Adapted from: Fang-Shu Ou et al.
Biomarker Discovery and Validation: Statistical Considerations. Journal of
Thoracic Oncology, Volume 16, Issue 4, 2021, Pages 537-545, ISSN 1556-0864, https://doi.org/10.1016/j.jtho.2021.01.1616, and Mishra, A.;
Verma, M. Cancer Biomarkers: Are We Ready for the Prime Time? Cancers 2010, 2,
190-208. https://doi.org/10.3390/cancers2010190
3.4. Advanced data integration and AI‑driven subtyping
The integration of advanced technologies, including
bioinformatics and bioanalkytics, enhances the prediction of AIH patient
responses and identifies novel biomarkers. Innovations in next-generation
sequencing (NGS), pharmacogenomics and mass spectrometry (MS) have also
advanced our understanding of genomics landscapes in AIH patients and
pre-illness persons-at-risk, and host responses, facilitating the development
of biomarker-driven multi-targeted therapies and personalized vaccines of the
next-step generation. By integrating genetic, environmental, and lifestyle
factors, the above-mentioned PPM-guided approach deepens the understanding of
network-related interactome interactions in AIH, ultimately advancing the goals
of individualized care and disease elimination (Figure 5).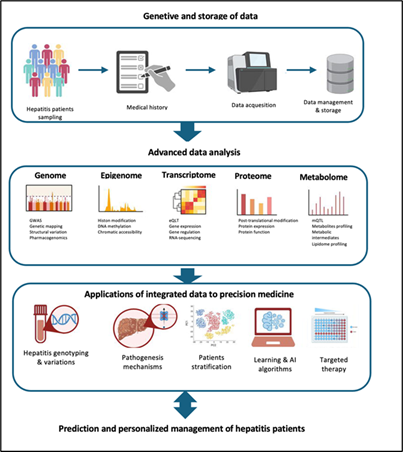
Figure 5: Advanced data integration in AIH subtyping theouth the View of
PPM-guided approaches.
This workflow demonstrates the use of advanced technologies - multi-OMICS, machine learning, and artificial intelligence -to refine hepatitis subtyping and personalize treatment strategies. It emphasizes identifying high-risk patients and ensuring high-quality data for analysis. Artificial intelligence and machine learning integrate these data sets to enable precise diagnostics, novel therapeutic target discovery, and customized treatments, improving patient outcomes. Abbreviations: GWAS, genome-wide association studies; eQTL, expression quantitative trait loci; mQTL, methylation quantitative trait loci; AI, artificial intelligence. Adapted from:
4. Immunosuppressive Therapy in the Context of PPM
4.1. Treatment phases
and response criteria
· Insufficient response: lack of normalization of serum transaminases and IgG (above ULN) 6 months after the start of treatment;
· Lack of response: decrease in serum transaminase levels by less than 50% within 4 weeks after the start of treatment;
· Remission: absence of clinical and laboratory signs of activity (complete biochemical response) combined with the absence of histological activity (histological activity index <4). This term is valid only when performing a control liver biopsy. In a broader sense, AIH remission implies the absence of disease activity (according to clinical data, laboratory and histological studies), accompanied by a significant reduction or complete regression of clinical symptoms, the possibility for the patient to return to normal life, complete or partial regression of fibrosis;
· Treatment intolerance: any adverse event, possibly related to the treatment (as assessed by the attending physician), which led to the discontinuation of therapy;
· Relapse: an increase in the activity of the disease after induction of remission and withdrawal or reduction of the IST doses.
Standard AIH treatment is inadequate for a proportion of patients, leading to the emergence of other treatment options and lines. New therapeutic strategies are needed, with the aim of obtaining long-lasting disease remission without inducing non-specific immunosuppression and a focus on inhibiting the intrahepatic proinflammatory milieu or expanding the pool of regulatory T cells, or both11,18.
4.2. Induction
therapy
Previously, prednisone was positioned as an induction
therapy at an initial dose of 60 mg/day for monotherapy and 40 mg/day for
combination therapy. However, later it became clear that a personalized
approach to determining the dose of the drug was required. As a first-line
therapy for AIH, except in cases of decompensated LC and acute severe
hepatitis, a combination of prednisone (0.5–1 mg / kg / day) and azathioprine
(1-2 mg / kg / day), or prednisone (0.5-1 mg / kg / day) and mycophenolate
mofetil (MMF; 1-2 g / day) is prescribed in individually selected doses.
Parenteral administration of prednisone (followed by oral administration) can be used in severe cases, suspected malabsorption, as well as in cases of insufficient compliance.
The rate of prednisone dose reduction is selected individually, depending on the patient's condition, the presence or absence of signs of liver failure and the response to therapy. Dose reduction to maintenance one is carried out under the control of laboratory parameters of the liver and the safety of treatment. Usually, dose reduction begins 2 weeks after the start of treatment. The rate of reduction is 5 mg of prednisone per week to a dose of 15-20 mg/day. Further reduction is considered only after normalization of transaminase activity. The maintenance dose ranges from 5 to 15 mg /day under the control of laboratory liver parameters.
An alternative to systemic glucocorticoids is budesonide, which has demonstrated efficacy in inducing and maintaining remission and has fewer side effects. The disadvantages of budesonide, which prevent its use as a first-line therapy, include the lack of systemic action (which is necessary for extrahepatic manifestations of AIH), the risk of activation of concomitant autoimmune diseases against the background of topical therapy, and the inability to titrate the dose to maintenance. In addition, budesonide is contraindicated in portal hypertension due to an increased risk of thrombosis in the portal vein system. At the same time, with portosystemic bypass surgery, there is no effect of the first passage through the liver, and the systemic side effects of budesonide will be the same as those of traditional glucocorticoids.
In induction therapy, prednisone is usually combined with azathioprine, an immunosuppressant from the group of thiopurine antimetabolites. Prednisone monotherapy and prednisone and azathioprine combination therapy are considered equally effective in inducing a response, but combination therapy helps to reduce side effects. Azathioprine is optimally added on average 2 weeks after the start of prednisone treatment, optimally with a decrease in bilirubin levels <100 µmol/l. Delayed use is primarily due to the potential hepatotoxicity of azathioprine. Azathioprine is contraindicated in patients with decompensated LC and acute severe AIH.
Prednisone monotherapy is a possible option for intolerance to steroid-sparing drugs (azathioprine, mercaptopurine, MMF). However, it requires increased doses of the drug and, accordingly, leads to serious side effects2,4,11.
4.3. Azathioprine
metabolism and pharmacogenetic optimization
Precision pharmacogenomics has revolutionized the
personalization of drug prescriptions in AIH management by exploring how
individual genetic variations influence drug response, focusing primarily on
pharmacokinetics and pharmacodynamics.
The main goal is to optimize therapeutic efficacy and minimize adverse drug reactions by tailoring treatments and risk assessments to an individual’s genetic profile, where genome-wide association studies (GWAS) remain the gold standard for identifying pharmacogenomic associations, whilst focusing on identifying single nucleotide polymorphisms (SNPs) that influence the development and progression of AIH.
The tolerance of azathioprine is primarily due to the genetically determined features of its metabolism. Thiopurine methyltransferase (TPMT) is the most studied enzyme of thiopurine metabolism. Another enzyme involved in the metabolism of azathioprine is the protein NUDT15, the member of hydrolases family.
In routine practice in patients with AIH, the determination of TPMT and NUDT15 gene polymorphisms before starting azathioprine therapy is not carried out due to the rare occurrence of mutant alleles, although it may be useful for individual patients in case of drug intolerance. In addition, even the presence of a "wild" type of TPMT or NUDT15 does not exclude the possibility of side effects, since usually only the most common polymorphisms are identified, not all allelic variants are known, and, finally, there may be other reasons for the development of undesirable effects in a particular patient
Insufficient effectiveness of treatment may be due to the individual characteristics of azathioprine metabolism. In this case, its dose can be adjusted based on the determination of metabolites: 6-thioguanine nucleotides (6-TGN), including the main active compound 6-thioguanine triphosphate (6-TGTP), and 6-methylmercaptopurine metabolites (6-MMP), which do not have an immunosuppressive effect, but reflect the toxicity of the drug. At the level of 6-TGN <220 pmol per 8×108 erythrocytes (associated with failure to achieve stable biochemical remission), the patient's adherence to treatment should be assessed. If the patient follows the therapy regimen, it is recommended to increase the dose of azathioprine. If the level of 6-TGN is low, but the content of 6-MMP is high, the metabolism of azathioprine should be adjusted by prescribing a uric acid inhibitor, allopurinol (at a dose of 100 mg /day), which reduces the formation of 6-MMP. In this case, the dose of azathioprine is reduced to 1/4 of the initial dose. Next, the dose of azathioprine is gradually increased while taking allopurinol. The dose of azathioprine required for optimal drug interaction is close to 2-3 mg/ kg body weight, which provides a good safety profile. If it is impossible to measure 6-TGN, the mean erythrocyte volume (MCV) indicator can be used as a surrogate marker. When taking a sufficient dose of azathioprine, it should increase, exceeding ULN19,20.
The preferred regimen for azathioprine intolerance is the use of MMF in combination with prednisone. MMF is an immunosuppressive, selective, non-competitive reversible inhibitor of inosine monophosphate dehydrogenase. MMF is recommended to be administered at an initial dose of 0.5 g 2 times a day, followed by an increase in the dose to 1 g 2 times a day. Both MMF and 6-mercaptopurine (6-MP) were capable of maintaining biochemical response in patients with AIH who were intolerant to azathioprine, with no clear evidence of inferiority of either treatment. While MMF was generally better tolerated, 6-MP may present a safe and effective treatment option in women of reproductive age (see below). In addition, therapy with 6-MP enables clinicians to monitor drug metabolite levels, titrate dosages, and monitor adherence12.
4.4. Maintenance
therapy
After achieving a complete biochemical response, to
maintain remission, it is recommended to use prednisone and azathioprine (or
MMF) in minimally effective doses with regular monitoring of laboratory
parameters of disease activity (for the first six months - 1 time per month,
then 1 time every 3-6 months). Optimally, maintenance monotherapy with
azathioprine should be considered. During reducing doses, it is necessary to
focus on laboratory indicators of disease activity (ALT, AST, IgG)2,3.
4.5. Management of
insufficient or no response
First of all, if
there is an insufficient response or lack of a biochemical response to 1st or
2nd line therapy, it is recommended to clarify compliance and re-examine to
exclude alternative causes of the disease and overlap syndromes.
With good compliance and the absence of overlap syndromes, a 3rd-line treatment with calcineurin inhibitors (tacrolimus, cyclosporine) is prescribed with a gradual selection of an individual dose under control of blood concentration: for cyclosporine, an estimated dose of 2 mg / kg 2 times a day, with blood concentration <120 ng / ml; for tacrolimus, the initial dose is 1 mg 2 times a day, an estimated dose of 0.1 mg / kg 2 times a day (for the prolonged form - 1 time a day), with blood concentration of 6-8 ng /ml. When prescribing calcineurin inhibitors, special attention should be paid to monitoring renal function due to their potential nephrotoxicity and glycemic levels. In addition, the risk of opportunistic infections should be considered10.
4.6. Rescue therapy
“Rescue therapy” is
considered if line 3 therapy is ineffective and should be administered after
consilium. When choosing a drug, it should be borne in mind that there are
descriptions of the positive effect on the course of AIH of the following
drugs: rituximab, infliximab, belimumab, barcitinib. The greatest experience
has been gained in adult patients with AIH in the use of rituximab. The drug
improves laboratory parameters in 70% of patients during the two-year follow-up
period, while simultaneously reducing the dose of prednisone in 2/3 of patients
and is characterized by a good safety profile. The algorithm of
immunosuppressive therapy in AIH is shown in (Figure 6).
Figure 6: Algorithm of immunosuppressive therapy in autoimmune hepatitis.
The flowchart outlines first‑, second‑ and third‑line immunosuppressive therapy (IST) options, including induction regimens with prednisone in combination with azathioprine or mycophenolate mofetil, criteria for complete, incomplete and absent response, and subsequent treatment steps in cases of intolerance, inadequate response, and relapse. It also summarizes pathways towards long‑term maintenance therapy, prolonged IST, and referral for liver transplantation, providing a practical overview of stepwise decision‑making in AIH management.
4.7. Withdrawal of
therapy and relapse management
When deciding whether
to discontinue IST in a patient with AIH in order to prevent recurrence, the
following indicators are taken into account: complete biochemical response
within two years; during the period of consideration of discontinuation of
therapy, ALT activity is less than half of ULN and IgG level is no more than 12
g/l; absence of non-inflammatory activity in liver tissue (histological
activity index <4), if performed control liver biopsy.
To prevent reactivation of AIH and decompensation of liver disease, it is recommended not to cancel IST in patients with AIH in patients with LC. When IST resumes after a recurrence of AH, subsequent attempts to discontinue treatment in almost all patients lead to a new relapse.
When deciding whether to discontinue IST in a patient with AIH in order to prevent recurrence, the following indicators are taken into account: complete biochemical response within two years; during the period of consideration of discontinuation of therapy, ALT activity is less than half of ULN and IgG level is no more than 12 g/l; absence of non-inflammatory activity in liver tissue (histological activity index <4), if performed control liver biopsy10,12.
5. Management in special clinical scenarios
5.1. Acute severe AIH
In patients with
acute severe AIH, but without signs of severe hepatic encephalopathy (HE),
prednisone therapy at an initiating dose of 1-1.5 mg/kg (intravenously or
orally) is considered. If the serum bilirubin level is below 100 µmol/l,
azathioprine may be added. The appointment of MMF is not recommended. Given the
risk of developing systemic infections, which reaches 20%, as well as the
overall high mortality rate in this group, such patients should be included on
the LT waiting list2,21. If severe coagulopathy develops and HE grade III–IV
LT should be performed as soon as possible; prescribing prednisone is advisable
in cases of unavailability of emergency transplantation (Figure 2).
The MELD (Model for End-Stage Liver Disease) index and its modifications in patients with acute severe AIH are evaluated before the start of therapy, then on the 3rd and 7th days of therapy and upon deterioration of the condition to predict the course, determine the need for treatment and evaluate the response to treatment. Additionally, a SURFASA score is recommended on day 3.
MELD = 11.2 x ln
(INR) + 9.6 x ln (creatinine, mg/dl) + 3.8 x ln (bilirubine, mg/dl) + 6.4
ln – natural
logarithm, INR – international
normalized ratio.
Interpretation:
MELD ≥15 – indication
for inclusion in the LT waiting list.
SURFASA = [–6,80 +
1,92 ∗ (D0-INR) + 1,94 ∗ (%3-INR) + 1,64 ∗ (%3-bilirubin)]
D0-INR – change in
INR at the time of initiation of treatment, %3-INR – percentage change in INR
by day 3, %3-bilirubin – percentage change in bilirubin by day 3.
Interpretation:
A score of less than
-0.9 indicates a 75% chance of responding to treatment; above 1.75— the risk of
death or LT is 85-100%.
Figure 7: Management algorithm for acute severe autoimmune hepatitis.
The scheme depicts the evaluation and treatment of patients with acute severe AIH, starting from diagnosis and assessment of liver failure (LF) and hepatic encephalopathy (HE), through initiation of high‑dose prednisone, and dynamic risk stratification using MELD and SURFASA scores at baseline, day 3 and day 7. It defines thresholds for inclusion on the liver transplantation waiting list, identifies non‑responders who require urgent transplantation, and delineates the pathway for continued IST in responders.
5.2. AIH with liver
cirrhosis
All patients with compensated
LC (Child-Pugh class A), regardless of histological activity, are recommended
to undergo standard IST.
The transition from compensated CP to uncompensated is a critical moment in the course of the disease, because patients experiencing complications such as ascites, HE, and bleeding from varices of the upper gastrointestinal tract. It is necessary to minimize the risk of ineffective treatment and take into account the prognostic factors affecting survival.
The decision on the appointment of IST in patients with subcompensated LC (Child-Pugh class B) is made individually based on the results of the examination, weighing the risks of complications of treatment, disease progression and the likely benefits of treatment. At the same time, age, concomitant pathology, laboratory and instrumental research results, and compliance should be taken into account. As a rule, it is no longer advisable to prescribe IST to patients with decompensated LC (Child-Pugh class C).
Clinical data show that in some patients with uncompensated LC due to AIH, treatment with glucocorticoids can lead to recompensation of cirrhosis, which improves survival. A decrease in serum bilirubin and MELD levels during the 1st week of treatment is primarily considered as response criteria.
The possibility of using other immunosuppressive drugs for LC, such as azathioprine, MMF, and calcineurin inhibitors, is being discussed. These drugs can provide long-term immunosuppression with a lower risk of prednisone-related side effects, but they also carry a risk of infection and toxicity, especially in patients with advanced liver disease.
atients who do not respond to therapy during week 1, primarily with a significant decrease in total bilirubin levels, have a poor prognosis without LT2-4
5.3. Overlap
syndromes
In AIH–PBC overlap, treatment
typically includes a combination of prednisone, azathioprine and UDCA at 13-15
mg/kg/day, whereas AIH–PSC overlap requires IST for AIH and PSC-directed
management, as summarized in (Figure 8)2.
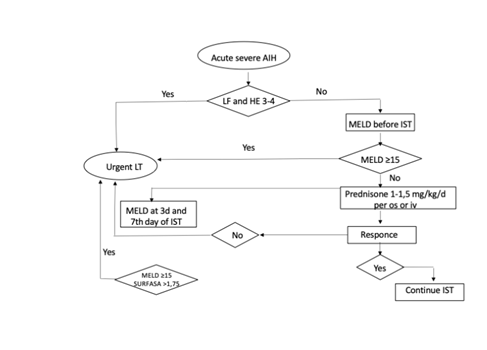
Figure 8. Diagnostic and therapeutic algorithm for patients with autoimmune hepatitis (AIH) and cholestatic features and/or ulcerative colitis (UC).
The flowchart integrates serum alkaline phosphatase (ALP) and gamma‑glutamyltranspeptidase (GGT) levels, the presence of UC, magnetic resonance cholangiopancreatography (MRCP), and antimitochondrial and PBC‑specific antibodies (AMA‑M2, anti‑gp210, anti‑sp100) to differentiate isolated AIH from AIH–primary biliary cholangitis (AIH–PBC) and AIH–primary sclerosing cholangitis (AIH–PSC) overlap syndromes, guiding subsequent treatment with immunosuppressive therapy and ursodeoxycholic acid (UDCA).
5.4. Other special
populations
Treatment of
AIH in elderly patients
It is recommended to consider induction therapy in elderly patients in the absence of contraindications with prednisone at a dose of no more than 0.5 mg/kg/day in combination with azathioprine at a dose of 1-2 mg/kg /day. In the presence of comorbid conditions that determine the risk of undesirable use of systemic glucorticoids and the absence of contraindications, it is recommended to use budesonide 9 mg/day in combination with azathioprine at a dose of 1-2 mg/kg/ day in elderly patients with AIH without LC.
In elderly patients with AIH, the prescribing of IST is determined based on the patient's constitutional status, the presence of concomitant pathology, disease status (severity of liver fibrosis, inflammatory activity), and biological age. When deciding on the appointment of IST for elderly patients, it is recommended to highlight certain categories of patients: with the presence of osteoporosis, diabetes, other components of the metabolic syndrome and other significant concomitant pathology at the onset of AIH. In such patients, it is recommended to pre-evaluate the possibilities of a response to IST and the risk of its side effects.
Treatment
of elderly patients with mild AIH without severe fibrosis is not strictly
mandatory and depends on an assessment of the balance between efficacy and
risks. For the remaining elderly patients (in the absence of acute severe AIH
or decompensated LC), the following principles should be used when deciding on
the appointment of IST:
· If there is a high probability of prednisone-related worsening of poorly controlled diabetes mellitus, osteoporosis, psychoemotional disorders and psychiatric diseases, hypertension, cataracts, it is recommended to consider prescribing budesonide at a dose of 9 mg /day and azathioprine 1-2 mg/kg /day in patients without LC;
· Reduce the prednisone dose as quickly as possible.
An initial assessment is required, followed by careful monitoring of bone mineral density, drug prevention and treatment of osteoporosis3,11.
Various variants of the course of AIH associated with pregnancy are described. According to available data, exacerbation of AIH during pregnancy is noted in 7-12% of cases, in the postpartum period - in 11-81% of cases. Also, in rare cases, both the onset of AIH during pregnancy and spontaneous remission are possible.
It is advisable to discuss with the patient the effect of the course of AIH and the drugs used to treat it on the health of the fetus and mother before pregnancy. Pregnancy is recommended to be planned when the biochemical response to treatment has been maintained for at least 1 year. Preservation of the biochemical response in AIH with the help of drug therapy during pregnancy is associated with its favorable outcomes and does not significantly affect the frequency of miscarriages, stillbirths, and prolongation of the gestational period. It is necessary to continue supportive IST in pregnant patients with AIH, since its cessation may lead to a relapse of the disease. In the absence of proper monitoring, AIH can be activated during pregnancy, which is associated with a high level of fetal and maternal complications (10-20%)
It is recommended to continue maintenance therapy with prednisone and/ or azathioprine throughout pregnancy and lactation to ensure control over the activity of the disease.
With AIH, the overall incidence of maternal complications during pregnancy or within 12 months after delivery is 38%, while premature birth is mainly due to the refusal of adequate treatment. The average live birth rate is 73%. Preterm birth occurs in about 20% of pregnancies, but no specific AIH-related birth defects have been described. Prematurity or fetal loss is also more often associated with poor control of AIH activity, including discontinuation of therapy before or during pregnancy
The
use of prednisone and azathioprine in patients with AIH who are planning
pregnancy seems safe. Studies on the course of AIH during pregnancy have not
revealed a negative effect of glucocorticoid and azathioprine therapy on the
fetus. Taking azathioprine is also considered safe during breastfeeding.
Although small amounts of their metabolites can enter breast milk, this does
not cause any complications in infants and does not affect their further
development.
MMF cannot be used during pregnancy and should be discontinued at least 3 months before conception planning due to the increased risk of embryo loss in the first trimester and congenital anomalies (most often ear defects, heart defects, and cleft palate). It is recommended to perform a pregnancy test within 1 week after taking MMF and use two effective methods of contraception for 4 weeks before and 6 weeks after using MMF. A small amount of MMF is found in the milk of nursing mothers.
It is recommended to monitor the activity of the disease in patients with AIH during pregnancy and after childbirth: ALT, AST, total bilirubin and the level of immunoglobulin G in the blood once a trimester and 12 weeks after delivery for detection possible recurrence of AIH and correction doses of IST. AIH relapses are three times more common after childbirth, which underscores the need for more careful monitoring and follow-up of patients. Loss of the biochemical response to IST is quite common after childbirth, when the level of estrogen in a woman's blood returns to normal. In this situation, an increase in IST should be considered - a short-term increase in prednisone doses2,3.
5.6. Treatment
of overlap syndromes AIH and cholestatic liver disease
No significant differences were found in the biochemical response and effect on the prognosis for life in patients receiving monotherapy with UDCA, IST (prednisone ± azathioprine) and their combination in patients with mild and moderate interface hepatitis with overlap syndrome AIH/PBC. At the same time, severe interface hepatitis was a predictor of a good biochemical response to combination therapy. In this regard, with moderate non-inflammatory activity, an alternative regimen is acceptable: UDCA monotherapy with a decision on the appointment of IST after 3 monthses with insufficient biochemical response (Figure 3)
The issue of prescribing IST to patients with AIH/PSC overlap syndrome remains very difficult. This is due not only to the absence of large randomized studies, but also to a higher risk of complications, including infectious ones, as well as a worse response to therapy and prognosis for the life of patients in comparison with AIH. In this regard, in clinical practice, the decision to prescribe IST to patients with AH/PSC should be made individually based on the leading clinical and morphological pattern, taking into account the possible risks of complications of concomitant diseases.
IST is indicated for patients with AIH/PSC who meet the histological criteria for diagnosis, i.e. with moderate to severe necroinflammatory activity. The average dose of prednisone is 0.5 mg/kg/day, azathioprine 50-75 mg/day, UDCA 13-15 mg/kg/day (Figure 3). At the same time, in the long term (more than 10 years), in most cases, the disease progresses to LC. In children with PSC/AIH (in pediatrics, the term "autoimmune sclerosing cholangitis" is used) who received IST, there was a biochemical response and a decrease in signs of inflammation in liver biopsies, however, cholangiographic changes progressed, and the 10-year survival rate without LT was lower (65%) than with classical AIH (100%)2.
5.7. Treatment
of AIH associated with immune-mediated diseases
According to cohort studies, immuno-mediated diseases
are detected in 14-44% of patients with AIH. It should be noted that the
milder, subclinical course of comorbid AIH immune-mediated diseases is more
typical. However, in some cases the severity of their manifestations dominates,
which requires appropriate correction of therapeutic tactics.
There is a wide range of immunosuppressive drugs for which a positive effect on the course of AIH has been demonstrated in small non-randomized studies, a series of clinical cases, or isolated clinical observations. These include, in descending order of evidence, calcineurin inhibitors (cyclosporine A, tacrolimus), m-TOR inhibitors (sirolimus), antibodies to TNF-α (infliximab, adalimumab) and B-cell receptors (rituximab, belimumab), as well as methotrexate
MMF is a standard therapeutic option in the treatment of patients with diffuse connective tissue diseases, methotrexate is used in the treatment of patients with rheumatoid arthritis, and anti-TNFα is used in the treatment of inflammatory bowel diseases3,4.
5.8. Treatment
of AIH associated with metabolically associated steatotic liver disease
Taking into account the negative effect of glucocorticoids on diabetes and other components of the metabolic syndrome, along with the classic IST regimen, a faster reduction in prednisone dose and lower doses during induction, as well as the use of budesonide along with the early addition of steroid-sparing drugs, can be used. Short-term use of glucocorticoids, even at the standard doses recommended for AIH, does not seem to have a significant side effect in patients with AIH and MAFLD, however, it requires monitoring of the condition by patients. When treating patients with AIH and MAFLD receiving IST, strict adherence to all prescriptions aimed at correcting the components of the metabolic syndrome in combination with lifestyle modification (reducing calorie intake, avoiding foods containing processed fructose, minimizing alcohol intake and increasing physical activity) is necessary.
Taking prednisone at a dose of >10 mg/day for more than 6-8 months can lead to a worsening of the course of diseases associated with MAFLD, and therefore requires careful monitoring and timely review of therapy aimed at the treatment of diabetes and hypertension3,4.
5.9. Treatment
of AIH in combination with viral hepatitis
If the patient has AIH and HCV or hepatitis B virus HBV replication is detected, treatment begins with the use of interferon-free antiviral therapy, taking into account the stage of fibrosis, and after elimination or suppression of the hepatotropic virus, the possibility of prescribing IST should be considered.
HBV reactivation can occur in both chronic hepatitis B and resolved HBV infection and is defined as an increase or rediscovery of HBV DNA. Patients receiving IST are at risk of HBV reactivation: 17-55% of patients who were previously exposed to HBV during and after IST are at risk of HBV reactivation. In the initial HBsAg positivity, the proportion increases to 100% with the development of acute liver failure in 15% of cases.
HBV
reactivation is defined as follows:
·
Upon initial detection of
HBsAg:
o
≥ 2 log increase in HBV DNA
level from baseline;
o
detection of HBV DNA at a
level > 100 IU/ml in a patient with initially undetectable HBV DNA levels;
·
With resolved HBV infection
(HBsAg-negative, anti-HBc-positive):
o
Reverse HBsAg seroconversion
(HBsAg turns from negative to positive);
o The appearance of HBV DNA in the absence of HBsAg, previously undetectable HBV DNA is detected.
It is difficult to determine the specific risk of a particular drug or medication regimen due to the lack of systematically collected data and the exclusion of patients with HBV infection. The risk of HBV reactivation in patients with AIH who receive treatment with traditional prednisone and azathioprine regimens is unknown. Traditional immunosuppressants (azathioprine, mercaptopurine, methotrexate, low-dose cyclosporine), as well as prednisone therapy for ≥4 weeks in autoimmune diseases, were associated with a low (<1%) risk for both HBsAg-positive and HBsAg-negative/anti-HBcore-positive patients
The risk increases in HBsAg-negative and anti-HBcore-positive patients with an increase in the dose and duration of prednisone treatment. High-dose therapy with glucocorticoids, B-cell antibodies, cytokine antagonists, and calcineurin inhibitors increases the risk of reverse seroconversion, which should be considered in HBsAg-negative/anti-HBcore-positive patients when considering the feasibility of prescribing proactive antiviral treatment. Reverse seroconversion (detection of HBsAg and HBV DNA in a previously HBsAg-negative patient) was observed mainly in patients treated with IST from groups of moderate and high risk of HBV reactivation: rituximab, anti-TNFα and chemotherapeutic agents. The optimal duration of antiviral therapy during rituximab treatment should be at least 12 months.
5.10. Treatment
of AH in combination with HIV infection
Treatment of AIH in HIV-infected patients should be individual, taking into account the possible risks and benefits of therapy. Standard IST is effective, but it is necessary to point out the possibility of developing infectious complications2-4.
5.11. Treatment
of drug-induced autoimmune-like hepatitis
In cases where it is difficult to differentiate between DI-ALH and AIH, it is recommended to prescribe prednisone at a dose of 0.5–1 mg/kg/day, followed by withdrawal and prolonged monitoring of the patient. The course of prednisone for DI-ALH is usually 1-2 months. Other IST regimens have been developed for steroid-refractory hepatotoxicity, including MMF, cyclosporine, tacrolimus, and anti-lymphocytic globulin.
The main difference between DI-ALH and true AIH is the absence of relapses. All patients who have been diagnosed with DI-ALH are recommended to have long-term follow-up with ALTdetection every 6 months for three years2.
5.12. Treatment of autoimmune hepatitis after liver
transplantation
Diagnosis of relapse is quite difficult due to the lack of specific markers. In addition, IST may mask some of the signs of the underlying disease. A recurrence of AIH in transplant recipients should be suspected with increased transaminase activity, or the appearance of unmotivated fever, or the appearance of weakness and fatigue. In rare cases, relapse may result in symptoms such as jaundice, abdominal pain, skin rashes, and arthralgia. A recurrence of AIH may be indicated by an increase in the level along with the detection of ANA and/or ASMA.
The main histopathological feature of AIH recurrence after LT is the pronounced activity of the lymphocytic periportal process with or without infiltration by plasma cells in the liver biopsy. Other pathological changes include acute lobular hepatitis with focal necrosis of hepatocytes, acidophilus bodies with lymphoplasmocytic cells, pseudorozetts hepatocytes, perivenular lymphoplasmocytic inflammation, and piecemeal and bridge necroses with lymphoplasmocytic infiltration.
If a recurrence of AIH is suspected after LT, differential diagnosis with T-cell rejection, drug hepatotoxicity, de novo steatohepatitis, and viral hepatitis should be performed to determine treatment tactics.
The recurrence of AIH after LT requires immediate treatment, since almost half of the cases are resistant to therapy and lead to graft rejection. In mild cases, the doses of maintenance therapy are increased., In severe cases prednisone is prescribed at a dose of at least 30 mg/day and azathioprine 1-2 mg /kg/ day, followed by a gradual decrease in prednisone doses. A combination of prednisone and MMF may also be used. When laboratory parameters improve, the prednisone dose is reduced to 5-10 mg for 1-2 months. In cases with severe hepatic failure, repeated LT may be required, which has to be performed in 33-60% of patients with recurrent AIH4,12
6. Novel and Experimental PPM‑Oriented Approaches
6.1. Biologics and
targeted immunomodulators
In this sense, novel immunomodulatory agents, a class
of drugs that regulate the body's immune functions, have been confirmed to
possess properties that modulate immune balance and induce immune tolerance. In
recent years, these agents, including including immune cell nucleic acid
inhibitors, calmodulin phosphate inhibitors, mammalian target of rapamycin
inhibitors, TNF-α inhibitors, IL-2, anti-CD20 mAbs, and B cell-activating
factor inhibitors, have played an increasingly significant role in the clinical
management of AIH18.
Of particular interest are macrophages as pivotal players in AIH pathogenesis, attributable to their multifaceted roles in inflammation amplification, immune regulation, and fibrogenesis. Through complex interactions with T lymphocytes and hepatic stellate cells, macrophages orchestrate a pathological milieu promoting inflammation and fibrosis. Notably, diverse programmed cell death (PCD) modalities-autophagy, necroptosis, pyroptosis, and ferroptosis-not only determine macrophage survival and functional phenotype but also significantly impact cytokine release, phenotypic plasticity, and the trajectory of immunopathological progression. Comprehensive elucidation of the interplay between macrophage immunological activity and programmed cell death pathways promises to inform novel, PPM-guided therapeutic approaches for AIH patients22.
6.2. Small molecules and gut microbiota‑driven strategies
New investigations aim to explore safer and more
specific therapeutic candidates such as Garcinone E (GE) against concanavalin-A
(Con-A)-induced AIH - pretreatment with GE significantly reduces key serum
markers and histopathological liver lesions and demonstrates potent
hepatoprotective effects, possibly attributed to its anti-inflammatory,
antioxidant, and anti-apoptotic properties, which modulate HO-1/Nrf2 signaling
and suppress the NF-κB-mediated inflammatory cascade and TNF-α/JNK-induced
apoptosis pathway23
Another type of PPM-driven strategy using gut microbiota and their metabolic products (thiopurine metabolites), significantly influencing AIH progression, can also optimise treatment regimens in AIH, resulting in fewer adverse drug reactions (ADRs) whilst maintaining biochemical (metabolite) remission (BR) - gut microbiota-derived metabolites modulate Treg/Th17 balance by shaping immune cell differentiation and function, thereby revealing novel therapeutic opportunities - the latter is promising treatment strategies, including the use of probiotic supplementation, engineered prebiotic compounds, microbiota transfer procedures, and specific medications targeting gut microorganisms and their byproducts. These approaches could potentially reduce immune-triggered hepatic damage, offering potential new avenues for AIH management and simultaneously outlining priorities for future research, focusing on multi-OMICS integration, the development of individualized therapeutic strategies, and rigorous clinical evaluation, to facilitate the development of safe and effective microbiota-based therapies for AIH23-25. Moving forward, large-scale, longitudinal studies integrating metagenomics, metabolomics, and host genomic data are needed to establish subtype-specific microbial markers and assess the efficacy of targeted interventions such as probiotics and bacteriophage therapy in AIH management and treatment.
6.3. Stem cell and
MSC‑based therapies
Emerging therapies, including SC treatments and
immunomodulatory agents, show potential to revolutionize AIH patient care26.
Stem cell (SC) therapies have been investigated as treatment for a variety of AIH subtypes in latest years6.
MSC transplantation, is widely used in treating different liver diseases (including AIH), focusing on the therapeutic mechanisms, including differentiation into hepatocyte-like cells, immunomodulating function with a variety of immune cells, paracrine effects via the secretion of various cytokines and extracellular vesicles, and facilitation of homing and engraftment. MSCs have great potential in the treatment of AIH based on their multi-faceted characteristics, and more accurate mechanisms and novel therapeutic strategies stemming from MSCs will facilitate clinical practice7.
For instance, MSC-exosome therapy appears to be promising, and the use of these bodies may be able to assist in lowering the risk of MSCs administration27. Methods for delivering MSC-derived exosomes (MSC-Exos), more specifically into target areas must be developed28,29. However, to yet, no clinical investigations relying on MSC treatment have been conducted. Therefore, the clinically beneficial effect of MSCs in therapy of AIH needs approval, and further basic and clinical studies focused on SC therapies for AIH are required.
6.4. MSC‑derived
exosomes and antifibrotic strategies
AIH provokes activation of myofibroblasts to secrete
ECM proteins, resulting in the formation of fibrous scars on the liver.
Fibrosis regression is possible through the targeted removal of
pathophysiological causes as well as the elimination of activated
myofibroblasts, resulting in the reabsorption of the scar tissue. To date, a
wide range of antifibrotic therapies has been tried and tested, including
growth factors, cytokines, miRNAs, mAbs, SC-based approaches, and other
approaches that target the ECM28,30. In this context, of particular interest is the use
of MSC-Exos, which have shown promise in treating AIH, and as microvesicles
with low immunogenicity, high safety, and permeability, can deliver RNA, DNA,
proteins, lipids, and various drugs for disease treatment, showing promising
clinical application prospects. The above-mentioned approach highlights the
significant regulatory effects of MSC-Exos on immune cells and their ability to
modify the microenvironment, demonstrating anti-inflammatory and anti-fibrotic
properties while promoting liver regeneration in AIH patients6,26,27. MSCs and
MSC-secreted exosomes can be used as an alternative therapy for treating AIH,
especially for liver cirrhosis and liver failure28-30
Moreover, therapeutic effect and underlying mechanism of MSC-Exos treatment on CD4+ T-cell overactivation and associated inflammatory liver injury in AIH, provoked and induced by disordered glucose metabolism, can be dependent on exosome-mediated mitochondrial protein transfer between cells. This study highlights that MSC-EV therapy may represent a new avenue for treating AIH.
The beneficial effects of MSCs mainly rely on cell-to-cell communication through the secretion of extracellular vesicles (EVs) and soluble factors. MSC-derived EVs (MSC-EVs) could modulate adjacent and distinct cells in the microenvironment by transferring various DNA, mRNA, ncRNAs, proteins, and lipids from parent cells to recipient cells. Compared to fluid, MSC-EVs delivery can protect miRNAs from the degradation of ribonucleases, ensuring that miRNAs negatively regulating multiple target genes at the post-transcriptional level and being involved in AIH, are able to perform their respective crucial roles in AIH recipient cells and thus to demonstrate their therapeutic challenges31,32.
6.5. miRNA‑based
targets
Regarding miRNAS,
special attention would have to be focused on hsa-miR-122-5p, as a potential
therapeutic target, promoting ferroptosis by inhibiting the expression of
SLC7A11 and thus providing new insights into the pathogenesis of AIH and paving
the way for innovative treatment strategies31,32.
Exos secreted from MSCs provide protection for Con A-induced liver injury - protective effect of Exos is mediated via efficient improving the accumulation of Exos in AIH in the liver and can illustrate the Exos as a promising drug carrier for AIH and exhibit MSC-Exos nanotherapeutic potential in AIH23,29.
6.6. MSC‑Exos as drug
carriers and immunoregulatory MSCs
Of special attention is a significance of PD-L1 and
ICAM1 expression levels of MSCs which empower MSCs immuno-regulation and could
be regarded as a bioindicator to choose optimal MSCs for treating AIH33-35. Those empowered
MSCs are highly effective treatment for AIH - those findings provide support
for the translation of the results to the clinic for treatment of AIH patients7,26,30,36
7. Unmet Needs and Future Directions
PPM focuses on
personalized predictive and preventive measures that contribute to the
development of individualized strategies for controlling AIH. To achieve the
implementation of the PPM concept, it is necessary to combine the assets of the
newest advances in basic science, design-driven translational applications with
clinical medicine, followed by the introduction and promotion of new
generation’s translational applications.
Innovations like multi-targeted therapies, immunotherapies, comprehensive genomics and pharmacogenomics testing, NGS and bioinformatics have shown promising advancements in improving AIH patient outcomes and potential cures. However, further biodesign-driven research and translational applications are essential to refine PPM-guided treatment strategies and validate the impact of genetic variations on therapeutic responses. Future Hi-Tech therapeutic agents are anticipated to be more selective, targeting both autoimmune destructions and host factors, demonstrating the advantage of PPM in managing and eradicating AIH17.
8. Conclusion
9. Abbreviations
6-MP –
6-mercaptopurine
AIH – autoimmune
hepatitis
ALP – alkaline
phosphatase
ALT – alanine
aminotransferase
AMA –
antimitochondrial antibodies
ANA – antinuclear
antibodies Anti-LC-1 – liver cytosol type 1 antibodies
Anti-LKM-1 – liver
kidney microsomal type 1 antibodies
Anti-SLA/LP – soluble
liver antigen/liver-pancreas antibodies
ASMA – smooth muscle
antibody
AST – aspartate
aminotransferase
autoAbs –
autoantibodies
DI-ALH – drug induced
autoimmune-like hepatitis
DNA –
deoxyribonucleic acid
GGT – gamma glutamyl
transpeptidase
GWAS – genome-wide
association studies
HBV – hepatitis B
virus
HCV – hepatitis C
virus
HE – hepatic
encephalopathy
HIV – human
immunodeficiency virus
HLA – Human leukocyte
antigens
Ig – immunoglobulin
INR – International
normalized ratio
IST –
immunosuppressive therapy
MAFLD – metabolic
dysfunction-associated fatty liver diseaseMHC – major
histocompatibility complex
MMF – mycophenolate
mofetil
MRCP – magnetic
resonance cholangiopancreatography
pANCA – perinuclear
antineutrophil cytoplasmic antibodies
PBC – primary biliary
cholangitis
PSC – primary
scclerosing cholangitis
RNA – ribonucleic
acidTh – T-helper(s)
TNF – tumor necrosis
factor
TPMT –thiopurine
S-methyltransferase
UDCA –
ursodeoxicholic acid
ULN – upper limit of
normal
References
1.
Vargas A, Harris C. Biomarker development in the
precision medicine era: lung cancer as a case study. Nat Rev Cancer.
2016;16:525–537.
2.
Tanaka A. Autoimmune Hepatitis: 2019 Update. Gut
Liver. 2020;14:430–438.
3.
Mack CL, Adams D, Assis DN, Kerkar N, Manns MP, Mayo
MJ, et al. Diagnosis and Management of Autoimmune Hepatitis in Adults and
Children: 2019 Practice Guidance and Guidelines from the American Association
for the Study of Liver Diseases. Hepatology. 2020;72:671–722.
4.
Sierra R, Marenco-Flores A, Alsaqa M, Barba R,
Cuellar-Lobo M, Barberan C, et al. Autoimmune Hepatitis Management: Recent
Advances and Future Prospects. Livers. 2024;4:240–252.
5.
Sirbe C, Simu G, Szabo I, Grama A, Pop TL.
Pathogenesis of Autoimmune Hepatitis – Cellular and Molecular Mechanisms. Int J
Mol Sci. 2021;22(24):13578.
6.
Chen L, Lu FB, Chen DZ, Wu JL, Hu ED, Xu LM, et al.
BMSCs-derived miR-223-containing exosomes contribute to liver protection in
experimental autoimmune hepatitis. Mol Immunol. 2018;93:38–46.
7.
Chen L, Zhang N, Huang Y, Zhang Q, Fang Y, Fu J, et
al. Multiple Dimensions of Using Mesenchymal Stem Cells for Treating Liver
Diseases: From
Bench to Bedside. Stem Cell Rev Rep. 2023;19(7):2192–2224.
8.
Lu FB, Chen DZ, Chen L, Hu ED, Wu JL, Li H, et al.
Attenuation of experimental autoimmune hepatitis in mice with bone mesenchymal
stem cell-derived exosomes carrying microRNA-223-3p. Mol Cell.
2019;42(12):906–918.
9.
Weltzsch JP, Ziegler A, Lohse A. Autoimmune Hepatitis:
Von Autoantikörpern bis Zirrhose. Inn Med (Heidelb). 2023;64(7):655–667.
10.
Muratori L, Lohse AW, Lenzi M. Diagnosis and
management of autoimmune hepatitis. BMJ. 2023;380:e070201.
doi:10.1136/bmj-2022-070201.
11.
Liberal R, de Boer YS, Heneghan MA. Established and
novel therapeutic options for autoimmune hepatitis. Lancet Gastroenterol
Hepatol. 2021;6(4):315
326.
12.
Reau NS, Lammert CS, Weinberg EM. Autoimmune
hepatitis: Current and future therapies. Hepatol Commun. 2024;8(6):e0458.
13.
Arinaga-Hino T, Ide T, Akiba J, Suzuki H, Kuwahara R,
Amano K, et al. Growth differentiation factor 15 as a novel diagnostic and
therapeutic marker for
autoimmune hepatitis. Sci Rep. 2022;12:8759.
14.
Yao M, Wang L, Wang J, Liu Y, Liu S, Zhao J, et al.
Diagnostic Value of Serum Golgi Protein 73 for Liver Inflammation in Patients
with Autoimmune
Hepatitis and Primary Biliary Cholangitis. Dis Markers.
2022;2022:4253566. doi:10.1155/2022/4253566.
15.
Wang L, Yao M, Liu S, Yang D, Wen X, Ning J, et al.
Serum Golgi Protein 73 as a Potential Biomarker for Hepatic Necroinflammation
in Population with
Nonalcoholic Steatohepatitis. Dis Markers.
2020;2020:6036904. doi:10.1155/2020/6036904.
16.
Reddy V. Using AI To Identify Biomarkers That
Facilitate Personalized Medicine. Proxzar Blog. 2019 Nov 25. Available at:
https://www.proxzar.ai/blog/using-ai-to-identify-biomarkers-that-facilitate-personalized-medicine.
17.
Arroyave E, Saldarriaga OA, Bhatti S, Bergman I,
Graham R, Tana M, et al. Integrating Molecular Testing With Clinical Criteria
and Histopathology Improves Diagnostic Precision in Immune-Mediated Liver
Diseases. Mod Pathol. 2025;38(5):100728.
18.
Li J, Wang H, Lin J, Wang A, Miao S, Liu H. The Role
of Novel Immunomodulators in the Treament of Autoimmune Hepatitis. J Clin Transl
Hepatol. 2025;13(6):493–503.
19.
Candels LS, Rahim MN, Shah S, Heneghan MA. Towards
personalised medicine in autoimmune hapatitis: Measurement of thiopurine
metabolites results in higher biochemical response rates. J Hepatol.
2021;75(2):324–332. doi:10.1016/j.jhep.2021.03.023.
20.
Bolia R, Srivastava A. The search for optimum
thiopurine metabolite levels in autoimmune hepatitis continues. J Hepatol.
2022;76(1):220–222. doi:10.1016/j.jhep.2021.06.040.
21.
Nagesh VK, Martinez E, Badam S, Harrison JL, Basta M,
Varughese VJ, et al. Management of acute liver failure – an updated literature
review. World
Crit Care Med. 2025;14(4):108840.
22.
Liu T, Wang Y, Huang Y, Zhao R, Shen H. Macrophages in
Autoimmune Liver Diseases: From Immune Homeostasis to Precision-Targeted
Therapy.
Biomedicines. 2025;13(10):2520.
23.
Zhang X, Wan L, Zheng Y, Ai Y. Gut Barrier, Microbial
Metabolites, and Immune Homeostasis in Autoimmune Hepatitis: From Molecular
Mechanisms to
Strategies. Front Biosci (Landmark Ed). 2025;30(5):27747.
24.
Li G, Xiong Y, Li Z, Yu Q, Li S, Xie J, et al. Gut
microbiota-derived metabolites modulate Treg/Th17 balance: novel therapeutic
targets in autoimmune diseases. Front Immunol. 2025;16:1710733.
25.
Iftikhar F, Iftikhar A, Khalid M, Talha M, Waafira A.
Gut microbial signatures in autoimmune hepatitis: unlocking diagnostic and
therapeutic potential. Ann
Med Surg (Lond). 2025;87(10):6868–6869.
26.
Lotfy A, Elgamal A, Burdzinska A, Swelum AA, Soliman
R, Hassan AA, et al. Stem cell therapies for autoimmune hepatitis. Stem Cell
Res Ther.
2021;12(1):386.
27.
Wu L, Zhang L, Huang M, Wu Y, Jin S, Zhang Y, et al.
Mesenchymal Stem Cell-Derived Exosomes: Emerging as a Promising Cell-Free
Therapeutic
Strategy for Autoimmune Hepatitis. Biomolecules. 2024;14(11):1353.
28.
Shen M, Zhou L, Fan X, Wu R, Liu S, Deng Q, et al.
Metabolic Reprogramming of CD4+ T Cells by Mesenchymal Stem Cell-Derived
Extracellular Vesicles
Attenuates Autoimmune Hepatitis Through Mitochondrial
Protein Transfer. Int J Nanomedicine. 2024;19:9799–9819.
29.
Li L, Liu Y, Wang K, Mo J, Weng Z, Jiang H, et al.
Stem cell exosomes: new hope and future potential for relieving liver fibrosis.
Clin Mol Hepatol.
2025;31(2):333–349.
30.
Jangra A, Kothari A, Sarma P, Medhi B, Omar BJ,
Kaushal K. Recent Advancements in Antifibrotic Therapies for Regression of
Liver Fibrosis. Cells. 2022;11(9):1500.
31.
Liu H, Chen Y, Yin G, Xie Q. Therapeutic prospects of
microRNAs carried by mesenchymal stem cell-derived extracellular vesicles in
autoimmune
diseases. Life Sci. 2021;277:119458.
32.
Suo Y, Wang Y, Su Y, Wang Q, Jia J, Zhao X. MiR-122-5p
modulates ferroptosis via SLC7A11: A potential therapeutic target in autoimmune
hepatitis. J
Transl Autoimmun. 2025;11:100303.
33.
Bai X, Chen T, Li Y, Ge X, Qiu C, Gou H, et al. PD-L1
expression levels in mesenchymal stromal cells predict their therapeutic values
for autoimmune
hepatitis. Stem Cell Res Ther. 2023;14(1):370.
34.
Bai X, Liu T, Li C, Qiu C, Ge X, Gou H, et al. PD-L1
and ICAM1 overexpression empowers immunoregulation of mesenchymal stromal cells
to improve the
autoimmune hepatitis treatment efficacy. Stem Cell Res Ther.
2025;16(1):209.
35.
Zhao J, Li Y, Jia R, Wang J, Shi M, Wang Y.
Mesenchymal Stem Cells-Derived Exosomes as Dexamethasone Delivery Vehicles for
Autoimmune Hepatitis
Therapy. Front Bioeng Biotechnol. 2021;9:650376.
36.
Yang F, Ni B, Liang X, He Y, Yuan C, Chu J, et al.
Mesenchymal stromal cell-derived extracellular vesicles as nanotherapeutics for
concanavalin A-induced hepatitis: modulating the gut–liver axis. Stem Cell Res
Ther. 2025;16(1):4