Hepatoprotective Effects of Ethanol Extract of Acetate Induced Oxidative Damage in Albino Rats
Abstract
This study evaluated the hepatoprotective effects of ethanol extract of
Platycerum bifurcatum in lead acetate induced oxidative damage. Thirty male
albino rats of mean weight 120 g were divided into 5 groups of six rats each.
Groups 1 - 3 served as normal control, lead acetate control (100 mg/kg body
weight), extract group only (400 mg/kg body weight), and while groups 4 and 5
were lead acetate induced groups treated with 200 and 400 mg/kg body weight of
ethanol extract of Platycerum bifurcatum respectively. Treatment lasted for 28
days, after which the animals were sacrificed under mild ether anaesthesia.
Blood samples were collected for biochemical analysis. The result from the
study showed that there was statistically significant (p< 0.05) decrease in
the concentration of ALT, AST, ALP, total bilirubin and albumin in the treated
groups, when compared with the lead acetate untreated group. Also, there was
statistically significant (p< 0.05) increase in the concentration of total
protein in the treated groups, when compared with the lead acetate untreated
group. These findings indicate that ethanol extract of Platycerum bifurcatum
possesses hepatoprotective effects in lead acetate induced oxidative damage in
albino rats, and thus could be utilized pharmacologically in the management and
treatment of oxidative damage and organ toxicity.
Keywords: Hepatoprotective, Platycerum bifurcatum, Lead acetate, Oxidative damage, Alanine aminotransferase, Aspartate transaminase.
1.
Introduction
Lead (Pb), a toxic heavy metal, poses a
significant threat to human health on a global scale1. This insidious substance exists in
various forms and sources, including food and air pollution2. It exacts a toll on multiple vital
organs, ranging from the brain and kidneys to hematopoietic tissues3. As lead-contaminated food and water are
absorbed through the duodenum, more than 95% of lead binds to erythrocyte
proteins and is stored in internal organs, particularly the liver4. One of the key mechanisms implicated in
lead-induced toxicity is oxidative stress, an imbalance between oxidant and
antioxidant systems due to the excess production of reactive oxygen species
(ROS). This unchecked generation of ROS by lead exposure leads to mitochondrial
impairment and cell damage5. In
response to lead toxicity, various heavy metal chelating agents have been
employed as therapeutic drugs, but they come with side effects6.
In this context, the study explores the
potential of Platycerium bifurcatum, commonly known as the “staghorn
fern,” to mitigate liver function impairment caused by lead toxicity. This
unique plant, naturally found in the canopies of tropical and subtropical
forests, has gained recognition for its medicinal properties and antioxidant
potential7,8. It is traditionally
employed in Nigeria for various therapeutic purposes, such as addressing ulcers
and preventing miscarriages9,10.
The antibacterial properties of this fern have been well-documented, with
activity against clinical strains of Escherichia
coli, Staphylococcus aureus, and Klebsiella spp. Furthermore, Platycerium
bifurcatum's antioxidative potential, linked to its rich phenolic content,
makes it a promising candidate for addressing lead-induced oxidative stress11. Notably, the chloroform fraction of this
plant has demonstrated antioxidant properties comparable to ascorbate12.
This study embarks on an exploration of
the potential of Platycerium bifurcatum
in alleviating liver function impairment induced by lead toxicity in Wistar
albino rats. By examining the potential of this plant to safeguard the liver in
concomitant administration of lead, this research aspires to deepen our
comprehension of lead-induced health challenges and to unravel the promising
avenues offered by natural remedies, with a particular emphasis on Platycerium
bifurcatum.
2.
Materials and methods
2.1. Collection and identification of specimen
Fresh frond (leave) and stalk of Platycerium bifurcatum
were collected in the mangrove vicinity of university of Lagos and was
identified and authenticated by a Taxonomist, Dr. Akinnibosun Henry Adewale in
the department of Plant Biology and Biotechnology, University of Benin, and
given a voucher number UBH-A650.
2.2. Preparation of sample and extraction
Fresh frond (leaves) of Platycerium
bifurcatum were rinsed in running
tap water to remove debris and then air dried under shade for two weeks. The
leaves were pulverized using a mechanical blender to coarse powder. Three
hundred (300) g of pulverized sample was macerated in 2100 ml of ethanol and
shaken severally. After 72 hours, total extract obtained were filtered using a
muslin cloth and subsequently with Whattman filter paper No. 1 (125 mm).
Filtrate was obtained after concentration using a rotary evaporator at 45°C to
obtain the crude extract.
2.3. Experimental design
Thirty (30) wistar albino rats of mean weight 120 g were purchased from
the animal facility Centre of the Faculty of Pharmaceutical Science, University
of Nigeria Nsukka. The animals were allowed to acclimatize for two weeks prior
to start of experiment, at the animal facility Centre of the Department of
Biochemistry, Michael Okpara University of Agriculture, Umudike, with access to
standard rodent feed and water ad libitum.
The animals were fasted overnight and randomly distributed into five
groups of six rats each. Among the five
(5) groups of animals, Group I received only food and water serving as normal
control, while group 2 received lead acetate only (100 mg/kg body weight),
group 3 received extract only (400 mg/kg body weight), Group 4 received lead
acetate and extract (200 mg/kg), and Group 5 received lead acetate and extract
(400 mg/kg body weight). Treatment
lasted for 28 days after the animals were sacrificed under mild ether
anaesthesia, blood samples were collected for biochemical analysis.
3. Evaluation of In Vitro Antioxidant Activity
2,2-diphenyl-1-picrylhydrazine (DPPH) scavenging activity was carried
out as described by13. FRAP assay
was carried out following the method described by14,15.
3.1. Evaluation of in vivo antioxidant
activity
The activity of catalase was assayed by the
method of 16. Superoxide dismutase activity was assayed by
the method of17 as contained in
Randox kit. Estimation of glutathione peroxidase was done according to the method of18. Estimation
of reduced glutathione was determined
by the method of19. Estimation
of malondialdehyde (MDA) concentration was estimated by measuring
spectrophotometrically the level of the lipid peroxidation product,
malondialdehyde (MDA) as described by20.
4. Statistical Analysis
All data were treated statistically using Statistical Package for Social
Science (SPSS) (Version 20). The data are expressed as mean ± standard
deviation using bar charts. Comparisons were made between the control and test
groups using the one-way analysis of variance (ANOVA) and multiple comparisons
(Tukey) at p≤0.05 level of significance.
5. Results
5.1.
Result of liver function assay
There was a
significant (P<0.05)
decrease in ALT for all extract treated groups
when compared to the lead acetate control group (Figure 1)
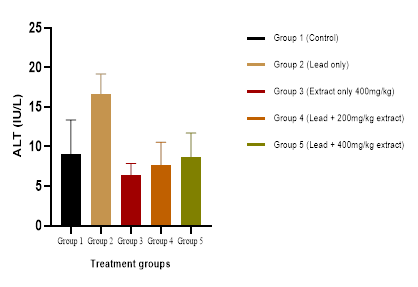
Figure 1:
Effect of administration of crude extract of Platycerium bifurcatum on ALT activity.
There was a significant (P<0.05) decrease in AST for all extract treated groups when compared to the lead acetate control group (Figure 2).
Figure 2: Effect of administration of crude extract of Platycerium bifurcatum on AST activity.
Figure 3: Effect of administration of crude extract of Platycerium bifurcatum on ALP activity.
There was a significant (P<0.05) decrease in total bilirubin (TB) for all extract treated groups when compared to the lead acetate control group (Figures 4-6).
Figure 4: Effect of administration of crude extract of Platycerium bifurcatum on Total
bilirubin concentration.
Figure 5:
Effect of administration of crude extract of Platycerium bifurcatum on Albumin concentration.
Figure 6:
Effect of administration of crude extract of Platycerium bifurcatum on Total protein concentration.
5.2.
Histopathological examination of the liver
The histopathological examination of the
liver showed variable necrosis for all groups except group 5 which showed the
normal liver histomorphology. The lead only treated group (Figure 8)
showed multifocal hepatocellular necrosis. There is also mild infiltration of
anti-inflammatory leukocytes in group 1, 2, 3 and 5 (Figures 7-9, 11).
Figure 7:
Histopathology of liver for group 1.
The sections of the liver presented in
group 1 showed piecemeal necrosis with moderate periportal infiltration of
inflammatory leukocytes (arrow). Central vein (V), Portal triad (P).
Figure 8:
Histopathology of liver for group 2.
The sections of the liver presented in
group 2 showed multifocal areas of hepatocellular necrosis with mild to
moderate infiltration of inflammatory leukocytes (arrow). Portal triad (P).
Figure 9:
Histopathology of liver for group 3.
The sections of the liver presented in
group 3 showed mild, random, areas of hepatocellular necrosis (red arrow).
Also, periportal piecemeal necrosis with mild infiltration of inflammatory
leukocytes were observed (black arrow). Central vein (V), Portal triad (P).
Figure 10:
Histopathology of liver for group 4.
The sections of the liver presented in
group 4 showed the normal hepatic histomorphology. Central vein (V), portal
triad (P).
Figure 11:
Histopathology of liver for group 5.
The sections of the liver presented in
group 5 showed mild, periportal piecemeal necrosis with mild infiltration of
inflammatory leukocytes were observed (black arrow). Portal triad (P).
6. Discussion
Lead is a ubiquitously found environmental
and industrial pollutant that has been detected in nearly all phases of
environment and biological system. Its persistence in human and animal tissues
has quite often been associated with considerable health risks21. In the present study, the impact of lead
in tissues of liver were significantly higher in lead acetate treated group
than controls and groups administered with extracts. Also, multifocal necrosis
of the hepatocytes was observed in lead treated group (Figure 8). In
addition to necrosis, inflammatory leukocytes infiltration was noticed in some
other cases. Ingestion of lead is one of the primary causes of hepatotoxic
effects. The necrosis observed in the control group may be due to other factors
that may not have been easily identified or controlled. The molecular
understanding of lead effects on hepatic drug metabolizing enzymes, cholesterol
metabolism, oxidative stress, and hepatic hyperplasia suggest a potential role
for lead in damaging extrahepatic systems, including the cardiovascular system.
Groups treated with lead acetate and low dose of crude extract of Platycerium
bifurcatum extract showed normal histopathology of the liver when compared
to the control and lead acetate treated groups, representing a good sign of
regeneration22. Also, similar
results were reported by23 who
demonstrated that treatment with Quercetin, the major flavonoid component of Platycerium
bifurcatum24,
by oral administration significantly protects the liver after alcohol induced
liver injury, possibly through its antioxidant, anti-inflammation and
anti-apoptosis effects by STAT3, Akt and NF-κB pathway.25 found a significant higher levels of lead
in liver of rats exposed to Pb for 4 weeks and a significant reduction of lead
levels after treatment with chelating agent, EDTA, during 5th week26-28 reported that Chlorogenic acid, a
natural phenolic product and major constituent of Platycerium bifurcatum
plays an increasingly positive role in removing the toxicity of heavy metals
owing to its antioxidative activity and metal-chelating properties.
We suggest one possibility that Platycerium bifurcatum complexes with
lead ion decreasing its lipophilicity, and thus its gastrointestinal
absorption. The chelating agents form an insoluble complex with Pb to remove it
from Pb-burdened tissues29. This
means that, the more lead is chelated from circulating freely in the biological
system, the less impact it will have on organs30
found sodium molybdate supplementation provide significant protection from Pb
uptake by blood, liver and kidney. The liver enzyme assays indicated that lead
acetate ingestion induced a significant elevation of plasma ALT, AST and ALP
levels at four weeks of lead acetate treatment. Since aminotransferases (ALT
and AST) are an important class of enzymes linking carbohydrate and amino acid
metabolism, the relationship between the intermediates of the citric acid cycle
is well established. These enzymes are regarded as markers of liver injury. The
observed result showed that the administration of Platycerium bifurcatum
extract doused the influence lead had on ALT and AST, however statistical
evaluations of the result showed non-significance. This may be as a result of
the dose of extract used, thus, there may be a more significant influence of Platycerium
bifurcatum extract on ALT
and AST levels altered by lead at higher doses.
In addition, ALP is membrane bound and its
alteration is likely to affect the membrane permeability and produce
derangement in the transport of metabolites. Moreover, elevated ALP activity,
which was used as a marker of liver adaptation to damaging factors, has been
reported frequently in lead exposed animals31,32.
The findings from this study showed a significant decrease in ALP levels
indicating ameliorative effect of the extract on the impact of lead when administered.
It is well known that lead binds to plasmatic proteins, where it causes
alterations in a high number of enzymes. It can also perturb protein synthesis
in hepatocytes33. It was observed
that the administration of lead and co-administration of lead and extract did
not show any significant alteration of the synthetic capacity of the liver.
Again, this may be as a result of the doses administered for both the toxicant
and treatment intervention. However, there was a significant reduction in
bilirubin level (Figure 4). Overall, the study showed that Platycerium bifurcatum offer mild
protective ability to the liver in the case of lead toxicity.
7.
Conclusion
This study indicates that Platycerum
bifurcatum mitigates liver function impairment induced by lead toxicity in
wistar albino rats. Lead being a heavy metal can accommodate in the biological
system through exposure to environment and it has an impact on the liver which
is a vital organ responsible for various metabolic processes. The result of
this study indicates that the phytochemicals present in the plant could be a
potential source of therapeutic agent in management of lead induced liver
toxicity.
The
authors hereby declare that “Principles of laboratory animal care” (NIH
Publication No. 85- 23, revised 1985) were followed in this study, as well as
specific national laws, where applicable. All experiments were examined and
approved by the appropriate ethical committee.
9. Acknowledgement
We sincerely appreciate the Department
of Biochemistry, College of Natural Sciences, Michael Okpara University of
Agriculture Umudike, Abia State, Nigeria for providing the facility for this
study.
10. Conflicts
of Interest
The authors declare that they have no
conflict of interest.
8. Correia
MA. Drug biotransformation. Basic
& Clinical Pharmacology, 2018;53.
16.
Sinha KA.
Colorimetric Assay of Catalase. Analytical
Biochemistry, 1972;47: 389-394.
19. Hoekstra
LT, de-Graaf W, Nibourg GA, et al. Physiological and biochemical basis of
clinical liver function tests: a review. Annals
of Surgery, 2013;257(1): 27-36.
23. Jones
MW, Small K, Kashyap S, et al. Physiology, gallbladder, 2018.
24. Kalra
A, Yetiskul E, Wehrle CJ, et al. Physiology,
Liver, 2018.
25.
Kosnett
MJ. Lead. In Brent, J. (Ed.), Critical
Care Toxicology: Diagnosis and Management of the Critically Poisoned Patient,
Gulf Professional Publishing, 2005.
26.
Kumari
K, Pahuja SK, Kumar S. An Evolution of Bilirubin Physiology and Analysis. Current Signal Transduction Therapy, 2023;18(2):
1-13.
27.
Merill
JC, Morton JJP, Soileau SD. Metals. In Hayes, A.W. (Ed.), Principles and Methods of Toxicology (5th
ed.), CRC Press, 2017.
29.
Mycyk
M, Hryhorcu D, Amitai Y. Lead. In Erickson TB, Ahrens WR, Aks S, Ling L.
(Eds.), Paediatric Toxicology:
Diagnostic and Management of the Poisoned Child, McGraw Hill
Professional, 2005.
31. Needleman
H. Lead poisoning. Annu Rev Med, 2004;55:
209-222.