From Imhotep to Medicine Security Builders: Repositioning Pharmacy for Pharmaceutical Sovereignty and Health System Resilience in Africa
Graphical
Abstract: From Pharmacy Practice to Medicine Security Builders
Conceptual description for journal submission: The graphical abstract illustrates the
transformation of pharmacy from a dispensing-centred
profession to a systems-leadership
profession that supports pharmaceutical sovereignty.
Education reform
Industrial partnerships
Regulatory strengthening
Digital pharmaceutical infrastructure
↓
Transformation Engine
Medicine Security
Builders
Pharmacists are positioned as industrial scientists, regulatory strategists, digital health innovators and pharmaceutical entrepreneurs.
↓
System Outcomes
Medicine security
Resilient pharmaceutical supply chains
Local pharmaceutical manufacturing
Regional pharmaceutical trade under AfCFTA
Improved access to affordable medicines
↓
Long-Term Impact
Pharmaceutical sovereignty
Health system resilience
Economic development and industrialisation in
Africa
The Medicine Security Builders framework illustrates the transformation of pharmacy education and practice into a strategic driver of pharmaceutical sovereignty, industrial development and medicine security across Africa.
Abstract
Background
Pharmacy is one of the oldest scientific
professions, with its roots traced to ancient Egypt under the intellectual
legacy of Imhotep. Over several millennia, the discipline evolved from herbal
preparation and compounding to industrial pharmaceutical manufacturing,
biotechnology innovation and digital health integration. Despite this long
historical evolution, pharmacy practice across much of Africa remains
disproportionately retail-oriented and highly dependent on imported medicines.
This structural imbalance weakens medicine security and constrains
pharmaceutical sovereignty.
Objective
This paper examines the historical evolution of
pharmacy. It proposes a transformation framework that repositions the
profession from producing résumé-oriented graduates to developing medicine security builders capable of
strengthening pharmaceutical manufacturing, regulatory science, digital health
integration and policy leadership.
Methods
A historical-analytical approach was used,
drawing on literature from pharmaceutical history, health systems governance,
regulatory science and pharmaceutical industrial policy.
Results
The analysis identifies key structural gaps in
pharmaceutical education, regulatory capacity and industry integration. A
conceptual model-the Medicine Security
Builders Framework-is proposed to guide the transformation of pharmacy
education and practice toward pharmaceutical sovereignty.
Conclusion
Repositioning pharmacy toward medicine security
leadership will strengthen health security, improve pharmaceutical
manufacturing capacity and enable Africa to build resilient pharmaceutical
ecosystems.
Keywords: Medicine security, Pharmaceutical sovereignty, Pharmacy evolution, Pharmaceutical manufacturing, Africa, Health systems
1.
Introduction
The intellectual origins of pharmacy can be
traced to Imhotep, the renowned
physician, architect and scholar of ancient Egypt during the 27th century BCE.
Imhotep is widely regarded as the earliest recorded physician-pharmacist and
symbolises the integration of scientific observation, medicinal formulation and
health system leadership1.
Ancient Egyptian medical texts such as the Ebers Papyrus (c. 1550 BCE) documented more than 800 medicinal formulations, demonstrating sophisticated pharmacological knowledge involving botanical, mineral and biological preparations2,3.
As previously discussed in work examining the evolution of pharmacy across civilisations, the profession has consistently adapted to scientific, technological and societal transformations4,5. From herbal medicine traditions to synthetic chemistry and biotechnology, pharmacy has evolved as both a clinical and industrial discipline.
During the Islamic Golden Age, pharmacy emerged as a specialised profession with the establishment of early pharmacies in Baghdad and Persia. Scholars such as Ibn Sina (Avicenna) formalised pharmacopoeias and dosage forms, laying the foundations for pharmaceutical standardisation6.
The industrial revolutions of the nineteenth and twentieth centuries further transformed pharmacy through advances in synthetic chemistry, microbiology and industrial pharmaceutical manufacturing7.
In the twenty-first century, pharmaceutical systems are increasingly shaped by artificial intelligence, telepharmacy, blockchain-enabled traceability and digital pharmacovigilance8.
Despite these advances globally, the pharmaceutical landscape across much of Africa remains characterised by high dependence on imported medicines, limited manufacturing capacity and fragmented pharmaceutical supply chains.
Addressing these structural challenges requires repositioning the pharmacy profession toward medicine security leadership and pharmaceutical sovereignty.
2. Medicine
Security as a Strategic Imperative
Medicine security refers to the sustained
capacity of healthcare systems to ensure reliable access to safe, effective,
quality-assured and affordable medicines.
The COVID-19 pandemic exposed the fragility of global pharmaceutical supply chains and highlighted the risks faced by regions heavily dependent on imported medicines.
Africa imports a significant proportion of finished pharmaceutical products and active pharmaceutical ingredients (APIs), leaving healthcare systems vulnerable to geopolitical disruptions, logistical bottlenecks and currency fluctuations9.
In previous work on Africa’s path to pharmaceutical sovereignty, it was argued that medicine security should be viewed as both a public health and economic development priority4.
Strengthening medicine security requires
capabilities in:
·
Pharmaceutical manufacturing
·
API synthesis
·
Regulatory science
·
Health technology assessment
·
Digital pharmaceutical supply chains
· Industrial pharmaceutical financing
These capabilities demand a pharmacy workforce trained beyond traditional dispensing roles.
3. From
Résumé Builders to Medicine Security Builders
Pharmacy education in many African universities
continues to focus primarily on preparing graduates for employment within community and hospital pharmacy practice.
While these roles remain vital, they are insufficient to support pharmaceutical industrialisation and medicine security.
A new orientation is therefore required. Pharmacy graduates must evolve from résumé builders seeking employment to medicine security builders capable of strengthening national pharmaceutical ecosystems.
This expanded professional identity requires
competencies in:
·
Pharmaceutical formulation science
·
API synthesis and industrial chemistry
·
Regulatory science and dossier development
·
Pharmacoeconomics and health technology assessment
·
Digital health systems
· Pharmaceutical entrepreneurship
Pharmacy education must therefore align more directly with the strategic goal of pharmaceutical sovereignty.
4. The
Medicine Security Framework
Medicine security can be conceptualised as a
systems architecture consisting of interconnected components that collectively
ensure reliable access to medicines.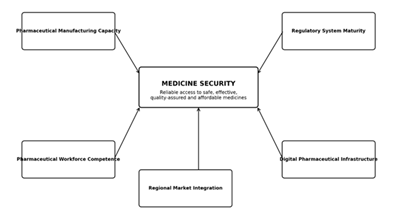
Figure 1: Medicine Security Framework.
The Medicine Security Framework illustrates five interdependent pillars required for resilient pharmaceutical systems: pharmaceutical manufacturing capacity, regulatory system maturity, pharmaceutical workforce competence, digital pharmaceutical infrastructure, and regional market integration. Together, these pillars enable reliable access to safe, effective, quality-assured and affordable medicines while supporting pharmaceutical sovereignty and health system resilience.
5. Pharmacy
Workforce Transformation Model
Transforming pharmacy education requires a
shift from traditional practice roles toward a diversified pharmaceutical
workforce capable of supporting industrial development.
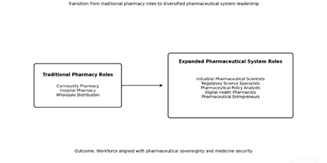
Figure 2: Pharmacy Workforce Transformation Model.
The model illustrates the transition of pharmacy practice from traditional roles centred on community and hospital pharmacy toward diversified professional roles within pharmaceutical manufacturing, regulatory science, policy leadership, digital health systems and pharmaceutical entrepreneurship. This transformation aligns the pharmacy workforce with the broader objective of strengthening medicine security and pharmaceutical sovereignty.
This transformation aligns pharmacy education with broader national development priorities.
6. The
Medicine Security Builders Framework 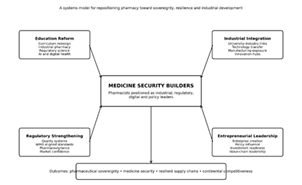
Figure 3: The Baale Medicine Security Builders Framework (Baale Model).
The Baale Model conceptualises pharmacy transformation through four mutually reinforcing pillars-education reform, industrial integration, regulatory strengthening and entrepreneurial leadership. These pillars reposition pharmacists as medicine security builders capable of strengthening pharmaceutical manufacturing capacity, regulatory credibility, innovation ecosystems and resilient medicine supply chains.
6.1.
Problem
Many African countries
remain highly dependent on imported medicines and active pharmaceutical
ingredients. This dependence exposes healthcare systems to supply disruptions,
currency shocks and geopolitical risks, undermining medicine security and
weakening pharmaceutical sovereignty.
Despite decades of discussion on pharmaceutical industrialisation in Africa, the role of pharmacy education and the pharmaceutical workforce in enabling pharmaceutical sovereignty has received limited conceptual attention.
6.2.
Core concept
This paper introduces
the Medicine Security Builders
Framework (Baale Model), which proposes transforming pharmacy education
and professional practice from a dispensing-centred
model to a systems leadership
model that supports pharmaceutical sovereignty.
Under this framework, pharmacists are repositioned as medicine security builders-professionals capable of strengthening pharmaceutical manufacturing, regulatory science, digital health systems and pharmaceutical policy.
6.3.
Four strategic pillars
The framework
identifies four mutually reinforcing pillars required to transform the pharmacy
profession.
6.4.
Education reform
Pharmacy curricula must
expand beyond dispensing to include industrial pharmacy, regulatory science, Pharmacoeconomics,
pharmaceutical policy, artificial intelligence and digital health systems.
6.5.
Industrial integration
Universities must
strengthen partnerships with pharmaceutical manufacturers, biotechnology firms
and innovation hubs to support research translation, technology transfer and
workforce development.
6.6.
Regulatory strengthening
Strong regulatory
systems aligned with global standards are essential for ensuring the quality of
medicines, attracting investment, and building confidence in locally
manufactured medicines.
6.7.
Entrepreneurial leadership
Pharmacists should be
encouraged to become pharmaceutical entrepreneurs, innovators and policy
leaders capable of driving value creation across pharmaceutical supply chains.
6.8.
Expected system outcomes
Implementation of the
Medicine Security Builders Framework contributes to:
· Reduced medicine import dependency
· Improved medicine affordability and access
· Resilient pharmaceutical supply chains
· Enhanced regional pharmaceutical integration through AfCFTA
6.9.
Strategic significance
The framework connects pharmacy workforce transformation with
pharmaceutical industrial policy, positioning the profession as a key
driver of health security and economic development in Africa.
By linking education reform, industrial development and regulatory strengthening, the Baale Model provides a conceptual roadmap for transforming pharmacy into a strategic pillar of medicine security and pharmaceutical sovereignty.
7.
Comparative Models of Pharmacy Practice
Table
1: Current Pharmacy Model vs Medicine Security Builder
Model.
|
Dimension |
Current
Pharmacy Model |
Medicine
Security Builder Model |
|
Primary orientation |
Dispensing and retail
supply |
Systems leadership
for medicine security |
|
Workforce identity |
Job-seeking,
résumé-oriented graduates |
Nation-building,
industry-shaping professionals |
|
Core practice setting |
Community and
hospital pharmacy |
Manufacturing,
regulation, research, digital health, policy and entrepreneurship |
|
Dominant competence
base |
Product knowledge,
dispensing, routine patient counselling |
Formulation science,
API strategy, regulatory science, Pharmacoeconomics, supply chain
intelligence and policy leadership |
|
Educational emphasis |
Licensure preparation
and clinical dispensing |
Industrial
competence, innovation, translational science and leadership for sovereignty |
|
Exposure during
training |
Hospitals, community
pharmacies, wholesalers |
Manufacturing plants,
regulatory agencies, research hubs, innovation ecosystems and industrial
clusters |
|
Regulatory outlook |
Compliance as
obligation |
Regulation as
market-enabling infrastructure and confidence builder |
|
Digital orientation |
Basic ICT support |
AI-enabled pharmacy,
digital pharmacovigilance, traceability and data-driven decision-making |
|
Research focus |
Academic output with
limited commercial translation |
Innovation, patents,
technology transfer and scalable product development |
|
Industry linkage |
Weak or episodic |
Structured
university–industry–government collaboration |
|
Economic contribution |
Medicine distribution
and service delivery |
Local value creation,
industrial expansion, export readiness and resilience |
|
Strategic metric of
success |
Employment rates,
licensure pass rates, dispensing volume |
Reduced import
dependency, stronger local manufacturing, improved affordability and better
health outcomes |
|
Continental role |
Nationally fragmented
practice |
Regional integration
through AfCFTA-aligned pharmaceutical ecosystems |
|
Long-term
contribution |
Professional service
continuity |
Pharmaceutical
sovereignty, medicine security and national resilience |
8.
Discussion
The analysis highlights
a structural paradox within the African pharmaceutical landscape. While demand
for medicines continues to grow due to population expansion and epidemiological
transitions, domestic pharmaceutical production capacity remains limited.
This imbalance reinforces reliance on imported medicines and undermines medicine security.
The transformation of pharmacy education, therefore, represents a strategic intervention capable of addressing this structural challenge.
Historically, the development of the pharmaceutical industry in Europe and North America was accompanied by the expansion of pharmaceutical sciences education and industrial pharmacy training.
Africa must pursue a similar trajectory.
However, strengthening pharmaceutical education alone is insufficient. Sustainable pharmaceutical development requires alignment among education, industry and government institutions, consistent with the Triple Helix model of innovation10.
Regulatory institutions also play a central enabling role. Strong regulatory systems increase confidence in locally manufactured medicines and encourage pharmaceutical investment.
As previously emphasised in work on pharmaceutical sovereignty5, the future of Africa’s pharmaceutical sector depends on building integrated pharmaceutical ecosystems that combine scientific capability, industrial production, and regulatory credibility.
The proposed Medicine Security Builders Framework provides a conceptual roadmap for achieving this transformation.
9.
Policy Recommendations for Governments and Universities
9.1.
For governments
· Public procurement policies should encourage quality-assured local manufacturing, while regulatory systems must be strengthened to meet the WHO maturity standards.
· Financial incentives, innovation funds and pharmaceutical industrial parks should be developed to support domestic pharmaceutical production.
· Regional cooperation through AfCFTA should be leveraged to harmonise regulatory standards and strengthen regional pharmaceutical markets.
9.2.
For universities
· Training should include placements in manufacturing plants, regulatory agencies and pharmaceutical innovation hubs.
· Universities should also establish stronger partnerships with pharmaceutical companies to facilitate research commercialisation and technology transfer.
· Entrepreneurship training should encourage pharmacists to create enterprises across the pharmaceutical value chain.
10.
Implications for Africa’s Pharmaceutical Industrialisation
Repositioning pharmacy
toward the Medicine Security Builder Model has profound implications for
Africa’s pharmaceutical industrialisation.
· Second, it strengthens the innovation ecosystem by linking pharmaceutical education more directly to product development and technology transfer.
· Third, it enhances confidence in locally manufactured medicines by strengthening regulatory and professional competencies.
· Finally, it supports regional competitiveness by producing professionals capable of operating within an integrated continental pharmaceutical market under AfCFTA.
The future of Africa’s pharmaceutical sector will therefore depend not only on factories and financing but also on the type of professionals the continent trains.
11.
Discussion
Recent global health
events have renewed attention on the strategic importance of local pharmaceutical manufacturing in Africa.
The COVID-19 pandemic and subsequent outbreaks such as mpox highlighted the vulnerability of import-dependent regions in accessing essential medicines and vaccines11. Strengthening local pharmaceutical production has therefore emerged as a key pillar of health security across the continent.
Recent policy reports emphasise that Africa must invest significantly in domestic pharmaceutical production to reduce reliance on imports and build resilient health systems. More than 70 % of medicines consumed in Africa are currently imported, while pharmaceutical production capacity remains concentrated in a small number of countries12.
Similarly, continental policy initiatives, such as the Pharmaceutical Manufacturing Plan for Africa (PMPA), aim to develop a competitive, sustainable pharmaceutical manufacturing industry capable of improving access to essential medicines while generating economic benefits13.
Recent global health analyses also highlight that weak pharmaceutical manufacturing capacity contributes directly to inequitable access to medicines across Sub-Saharan Africa. Structural barriers, such as limited technology transfer, restrictive intellectual property frameworks, and underinvestment in pharmaceutical infrastructure, continue to limit local production.
These developments reinforce the central argument of this paper: pharmaceutical sovereignty requires not only industrial infrastructure but also a workforce capable of sustaining pharmaceutical ecosystems.
The transformation of pharmacy education, therefore, represents a strategic intervention within broader pharmaceutical industrial policy.
12.
Conceptual Contribution
This paper contributes
to the emerging discourse on pharmaceutical sovereignty by proposing a new professional transformation framework for
pharmacy education and practice in Africa.
The existing literature on pharmaceutical development in Africa has largely focused on industrial policy, regulatory strengthening, and supply chain reforms12,14. While these factors remain critical, relatively little attention has been given to the human capital architecture required to sustain pharmaceutical sovereignty.
The principal conceptual contribution of this study is therefore the introduction of the Medicine Security Builders Framework (Baale Model).
The model advances
three important theoretical propositions.
· Second, pharmacy education must evolve from a distribution-centred professional paradigm to a systems leadership paradigm that integrates manufacturing science, regulatory strategy, digital health and industrial policy.
· Third, pharmacists can function as strategic actors within national pharmaceutical ecosystems, contributing not only to clinical care but also to pharmaceutical innovation, industrialisation and policy development.
By linking workforce transformation to pharmaceutical industrial policy, the Baale Model provides a conceptual bridge between pharmacy education, health system resilience and economic development.
This contribution complements broader continental initiatives such as the Pharmaceutical Manufacturing Plan for Africa (PMPA) and the Africa CDC initiatives on local health product manufacturing, which aim to strengthen regional pharmaceutical production ecosystems13,15.
13.
Conclusion
The four-million-year
evolution of pharmacy demonstrates a profession characterised by continuous
reinvention.
In the African context,
the next stage of this evolution must focus on medicine security and pharmaceutical sovereignty.
Repositioning pharmacists as medicine security builders will strengthen pharmaceutical manufacturing, regulatory systems, digital health integration and policy leadership.
Through this transformation, pharmacy can reclaim its historic mission—not only dispensing medicines but also building resilient pharmaceutical systems that protect health and support economic development across Africa.
14.
References
1.
Blomstedt P. Imhotep and the
discovery of cerebrospinal fluid. Acta Neurochirurgica Supplement, 2014.
2.
Metwaly AM.
Traditional ancient Egyptian medicine: A review. Journal of Ethnopharmacology,
2021.
3.
Baldrick T. The
surprisingly advanced medicine of ancient Egypt. The Collector, 2022.
4.
Baale
L. Africa’s path to pharmaceutical sovereignty and renaissance: Building
champions at home-Archives of Biotechnology and Pharmaceutical Research, 2025.
5.
Baale
L. The evolution of pharmacy: From Imhotep to the modern era. Archives of
Biotechnology and Pharmaceutical Research, 2025.
6.
Toklu HZ. The changing
face of pharmacy practice-Journal of Pharmacy and Pharmaceutical Sciences,
2013.
7.
Urick BY. Evolution of
pharmacy practice and education. Research in Social and Administrative
Pharmacy, 2019.
8.
Hippensteele A. AI in pharmacy:
Transforming medication management. Pharmacy Times, 2024.
9.
WHO. Global
Benchmarking Tool for Evaluation of National Regulatory Systems. Geneva: World
Health Organisation, 2023.
10.
Etzkowitz
H, Leydesdorff L. The dynamics of innovation: From national systems to Triple
Helix. Research Policy, 2000.
11.
Ndembi N, et al. Local
pharmaceutical manufacturing and health security in Africa. Global Health Policy Review, 2024.
12.
UNCTAD.
Building the Case for Investment in
Local Pharmaceutical Production in Africa. Geneva: United Nations
Conference on Trade and Development, 2025.
13.
Africa
CDC. Africa’s Progress Towards
Sustainable Local Manufacturing of Health Products. Addis Ababa: Africa
Centres for Disease Control and Prevention, 2025.
14.
WHO.
Framework for Strengthening Local
Production of Medicines, Vaccines and Health Technologies in the African Region
2025-2035. Geneva: World Health Organisation, 2024. 15.
African Union.
African Continental Free Trade Area: Operational Framework and Pharmaceutical
Opportunities. Addis Ababa: African Union Commission, 2022.