Design and Performance Evaluation of Memantine Hydrochloride Orodispersible Tablets Incorporating Natural and Synthetic Superdisintegrants
Abstract
Introduction
This study aimed to develop memantine hydrochloride orodispersible tablets (ODTs) to improve medication compliance in geriatric Alzheimer’s patients who often face swallowing difficulties.
Materials and methods
ODTs were formulated by direct compression using synthetic superdisintegrants (sodium starch glycolate, croscarmellose sodium) and a natural alternative (mucilage from Plantago ovata) at concentrations of 0.5%, 1% and 2%. The formulations were evaluated for flow properties, hardness, friability, wetting time, disintegration time and in-vitro drug release.
Results
The optimized formulation (MM3), containing 2% Plantago ovata mucilage, demonstrated the most efficient performance with a rapid disintegration time of 28 ± 1.29 sec and 98.83 ± 2.11% drug release within 30 min.
Conclusion
The use of natural Plantago ovata mucilage as a super disintegrant provides a superior, biocompatible alternative to synthetic agents, ensuring rapid drug onset and enhanced patient adherence.
Keywords: Alzheimer’s Disease, Memantine hydrochloride orodispersible tablets, Plantago ovata mucilage
1. Introduction
Alzheimer’s
disease is a progressive neurodegenerative disorder characterized by cognitive
decline, memory impairment and behavioural disturbances, significantly
affecting the geriatric population1.
The pathophysiology involves neuronal degeneration and excitotoxicity mediated
by excessive glutamate activity, necessitating long-term pharmacological
intervention2. Memantine
hydrochloride, an N-Methyl-D-aspartate (NMDA) receptor antagonist, plays a
crucial role in modulating glutamatergic transmission and is widely prescribed
for moderate to severe Alzheimer’s disease due to its neuroprotective effects3,4.
Despite
therapeutic benefits, effective disease management is often compromised by poor
patient compliance, particularly in geriatric populations. Age-associated
physiological conditions such as dysphagia, reduced saliva secretion and
impaired neuromuscular coordination make swallowing conventional solid dosage
forms challenging. Additionally, cognitive impairment further reduces adherence
to medication regimens. These limitations highlight the need for advanced
patient-centric drug delivery systems that ensure ease of administration and
rapid onset of action. Recent
progress in these delivery systems has been driven by advances in materials and
technology, enabling improved therapeutic efficiency5.
Orodispersible
tablets (ODTs) have emerged as a promising and widely accepted alternative to
conventional oral dosage forms6,7.
These formulations rapidly disintegrate in the oral cavity without the need for
water, thereby improving patient compliance and convenience8,9. Recent advancements in ODT technology
have focused on enhancing disintegration efficiency, mechanical strength and
drug release profiles through the use of novel excipients and optimized
formulation strategies, including direct compression techniques and the
incorporation of efficient superdisintegrants9,10.
Furthermore,
the development of biomolecule-responsive systems, such as specialized
hydrogels, represents the cutting edge of personalized medicine in this field11.
Super
disintegrants play a critical role in ODT performance by facilitating rapid
tablet breakup through mechanisms such as swelling, wicking and deformation12,13. Synthetic super disintegrants like
sodium starch glycolate and croscarmellose sodium are widely used due to their
effectiveness; however, growing concerns regarding cost, biocompatibility and
sustainability have driven interest toward natural alternatives14,15. In recent years, natural polymers
have gained significant attention as pharmaceutical excipients owing to their
biodegradability, non-toxicity, economic feasibility and eco-friendly nature16,17.
Among
natural candidates, mucilage derived from Plantago ovata (Isabgol) has shown considerable promise as a super
disintegrant due to its high swelling index, rapid hydration capacity and
excellent water absorption properties18.
Recent studies have demonstrated that Plantago
ovata mucilage can enhance disintegration and dissolution performance,
sometimes exhibiting comparable or superior efficiency to synthetic
superdisintegrants19,20. However,
systematic comparative evaluations in specific therapeutic contexts,
particularly for central nervous system drugs like memantine hydrochloride,
remain limited.
Therefore,
the present study aims to develop and evaluate orodispersible tablets of
memantine hydrochloride using Plantago
ovata mucilage as a natural super disintegrant and to compare its
performance with commonly used synthetic super disintegrants such as sodium
starch glycolate and croscarmellose sodium21.
The study focuses on optimizing formulation parameters to achieve rapid
disintegration, enhanced drug release and improved patient compliance. This
work contributes to the growing field of natural excipient-based drug delivery
systems and provides a potential strategy for developing cost-effective and
patient-friendly formulations for geriatric care.
2. Materials
and Methods
Memantine
hydrochloride was obtained from Sun Pharmaceuticals Industries Ltd, India, as a
gift sample. Croscarmellose sodium, sodium starch glycolate, microcrystalline
cellulose, mannitol, aspartame and magnesium stearate were procured from SD
Fine Chem Ltd., Mumbai, India. Plantago ovata seeds were obtained from Yarrow
Chem Products, Mumbai, India.
2.1.
Procedure
2.1.1.
Isolation of mucilage: The seeds of Plantago
ovata were soaked in distilled water for 48 h and then boiled for a few minutes
to completely release the mucilage. The mixture was filtered through
a muslin cloth to separate the marc. An equal volume of acetone was
added to the filtrate to precipitate mucilage. The obtained mucilage
was dried at a temperature below 60°C, powdered, passed through a sieve no # 80
and stored in a desiccator until further use.
2.1.2.
Preparation of tablets: Memantine hydrochloride
is intended to exert immediate therapeutic effects. Oral dispersible tablets
were formulated using super-disintegrants to ensure rapid disintegration. In
this study, the mucilage of Plantago ovata, sodium starch glycolate and croscarmellose
sodium were used as super-disintegrants. The tablets were prepared using the
direct compression method. A total of nine formulations were prepared, as
presented in (Table 1).
Table 1: Formulations of memantine hydrochloride containing different
super-disintegrants.
|
Formulation |
Memantine HCl (mg) |
SSG (mg) |
CCS (mg) |
Mucilage powder (mg) |
MCC (mg) |
Magnesium stearate (mg) |
Talc (mg) |
Mannitol (mg) |
Aspartame (mg) |
Total (mg) |
|
MSG1 |
5 |
0.25 |
- |
- |
29.75 |
1 |
1 |
11 |
2 |
50 |
|
MSG2 |
5 |
0.5 |
- |
- |
29.5 |
1 |
1 |
10 |
2 |
50 |
|
MSG3 |
5 |
1 |
- |
- |
29 |
1 |
1 |
9 |
2 |
50 |
|
MCS1 |
5 |
- |
0.25 |
- |
29.75 |
1 |
1 |
11 |
2 |
50 |
|
MCS2 |
5 |
- |
0.5 |
- |
29.5 |
1 |
1 |
10 |
2 |
50 |
|
MCS3 |
5 |
- |
1 |
- |
29 |
1 |
1 |
9 |
2 |
50 |
|
MM1 |
5 |
- |
- |
0.25 |
29.75 |
1 |
1 |
11 |
2 |
50 |
|
MM2 |
5 |
- |
- |
0.5 |
29.5 |
1 |
1 |
10 |
2 |
50 |
|
MM3 |
5 |
- |
- |
1 |
29 |
1 |
1 |
9 |
2 |
50 |
2.2. Analytical methodology
2.2.1. Determination of λmax:
A 10 µg/mL solution of memantine hydrochloride was
scanned using a double-beam UV-visible spectrophotometer, with 0.1N hydrochloric acid serving as the blank. The
maximum absorption wavelength (λmax)
was identified at 254 nm, which was selected
for all further analytical studies22,23.
2.2.2. Construction of calibration
curve: Standard solutions with concentrations of 2, 4, 6,
8 and 10 µg/mL were prepared by appropriate
dilution of Stock III, while the 20 µg/mL solution
was derived from Stock II. The absorbance of each concentration was measured at
254 nm using 0.1N hydrochloric acid as the
reagent blank. All measurements were performed in hexaplicate (n=6) and the
resulting mean absorbance values were utilized to construct the calibration
curve22,23.
2.2.3. Fourier transform infrared spectroscopy: The compatibility
between the drug and excipients was studied using FTIR spectroscopy. The
spectra of pure drug and its physical mixtures with excipients were recorded in
the range of 400 to 4000 cm⁻¹. The obtained
spectra were analysed for characteristic peaks corresponding to functional
groups and were compared to detect any possible interactions24.
2.2.4. Angle of repose: The angle of repose was
measured using the fixed-funnel technique to assess powder flowability. A known
weight of the powder mix was allowed to flow freely through a funnel onto a
stationary surface, maintaining the funnel tip at the apex of the powder cone.
The height and diameter of the pile were recorded and the angle of repose was
determined according to the formula25,26.
where
h is the height and r is the radius of the pile.
2.2.5. Bulk density and tapped density:
Bulk density was determined by pouring the powder blend into a graduated
cylinder and measuring the bulk volume. It was calculated as the ratio of the
mass of powder to the bulk volume.
Tapped density was
determined by tapping the graduated cylinder containing the powder blend for a
fixed number of times until a constant volume was obtained. It was calculated
as the ratio of the mass of powder to tapped volume. Bulk density and tapped
density were determined using the given formulae25,26.
2.2.6. Carr’s compressibility index and
hausner’s ratio: The
compressibility index was calculated using the formula25,26
Hausner’s ratio was
calculated using the following formula
2.3. Evaluation of orodispersible
tablets
2.3.1. Hardness: Tablet hardness was
determined using a Monsanto hardness tester, which measures the force required
to break the tablet and indicates its mechanical strength27,28. The hardness is
expressed in terms of kg/cm2.
2.3.2. Thickness: The thickness of
tablets was measured using a screw gauge for 10 tablets and expressed as mean ±
standard deviation. It ensures uniformity and proper packaging27,28.
2.3.3. Weight variation: Twenty tablets were
randomly selected and weighed individually. The average weight was calculated
and the percentage deviation of each tablet from the average was determined
according to pharmacopeial limits27,28.
2.3.4. Friability: Friability was
evaluated using a Roche friabilator operated at 25 rpm for 100 revolutions.
Tablets were weighed before and after the test and percentage weight loss was
calculated to assess resistance to abrasion by the given formula27,28.
2.3.5. Content uniformity: Tablets were
powdered and an amount equivalent to the drug dose was dissolved in 0.1N HCl,
filtered and analyzed at 254 nm using a UV spectrophotometer27-29.
2.3.6. Wetting time and water
absorption ratio: A tablet was placed on tissue paper soaked in water containing a dye
and the time required for water to reach the upper surface of the tablet was
recorded as wetting time27,28.
The tablet was
weighed before and after water absorption and the water absorption ratio was
calculated using the standard formula. Water absorption ratio R was determined
using the following equation30,31.
Where Wa = weight of tablet after absorption,
Wb = weight of tablet before absorption.
2.3.7. In-vitro disintegration time: The tablet was placed
in a petri dish containing 10 mL of water at 37°C and the time taken for
complete disintegration into fine particles was recorded32.
2.3.8. Swelling index: The
swelling capacity of various superdisintegrants was evaluated to determine
their contribution to rapid tablet disintegration. The swelling index, defined
as the volume (mL) occupied by 1 g of material, including any adhering
mucilage, after 4 h of hydration in an aqueous medium, was measured for Plantago ovata, sodium starch
glycolate and croscarmellose sodium. All measurements were performed in
triplicate and the final index for each substance was recorded as the mean
value33.
2.3.9. In-vitro drug release: In vitro drug release was
evaluated using a USP dissolution apparatus II (paddle type). The study was
conducted in 900 mL of 0.1 N hydrochloric acid, maintained at 37 ± 0.5°C with a
paddle rotation speed of 50 rpm. At specified intervals (5, 10, 15, 20, 25 and
30 min), 5 mL aliquots were withdrawn and immediately replaced with an equal
volume of fresh medium to maintain sink conditions. Collected samples were
filtered and analysed via UV spectrophotometry at 254 nm. The cumulative
percentage of drug release was determined using a standard calibration curve to
characterize the dissolution profile and release kinetics of the formulated
tablets34.
2.3.10. Similarity and difference
factors: To
evaluate the comparability of the optimized formulation and the marketed
product, dissolution profiles were analysed using model-independent approaches:
the difference factor (f1) and the similarity factor (f2). While f1 calculates
the percentage difference between the two profiles at each time point to
indicate relative error, f2 provides a logarithmic reciprocal square root
transformation of the sum-of-squared error, representing the closeness of the
profiles. According to regulatory standards, f1 values between 0-15 and f2
values between 50-100 confirm that the optimized formulation exhibits
bioequivalent drug release behaviour relative to the marketed reference35,36.
3. Results and Discussion
3.1. Determination of λmax
The analytical method for Memantine hydrochloride was developed to determine
its absorption maximum (λmax) and to enable accurate quantification
of the dispersions before experimental studies. The drug was scanned in 0.1 N
hydrochloric acid over an appropriate wavelength range to identify its λmax.
An absorption maximum was observed at 254 nm, as shown in Figure 1.
Subsequently, a standard calibration curve of memantine hydrochloride in 0.1 N
hydrochloric acid was constructed at the determined λmax for
quantitative analysis37.
Figure 1: Absorption maximum of Memantine Hydrochloride
in 0.1N HCl.
3.2. Standard
graph of memantine hydrochloride in 0.1N HCl
Standard
solutions of Memantine hydrochloride in
the concentration range of 2-20 µg/mL were prepared and their absorbance was
measured at 254 nm using an appropriate blank. A calibration curve was
constructed by plotting absorbance versus concentration (Figure 2), with
absorbance on the Y-axis and concentration on the X-axis37.
Figure 2: Standard graph of memantine hydrochloride in
0.1N HCl.
3.3. Fourier transform infra-red
spectroscopy
The FTIR spectra
shown in (Figure 3) inferred that the characteristic peaks of the drug
were indicated in the formulation FTIR spectrum, implying the drug excipient
stability.
Fourier
Transform Infrared (FTIR) spectroscopy was performed to investigate the
potential interactions between Memantine hydrochloride and the selected
excipients, namely sodium starch glycolate, croscarmellose sodium and mucilage
of Plantago ovata. The spectra of the pure drug, individual excipients and
their physical mixtures were recorded and compared.
The
characteristic absorption peaks of memantine hydrochloride were observed at
786.39 cm⁻¹ corresponding to CH₃ stretching vibrations, 1352 cm⁻¹
attributed to C–N stretching, 1646 cm⁻¹ indicating amide functional
groups and 2940 cm⁻¹ corresponding to C–H stretching vibrations. These prominent peaks were
retained in the spectra of the drug–excipient mixtures without any significant
shift, disappearance or formation of new peaks.
The FTIR
spectra of the formulations (Figure 3) demonstrated that all major
characteristic peaks of the drug were preserved in the presence of excipients,
indicating the absence of any chemical interaction between the drug and the
excipients. Minor variations in peak intensity were observed, which may be
attributed to physical mixing and dilution effects rather than chemical
incompatibility38.
Figure 3: FTIR spectra of (A) Memantine hydrochloride (Drug) (B)Drug + SSG (C)
Drug + CCS (D) Drug+ Plantago ovata mucilage.
Overall,
the FTIR analysis confirms the compatibility and stability of memantine
hydrochloride with the selected excipients, supporting their suitability for
the formulation of immediate-release dosage forms.
3.4. Pre-compression parameters
As presented
in (Table 2), the pre-compression evaluation of all formulation blends
included parameters such as angle of repose, bulk density, tapped density,
Carr’s compressibility index and Hausner’s ratio to assess flowability and
compressibility, which are critical for direct compression. The angle of repose
values ranged from 23.52 ± 0.98° to 25.26 ± 1.03°, indicating excellent flow
properties (<30°). Carr’s compressibility index values were found to be
between 11.24 ± 1.49 and 14.53 ± 0.93, suggesting good compressibility. The
Hausner’s ratio was approximately 1.17 for all blends, remaining within
acceptable limits (<1.25), thereby further confirming good flowability.
Overall, the powder blends demonstrated satisfactory flow and compressibility
characteristics, making them suitable for direct compression39.
Table 2: Precompression parameters of the powder blend
of all formulations of SSG, CCS and mucilage of Plantago ovata.
|
Formulation |
Angle of repose(?)* |
Bulk density (gm/cm3)* |
Tapped density (gm/cm3)* |
Hausner’s ratio * |
Compressibility Index (%) * |
|
MSG1 |
25.26±1.03 |
0.642±0.014 |
0.735±0.004 |
1.144±0.019 |
12.58±1.520 |
|
MSG2 |
23.52±0.98 |
0.646±0.006 |
0.735±0.009 |
1.137±0.003 |
12.09±0.233 |
|
MSG3 |
24.78±0.82 |
0.617±0.004 |
0.722±0.003 |
1.170±0.013 |
14.53±0.926 |
|
MCS1 |
24.89±0.80 |
0.634±0.005 |
0.720±0.008 |
1.136±0.022 |
11.99±1.739 |
|
MCS2 |
24.21±0.72 |
0.645±0.005 |
0.742±0.005 |
1.150±0.001 |
13.24±0.169 |
|
MCS3 |
24.62±0.53 |
0.652±0.012 |
0.740±0.003 |
1.134±0.021 |
11.89±0.562 |
|
MM1 |
23.89±0.92 |
0.669±0.024 |
0.757±0.002 |
1.131±0.019 |
11.62±0.327 |
|
MM2 |
24.47±0.92 |
0.641±0.004 |
0.727±0.002 |
1.134±0.004 |
11.88±0.332 |
|
MM3 |
24.97±0.86 |
0.630±0.005 |
0.710±0.006 |
1.126±0.019 |
11.24±1.491 |
Values are expressed
as Mean ±SD, *n = 3, MSG= Formulations of SSG, MCS= Formulations of CCS, MM =
Formulation of mucilage
3.5.
Evaluation of the memantine HCl orodispersible tablets
In this study orodispersible tablets of
memantine hydrochloride were formulated using synthetic superdisintegrants,
namely sodium starch glycolate and croscarmellose sodium, along with a natural
superdisintegrant, Plantago ovata mucilage. All
formulations were evaluated for hardness, friability, thickness, weight
variation, drug content, wetting time, water absorption ratio, disintegration
time and in vitro drug release.
As shown in (Table 3), the hardness of
the tablets ranged from 3.0 ± 0.62 to 3.1 ± 0.64 kg/cm², while friability was
below 1%, indicating adequate mechanical strength. The thickness varied between
2.59 ± 0.04 and 2.77 ± 0.02 mm. All formulations complied with pharmacopeial limits
for weight variation (±10%). Drug content ranged from 99.3 to 101.13%,
confirming uniform distribution of the drug across all batches40.
Table 3: Evaluation of memantine HCl orodispersible tablets.
|
Formulation |
Weight variation (mg)**** |
Hardness (kg/cm2)** |
Thickness (mm)*** |
Friability (% )* |
Wetting time (sec)** |
Water absorption ratio** |
DT (sec)** |
Content uniformity (%)* |
|
MSG1 |
50± 1.19 |
3.1±0.64 |
2.77±0.02 |
0.27±0.12 |
72±1.21 |
110±1.23 |
83±1.21 |
101.13±0.73 |
|
MSG2 |
52±1.12 |
3.0±0.62 |
2.59±0.04 |
0.23±0.18 |
56±1.92 |
118±1.29 |
76±1.96 |
99.98±0.69 |
|
MSG3 |
52±0.99 |
3.1±0.53 |
2.65±0.01 |
0.21±0.12 |
44±1.08 |
121±1.30 |
62±1.23 |
99.80±1.23 |
|
MCS1 |
51±1.05 |
3.0±0.39 |
2.71±0.02 |
0.22±0.15 |
62±1.29 |
132±1.42 |
53±1.28 |
99.32±1.45 |
|
MCS2 |
49±1.10 |
3.1±0.32 |
2.69±0.01 |
0.25±0.24 |
53±1.02 |
135±1.46 |
48±1.18 |
100.82±1.12 |
|
MCS3 |
51±1.10 |
3.1±0.69 |
2.70±0.02 |
0.26±0.18 |
40±1.26 |
137±1.38 |
39±1.13 |
99.98±1.10 |
|
MM1 |
50±0.89 |
3.1±0.29 |
2.76±0.05 |
0.24±0.12 |
56±1.24 |
139±1.26 |
48±1.52 |
99.86±0.89 |
|
MM2 |
49±0.98 |
3.1±0.12 |
2.79±0.05 |
0.28±0.21 |
42±1.76 |
144±1.81 |
36±1.34 |
99.24±0.58 |
|
MM3 |
50±1.11 |
2.9±0.57 |
2.64±0.02 |
0.24±0.16 |
28±1.91 |
152±1.43 |
28±1.29 |
99.85±0.86 |
Values are expressed as Mean ±SD, *n=3, **n = 6, ***n=10, ****n= 20.
MSG - formulations of sodium starch glycolate, MCS – formulations of
croscarmellose, MM – formulations of mucilage powder.
3.6. Determination of swelling index
The mucilage
of Plantago ovata exhibited a significantly
higher swelling index (Table 4) compared to the synthetic super
disintegrants, sodium starch glycolate and croscarmellose sodium, indicating
its superior water uptake and swelling capacity, which may contribute to faster
tablet disintegration41.
Table 4: Swelling index of super-disintegrants.
|
S.No |
Name of super-disintegrants |
Swelling index (%v/v) |
|
1 |
Mucilage of Plantago ovata |
89±2.1 |
|
2 |
Crosscarmellose
sodium |
74±1.3 |
|
3 |
Sodium starch
glycolate |
67±1.6 |
Values are expressed
as mean ± S.D, n=3.
3.7. Disintegration time
Disintegration time is a critical parameter in
the development of orodispersible tablets. In the present study, the
disintegration time of all batches ranged from 28 ± 1.52 to 83 ± 1.21 sec
(Table 4), complying with the official requirement of less than 3 min for
dispersible tablets. Figure 4 illustrates the disintegration behaviour of the
tablets in water.
A decrease in disintegration time was observed
with increasing concentrations of Plantago ovata
mucilage, croscarmellose sodium and sodium starch glycolate, indicating the
effectiveness of these super disintegrants in promoting rapid tablet
disintegration. However, for the synthetic super disintegrants, croscarmellose
sodium and sodium starch glycolate, this reduction in disintegration time was
evident only up to the maximum studied concentration of 2%.
Among all formulations, batch MM3, containing
2% Plantago ovata mucilage, was identified
as the optimized formulation, exhibiting the shortest disintegration time of 28
sec. The results also demonstrated a direct relationship between swelling index
and disintegration efficiency, suggesting that higher swelling capacity
enhances the disintegration process. Based on the obtained results, formulation
MM3 was considered optimal, as it combined rapid disintegration with
satisfactory tablet properties42.
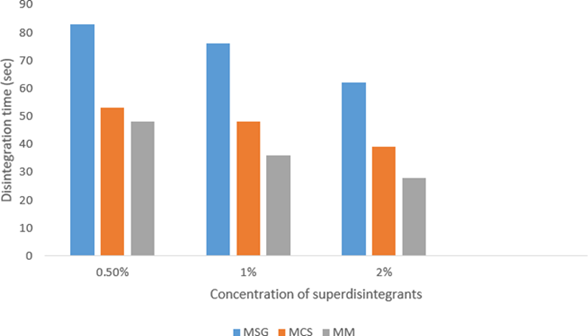
Figure 4: Disintegration time of different super
disintegrants with different concentrations.
3.8. Wetting time
(Figure 5) depicts the relationship between
the concentration of superdisintegrants and wetting time. Wetting time was
evaluated as an indirect parameter to correlate with disintegration behaviour
in the oral cavity. It serves as an important criterion for understanding the
ability of superdisintegrants to absorb moisture and swell in the presence of a
limited amount of water.
Figure 5: Wetting time of different super
disintegrants with different concentrations.
Since the
dissolution process of a tablet depends on initial wetting followed by
disintegration, rapid wetting facilitates the penetration of the aqueous medium
into the tablet matrix. This process replaces the air adsorbed on the
particles, weakens intermolecular interactions and promotes the breakdown of
the tablet into finer particles. However, disintegration is a multifactorial
process influenced by both wetting and swelling mechanisms. The wetting time of
the formulated tablets ranged from 28 ± 1.91 to 72 ± 1.21 sec42.
3.9. Water absorption ratio
(Figure 6) illustrates the relationship
between the concentration of super disintegrants and the water absorption
ratio. The water absorption ratio was evaluated to assess the moisture sorption
and water uptake capacity of the super disintegrants. An increase in water
absorption ratio was observed with increasing concentrations of super
disintegrants, accompanied by a corresponding decrease in both disintegration
time and wetting time. This indicates that enhanced water uptake facilitates
rapid tablet hydration and promotes faster disintegration. The water absorption
ratio of the formulated tablets ranged from 110 ± 1.23 to 152 ± 1.43.
Figure 6: Water absorption
ratio of different superdisintegrants with different concentrations.
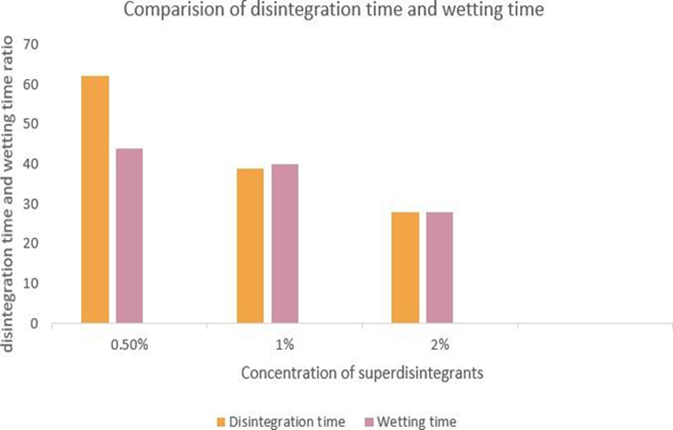
Figure 7: Comparison of disintegration time and wetting
time of MSG3, MCS3, MM3.
Disintegration
time and wetting time of formulations MSG3, MCS3 and MM3 were compared (Figure
7). Among these, formulation MM3 exhibited the shortest disintegration time
and wetting time compared to the other formulations42.
3.9. Invitro drug release studies
The in vitro drug release studies were conducted for all the formulations
containing different super disintegrants. (Table 5) and (Figure 8) indicate
that maximum drug release was observed at 30 min. Among all the formulations,
the optimized formulation containing Plantago ovata mucilage at 2%
concentration reported 98.83
± 2.11 at 30 min42.
Table 5: Cumulative Percentage Drug Release of
Different Formulations (Mean ± SD, n = 6).
|
Time
(min) |
MSG1 |
MSG2 |
MSG3 |
MCS1 |
MCS2 |
MCS3 |
MM1 |
MM2 |
MM3 |
Market
formulation |
|
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
5 |
85.29±1.87 |
85.37±2.27 |
85.24±1.87 |
90.86±0.69 |
91.12±1.31 |
91.82±1.89 |
89.89±2.12 |
89.87±1.28 |
89.85±1.37 |
80.25±0.92 |
|
10 |
89.21±1.92 |
89.26±2.87 |
89.63±2.19 |
93.25±0.87 |
92.82±1.26 |
92.95±1.28 |
92.46±2.54 |
92.52±1.53 |
93.14±1.46 |
81.42±1.12 |
|
15 |
90.28±0.98 |
90.34±2.96 |
90.83±1.97 |
94.13±1.23 |
93.49±1.20 |
93.98±1.86 |
94.76±2.98 |
94.78±1.39 |
94.85±2.31 |
83.73±1.98 |
|
20 |
91.25±1.11 |
92.32±1.19 |
92.98±2.34 |
94.37±1.41 |
94.79±1.12 |
95.12±1.22 |
95.28±1.92 |
95.86±1.23 |
95.92±1.21 |
88.05±1.54 |
|
25 |
94.19±1.86 |
94.58±1.27 |
95.28±1.23 |
98.22±1.29 |
96.99±0.98 |
96.87±1.62 |
96.62±1.28 |
96.98±1.99 |
96.92±2.34 |
90.41±1.19 |
|
30 |
96.92±1.28 |
97.13±1.28 |
97.98±2.39 |
99.79±2.57 |
98.42±0.82 |
98.97±1.62 |
98.52±2.34 |
98.75±2.13 |
98.83±2.11 |
96.64±0.89 |
Figure 8: Dissolution profiles of all 9 Memantine HCl
orodispersible tablet formulations (MSG1, MSG2, MSG3, MCS1, MCS2, MCS3, MM1,
MM2, MM3).
3.10. Calculation
of dissimilarity (f1) and similarity factor (f2)
In vitro drug release studies showed that all
formulations achieved more than 90% drug release within 30 min. The optimized
formulation, MM3, exhibited a drug release of 98.83 ± 2.11%, while the marketed
formulation showed 96.64 ± 0.89% release within the same time period (Table
6).
The dissolution profile of the optimized formulation was compared with that of the marketed formulation using similarity (f2) and difference (f1) factors. The calculated f1 and f2 values were found to be 9.62 and 52.46, respectively (Table 6). For two dissolution profiles to be considered similar, the f1 value should be between 0 and 15 and the f2 value should lie between 50 and 100. The obtained values indicate that the optimized formulation is comparable to the marketed formulation in terms of dissolution performance43.
Table 6:
Calculation of dissimilarity (f1) and similarity factor (f2).
|
Time (min) |
Reference |
Test |
Rt-Tt |
(Rt-Tt)² |
|Rt-Tt| |
|
NAMENDA |
MM3 |
||||
|
0 |
0.00 |
0.00 |
0.00 |
0.00 |
0.00 |
|
5 |
80.25 |
90.86 |
-10.61 |
112.57 |
10.61 |
|
10 |
81.42 |
93.25 |
-11.83 |
139.94 |
11.83 |
|
15 |
83.73 |
94.13 |
-10.4 |
108.16 |
10.4 |
|
20 |
88.05 |
94.37 |
-6.32 |
39.94 |
6.32 |
|
25 |
90.41 |
98.22 |
-7.81 |
60.99 |
7.81 |
|
30 |
96.64 |
99.79 |
-3.15 |
9.92 |
3.15 |
|
Sum |
520.5 |
|
|
471.54 |
-50.12 |
|
Number of time points or intervals excluding zero |
|
6 |
|||
|
Dissimilarity factor (f1) |
|
9.62 |
|||
|
Similarity factor (f2) |
|
52.46 |
|||
4. Summary
The
present study focused on the design and evaluation of orodispersible tablets
(ODTs) of memantine hydrochloride using both synthetic and natural super
disintegrants to enhance patient compliance, particularly in geriatric patients
with swallowing difficulties. Formulations were developed by direct compression
employing sodium starch glycolate, croscarmellose sodium and Plantago ovata mucilage at varying
concentrations.
Pre-compression
parameters indicated good flow properties and compressibility of all powder
blends, making them suitable for direct compression. Post-compression
evaluation confirmed that all formulations met pharmacopeial requirements for
hardness, friability, weight variation and drug content uniformity. Among the
formulations, those containing higher concentrations of super disintegrants
exhibited improved wetting, water absorption and faster disintegration.
The
optimized formulation (MM3), containing 2% Plantago ovata mucilage, demonstrated superior performance with
rapid disintegration time, reduced wetting time and enhanced water absorption
capacity. In vitro drug release studies revealed that all formulations achieved
more than 90% drug release within 30 minutes, with MM3 showing the highest
release profile. Comparative dissolution analysis with the marketed formulation
confirmed similarity, as indicated by acceptable f1 and f2 values.
5. Conclusion
The
present study achieved the successful formulation of orodispersible tablets of
memantine hydrochloride using both synthetic and natural super disintegrants.
Comparative evaluation of the formulations demonstrated that Plantago ovata mucilage exhibited superior performance in
terms of key parameters, including disintegration time, wetting time, water
absorption ratio and in vitro drug release. The optimized formulation (MM3),
containing 2% Plantago ovata mucilage, showed rapid
disintegration and enhanced dissolution characteristics. Furthermore, its
dissolution profile was found to be comparable to that of the marketed
formulation, as indicated by acceptable f1 and f2 values. These findings
establish Plantago ovata mucilage as an effective
natural super disintegrant for the development of orodispersible tablets.
6. Conflict of Interest
The
authors declare that there are no conflicts of interest regarding the
publication of this paper.
7. References
5. Maitra A, et al. Biomedical Engineering: Materials,
Technology and Applications. Weinheim, Germany: Wiley-VCH, 2022.
6. Goel H, Rai P.
Recent advances in orodispersible tablets. Saudi Pharm J, 2021;29(5): 441-450.
7. Zhang Y, et al.
Orodispersible tablet formulation technologies. Drug Dev Ind Pharm, 2022;48(4): 567-578.
9. Deshmukh VN, Jadhav
JK. Evaluation of wetting time in orodispersible tablets. Int J Pharm Sci Rev Res, 2021;68(2): 45-52.
10. Shah RB, Tawakkul
MA. Pharmaceutical excipients overview. Pharm
Dev Technol, 2020.
11. Lavrador P, et al.
Biomolecule-Responsive Hydrogels in Medicine. Advanced Healthcare Materials. 2017: 6:1-35.
12. Singh J, et al.
Role of super disintegrants in tablet formulation. J Appl Pharm Sci, 2021;11(6): 001-010.
13. Singh SK, et al.
Mechanisms of tablet disintegration. Pharm
Dev Technol, 2021.
14. Sharma P, Verma A.
Correlation between wetting time and disintegration. Asian J Pharm Sci, 2022;17(1): 89-96.
15. Sharma N, et al.
Natural super disintegrants in drug delivery systems. Int J Biol Macromol, 2022; 210: 552-565.
16. Bhusnure OG, et al.
Natural polymers as pharmaceutical excipients. Int J Pharm Sci Res, 2022;13(5): 2001-2010.
17. Mandal A, et al.
Overview on Natural Hydrophilic Polysaccharide Polymers in Drug Delivery. Polymers for Advanced Technologies, 2018;29:
2564-2573.
18. Pawar HA, et al.
Applications of mucilage in pharmaceuticals. Carbohydr Polym, 2021;251: 117090.
21. Kumar R, et al.
Comparative study of natural and synthetic superdisintegrants. J Drug Deliv Ther, 2022;12(2): 102381.
22. Sharma D, Singh R.
Analytical method validation in pharmaceuticals. J Pharm Biomed Anal, 2021;196: 113897.
23. Verma RK, Garg S.
Development of calibration curves in drug analysis. Drug Dev Ind Pharm, 2020.
24. Silverstein
RM, Webster FX. Spectrometric
Identification of Organic Compounds. Wiley, 2020.
25. Kulkarni GT,
Gowthamarajan K. Powder flow properties in pharmaceutics. Pharmaceutics, 2021;13(3): 350.
26. Aulton ME, Taylor
KM. Aulton’s Pharmaceutics: The Design
and Manufacture of Medicines. 5th ed. Elsevier, 2021.
27. Khan GM. Evaluation
of tablet dosage forms. J Pharm Sci,
2020;109(6): 2050-2060.
28. United States
Pharmacopoeia (USP 43–NF 38). Tablet evaluation tests. USP, 2020.
29. British
Pharmacopoeia. BP 2021. London:
TSO, 2021.
30. Deshmukh VN, Jadhav
JK. Evaluation of wetting time in orodispersible tablets. Int J Pharm Sci Rev Res, 2021;68(2): 45-52.
31. Patel DM, Shah SR.
Role of water absorption ratio in tablet disintegration. Int J Pharm Investig, 2021;11(2): 123-130.
32. Indian
Pharmacopoeia. IP 2022.
Ghaziabad: IPC, 2022.
33. Kulkarni U, Patil
BS. Evaluation of natural super disintegrants for tablet formulations. Int J Pharm Sci Res, 2020;11(5): 2345-2352.
34. Gohel MC, et al.
Dissolution studies of oral dosage forms. AAPS Pharm Sci Tech, 2020.
35. Costa P, Sousa Lobo
JM. Modelling and comparison of dissolution profiles. Eur J Pharm Sci, 2021;13(2): 123-133.
36. Shah VP, et al.
Dissolution profile comparison using similarity factor (f2). AAPS J, 2020.
37. Amin AH, El Sheikh R, Abdel Fattah GM, et al.
Spectrophotometric methods for the quantitative determination of memantine
hydrochloride. Int J Appl Pharm,
2022;14(2): 206-214.
38. Patil BS, Kulkarni
U. Drug-excipient compatibility studies using FTIR and DSC in solid dosage
forms. Int J Pharm Sci Res,
2015;6(3): 1030-1036.
39. Aulton ME, Taylor
KM. Aulton’s Pharmaceutics: The Design and Manufacture of Medicines. 5th ed.
Elsevier, 2018.
40. Ghogari IS, Jain
PS. Development of orally disintegrating tablets of memantine hydrochloride—a
remedy for Alzheimer’s disease. Int J
Appl Pharm, 2020;12(1): 147-152
41. Ashooriyan P,
Mohammadi M, Darzi GN, et al. Development of Plantago ovata mucilage-based systems and evaluation of
physicochemical properties. Int J Biol
Macromol, 2023;248: 125938.
42. Awandekar NB,
Tekade R, Dhawas S, et al. Formulation and evaluation of fast dissolving
tablets using Plantago ovata as
natural superdisintegrant. Res J Pharm
Technol, 2022;15(2): 633-638.
43. Péterfi O, Kovács
B, Casian T, et al. Comparison of surrogate models in tablet dissolution
prediction: addressing limitations of f2. AAPS J, 2025;27: 118.