Innovative Nose-To-Brain Drug Delivery Strategies for Neuroprotection in Diabetes Associated Cerebral Ischemia: Advances, Challenges and Translational Perspectives
Abstract
Intranasal delivery has gained significant attention as a non-invasive and efficient route to target neuroprotective agents directly to the brain, especially in managing diabetes-associated cerebral ischemia. This delivery bypasses the blood-brain barrier, maximizing therapeutic concentrations in the central nervous system while minimizing systemic exposure and associated side effects. Studies on intranasal insulin and insulin-like growth factor-1 (IGF-1) have demonstrated neuroprotection via vasodilatory, anti-inflammatory, antithrombotic effects and improved neuronal metabolism, functional connectivity and neurotransmitter regulation. Intranasal administration of exendin-4, a glucagon-like peptide-1 receptor agonist used for diabetes, has also shown promising neuroprotective effects in animal models of ischemic stroke by reducing infarct volume and neurological deficits without significant peripheral effects. Moreover, evidence suggests that intranasal insulin ameliorates experimental diabetic neuropathy by directly targeting the nervous system, avoiding systemic insulin exposure that can lead to hypoglycaemia and restoring key signalling pathways in dorsal root ganglia. Overall, intranasal delivery represents a feasible, safe and effective approach for neuroprotective therapy in diabetic stroke and neuropathy, demonstrating compelling potential for clinical translation.
Keywords: Intranasal delivery, Nose-to-brain transport, Diabetic stroke, Neuroprotection, Blood-brain barrier, Nanocarriers, Cerebral ischemia
Abbreviations Section: BBB, CNS, MCAO, IGF-1, PLGA,
NLC, NPs
1.
Introduction
Diabetes mellitus is a chronic metabolic disorder
characterized by persistent hyperglycemia resulting from defects in insulin secretion,
insulin action or both1.
Globally, the prevalence of diabetes is rapidly increasing, with significant
implications for public health due to its association with cardiovascular and
cerebrovascular complications2.
One of the most severe neurological complications observed in diabetic patients
is cerebral ischemia, commonly manifesting as ischemic stroke, which leads to
substantial morbidity and mortality3.
Diabetic patients not only have a higher risk of stroke but also experience
more severe neurological deficits and poorer recovery outcomes compared to
non-diabetic individuals4,5. The
pathophysiology underlying diabetes-associated cerebral ischemia involves
complex mechanisms such as impaired cerebral blood flow autoregulation,
increased oxidative stress, neuroinflammation and enhanced neuronal apoptosis6,7.
Despite advances in acute ischemic stroke management,
including thrombolytic therapies and mechanical thrombectomy, therapeutic
options specifically targeting neuroprotection in diabetic stroke remain
limited8. The development of
effective neuroprotective agents is critical for limiting infarct size and
improving neurological function post-stroke; however, systemic administration
of these agents faces major obstacles, primarily the blood-brain barrier (BBB)9. The BBB is a highly selective
semipermeable barrier that restricts the entry of most therapeutic molecules
into the brain, thereby limiting drug bioavailability at the ischemic site10. Consequently, novel drug delivery
strategies that can effectively circumvent the BBB and enhance targeted brain
delivery are urgently needed.
Intranasal (IN) drug delivery has emerged as a
promising non-invasive approach for delivering neuroprotective agents directly
to the brain, bypassing the BBB through the olfactory and trigeminal neural
pathways11,12. The nasal route
offers multiple advantages such as rapid drug absorption, avoidance of
first-pass metabolism and the potential for self-administration, thereby
improving patient compliance13.
Intranasal delivery exploits the unique anatomical connection between the nasal
cavity and the central nervous system (CNS), enabling direct transport of therapeutics
along neuronal pathways14. This
route provides an efficient means to deliver a wide range of drugs, including
peptides, proteins and small molecules, which are otherwise challenging to
administer systemically15,16.
Significant progress has been made in understanding
the nasal anatomy, physiology and the underlying mechanisms governing
nose-to-brain transport17. Key
advancements include the development of innovative formulations such as
nanoparticles, mucoadhesive gels and in situ forming systems, which enhance
drug stability, mucosal retention and brain targeting efficacy18,19. Alongside these technological
breakthroughs, various neuroprotective agents-both synthetic like insulin and
natural compounds such as melatonin-have been investigated for intranasal
delivery with encouraging preclinical and early clinical results20,21. These agents demonstrate the ability
to modulate key pathological processes including oxidative stress, inflammation
and apoptosis in diabetes-associated cerebral ischemia22.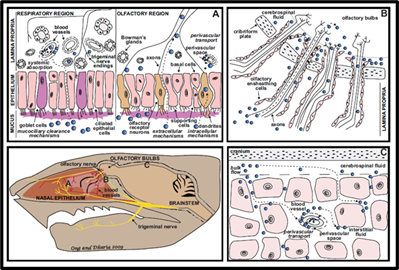
Figure 1: Mechanisms and
Pathways of Intranasal Drug Delivery to the Central Nervous System11.
(Figure 1) explain schematic
illustration of intranasal drug transport pathways to the central nervous
system. Following administration, drugs interact with the nasal epithelium and
underlying lamina propria and are transported via olfactory and trigeminal
neural pathways, perivascular channels and systemic circulation. In the
olfactory region, drugs access the cerebrospinal fluid and olfactory bulbs,
while distribution within the brain occurs through bulk CSF flow and
perivascular spaces. These routes also contribute to bidirectional solute
clearance between the CNS and the periphery.
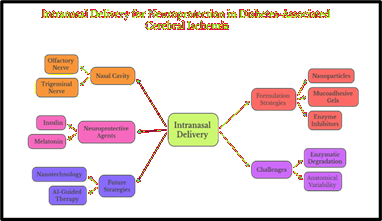
Figure 2: Intranasal Delivery Strategies for Neuroprotection in Diabetes-Associated Cerebral Ischemia.
(Figure
2)
demonstrates a conceptual overview of Intranasal Delivery as a promising route
for drug administration, particularly targeting the central nervous system. It
shows that drugs administered through the nasal cavity can reach the brain via
the olfactory and trigeminal nerve pathways, thereby bypassing the blood-brain
barrier. The diagram highlights neuroprotective agents such as insulin and
melatonin as therapeutic candidates delivered through this route. It also
outlines key formulation strategies-including nanoparticles, mucoadhesive gels
and enzyme inhibitors-designed to enhance drug absorption, stability and
residence time in the nasal mucosa.
However,
despite promising data from animal models and pilot clinical studies, several
translational challenges impede the widespread clinical adoption of IN
neuroprotective therapies23.
These include anatomical variability of the nasal cavity, mucociliary
clearance, enzymatic degradation of peptides and regulatory hurdles concerning
safety and efficacy evaluation24.
Additionally, there is a need for comprehensive clinical trials to establish
optimized dosing regimens and long-term safety profiles25.
In this
review, we provide an updated and comprehensive analysis of the physiology of
nose-to-brain drug delivery, current formulation strategies and the spectrum of
neuroprotective agents relevant to diabetes-associated cerebral ischemia and
evidence from preclinical and clinical studies. We also critically discuss
challenges and future perspectives focused on enhancing the translational
success of intranasal neuroprotective therapies. Collectively, this review aims
to highlight the promising role of intranasal delivery as a novel paradigm to
improve outcomes in patients suffering from diabetes-associated cerebral
ischemia and stroke.
2.
Anatomy and Physiology: Nose-to-Brain Transport
The nasal
cavity serves as a direct anatomical interface between the external environment
and the brain, allowing drug delivery strategies to bypass the restrictive
blood-brain barrier (BBB) via the nose-to-brain route26. The olfactory region, located in the
upper part of the nasal cavity, is characterized by olfactory sensory neurons
whose axons project directly into the olfactory bulb of the brain27. This unique neuroanatomical arrangement
permits intracellular axonal transport of molecules from the nasal mucosa to the
central nervous system (CNS)28.
Two major
pathways mediate nose-to-brain transport: the olfactory nerve pathway and the
trigeminal nerve pathway. The olfactory pathway transmits substances via
neuronal uptake and axonal transport across the cribriform plate to the
olfactory bulb29. The trigeminal
nerve pathway innervates the respiratory region of the nasal cavity and
provides an additional conduit for drug delivery toward the brainstem and
spinal cord30. Both pathways
support extracellular diffusion via perineural channels and endocytosis,
facilitating rapid drug movement into the CNS while minimizing systemic
exposure31.
The nasal
mucosa itself consists of pseudostratified ciliated columnar epithelial cells,
basal cells, mucus-secreting goblet cells and an underlying lamina propria with
a network of blood vessels and nerves32.
The epithelium forms tight junctions that regulate permeability, while the
mucus layer and mucociliary clearance mechanisms serve as physiological
barriers, influencing drug residence time and bioavailability33. These barriers pose challenges for
sustained drug absorption but are mitigated through formulation strategies like
mucoadhesive polymers and Nano-carriers34.
Enzymatic
degradation in the nasal cavity, owing to peptidases and proteases present in
the mucosa, further limits the stability of peptide and protein therapeutics
delivered intranasal35. However,
strategic use of enzyme inhibitors, protective carriers and permeation
enhancers can improve therapeutic efficacy without compromising safety36. Importantly, anatomical variations among
individuals in nasal cavity size, mucosal thickness and olfactory epithelium
extent impact the efficiency of nose-to-brain delivery and need consideration
in formulation design37.
Understanding
the complex interplay of nasal anatomy, physiology and transport mechanisms
lays the foundation for developing successful intranasal neurotherapeutics.
This knowledge is vital for tailoring formulations that optimize brain
targeting while minimizing systemic exposure and adverse effects, especially in
the context of neuroprotection for diabetes-associated cerebral ischemia38.
3.
Formulation Strategies for Intranasal Delivery
Intranasal
drug delivery has emerged as a promising non-invasive route to transport
therapeutic agents directly to the central nervous system (CNS) by bypassing
the blood-brain barrier (BBB). However, the nasal cavity's unique anatomy and
physiology present formulation challenges that must be addressed to achieve
optimal drug absorption, stability and brain targeting26. The development of advanced formulation
strategies such as nanoparticles, mucoadhesive systems, penetration enhancers,
enzyme inhibitors, gels and powders have been explored extensively to enhance
the efficiency and efficacy of intranasal delivery27,28.
3.1. Nanoparticles
and nanocarriers
Nanoparticles
(NPs), including polymeric nanoparticles, liposomes, solid lipid nanoparticles
(SLNs) and nanoemulsions, are among the most versatile carriers for intranasal
drug delivery29,30. Their
nanoscale size facilitates mucosal permeation and uptake by olfactory and
trigeminal nerve pathways. Polymeric NPs such as those based on poly (lactic-co-glycolic
acid) (PLGA) offer controlled drug release, improved stability and protection
from enzymatic degradation present in the nasal mucosa31. Lipid-based systems like SLNs and
nanostructured lipid carriers (NLCs) improve biocompatibility and drug
encapsulation efficiency, enhancing brain bioavailability32. Surface modifications, including PEGylation
or ligand conjugation, promote mucoadhesion, reduce clearance and improve
selective targeting of neuronal cells33.
3.2. Mucoadhesive
systems
Mucoadhesive
polymers such as chitosan, carbopol and hydroxypropyl methylcellulose (HPMC)
increase drug residence time in the nasal cavity by adhering to the mucosal
surface, thus counteracting rapid mucociliary clearance34. These polymers also transiently open
tight junctions in the nasal epithelium, enhancing paracellular drug transport.
Mucoadhesive nanogels and in situ gelling systems have gained attention for
their ability to form viscous gels upon contact with nasal fluids, thereby
sustaining drug release and improving patient compliance35. For example, in situ gels formed by
thermo-responsive polymers such as poloxamers transition from sol to gel at
nasal physiological temperature, prolonging drug retention36.
3.3. Penetration
enhancers and enzyme inhibitors
Nasal
formulations often incorporate penetration enhancers (such as bile salts,
surfactants and cyclodextrins) to increase epithelial permeability and
facilitate drug absorption across nasal mucosa37.
However, the safety of such enhancers needs thorough evaluation to prevent
mucosal irritation or toxicity. Enzymatic degradation of peptides and proteins
by nasal proteases limits drug bioavailability; hence, enzyme inhibitors like
aprotinin or bacitracin are co-formulated to protect labile drugs38. Combining penetration enhancers with
enzyme inhibitors maximizes drug stability and uptake in the nasal cavity.
3.4. Nasal
powders and microspheres
Nasal
powders present an alternative to liquid formulations that can enhance
stability, shelf life and dosing accuracy39.
Powders avoid issues related to solution spray deposition, dripping and short
residence time. Microsphere systems composed of biodegradable polymers such as
PLGA enable controlled drug release and protect drugs from enzymatic
degradation40. These solid
formulations can be formulated with mucoadhesive properties to enhance
retention and absorption.
3.5. Physicochemical
properties optimization
The
physicochemical characteristics of nasal formulations profoundly influence
their delivery success. Particle size is critical, as particles between 10-200
nm show optimal deposition in the olfactory region and enhanced transport via
neuronal pathways41. Surface
charge affects mucoadhesion-cationic particles tend to adhere better to the
negatively charged mucin layer, improving residence time42. Viscosity and pH of the formulation are
optimized to enhance comfort, stability and absorption; nasal formulations
typically have a pH range of 4.5-6.5 and viscosity moderations to avoid irritation
while maintaining adhesion43.
3.6. Device
and delivery technology
The
efficacy of intranasal formulations is also highly dependent on the delivery
device used. Advances in nasal spray pumps, nebulizers and breath-powered
devices improve dosing accuracy and delivery to the olfactory region44. Devices utilizing propellant-based or
bidirectional breath-powered technologies achieve deeper nasal penetration and
reduce drug loss due to anterior nasal deposition45.
Exhalation-assisted devices that seal the soft palate prevent pulmonary
exposure and improve CNS targeting.
3.7. Combination
and hybrid systems
To
overcome individual limitations, hybrid formulations combining nanoparticles in
mucoadhesive gels or powders loaded with enzyme inhibitors are being developed46. These combinations improve
bioavailability, stability and targeted delivery synergistically. Nanocarriers
decorated with targeting ligands or antibodies can be incorporated into gels to
provide both specific binding to brain receptors and prolonged nasal retention47.
3.8. Preclinical
success and considerations
Numerous
preclinical studies demonstrate that such optimized formulations significantly
increase brain concentrations of therapeutic agents, improve pharmacodynamics
and reduce systemic side effects48.
For example, PLGA nanoparticles loaded with neuroprotective peptides have shown
enhanced uptake in animal models of cerebral ischemia49. Lipid nanocarriers delivering melatonin
have decreased oxidative damage in diabetic ischemic rat models50. (Table 1) summarizes key
preclinical and clinical studies investigating intranasal neuroprotective
therapies-including melatonin nanocapsules, Exendin-4 and insulin-that
demonstrate reduced oxidative stress and infarct size, anti-inflammatory
effects and improved neurological and cognitive outcomes in diabetes-associated
cerebral ischemia.
Table
1:
Preclinical and Clinical Evidence of Intranasal Neuroprotective Therapies in Diabetes-Associated
Cerebral Ischemia.
|
Model/System |
Drug/Formulation |
Key
Results/Findings |
Reference |
|
Diabetic ischemic rats |
Melatonin nanocapsules |
Reduced oxidative stress and infarct size |
51 |
|
Rat focal cerebral ischemia |
Intranasal Exendin-4 |
Anti-inflammatory and neuroprotective
effects |
52 |
|
Diabetic MCAO rat model |
Intranasal Insulin |
Improved neurological outcomes, reduced
infarct volume |
53 |
|
Clinical ischemic stroke |
Intranasal insulin therapy |
Enhanced cognitive function and recovery
post-stroke |
54 |
4.
Drug Characteristics and Mechanisms of Nasal-to-Brain Delivery
4.1.
Types of drugs suitable for nasal-to-brain delivery
Intranasal
administration allows a diverse range of drugs to access the brain,
particularly those that face limitations with conventional systemic delivery
due to the blood-brain barrier (BBB). Peptides and proteins represent a major
class benefiting from intranasal delivery, as this route bypasses enzymatic
degradation in the gastrointestinal tract and first-pass metabolism in the
liver55,56. Insulin, insulin-like
growth factor-1 (IGF-1) and exendin-4 are widely studied for neuroprotection
owing to their roles in promoting neuronal survival and metabolic support57,58. Small molecule neuroprotective drugs,
including antioxidants like melatonin and calcium channel blockers, have
favorable molecular weight and lipophilicity that enable effective transport through
nasal mucosa59,60. Natural
compounds such as flavonoids and curcumin also exhibit neuroprotective
properties but often require nanocarrier formulations to enhance bioavailability
and stability61. The delivery of
large biomolecules such as monoclonal antibodies and nucleic acids represents
an emerging area with significant challenges due to size and enzymatic
degradation, which are often addressed by encapsulation in nanoparticles or
surface modification to enhance nasal uptake62.
4.2.
Physicochemical characteristics influencing nasal-to-brain transport
The
efficacy of nasal-to-brain delivery critically depends on drug physicochemical
properties. Molecular weight is a fundamental determinant; drugs below
approximately 1000 Da have higher permeability through nasal epithelium, while
larger molecules require specialized carriers63.
Lipophilicity significantly impacts mucosal membrane permeation, with
moderately lipophilic molecules demonstrating improved absorption64. Surface charge affects interaction with
the negatively charged mucin layer; cationic molecules or particles enhance
mucoadhesion and retention time thus improving absorption65. The pH and tonicity of nasal
formulations must align with physiological conditions (pH 4.5-6.5 and
isotonicity) to prevent mucosal irritation and ensure drug stability66.
4.3.
Mechanisms of drug uptake and transport via nasal mucosa
Drugs can
cross the nasal epithelium via paracellular (between cells) and transcellular
(through cells) pathways [67]. Small hydrophilic molecules Favor paracellular
transport via tight junctions, whereas lipophilic drugs utilize transcellular
diffusion. Endocytosis and receptor-mediated transport also play critical roles
for larger biomolecules and nanoparticles, facilitating their uptake and
transport along olfactory and trigeminal neural pathways68. The olfactory nerve pathway provides
direct access from nasal mucosa to olfactory bulb, while the trigeminal nerve
pathway targets brainstem and other deeper brain regions, enabling drug
trafficking into CNS tissue69.
4.4.
Formulation features enhancing drug delivery efficiency
Nanoparticles
protect drugs from enzymatic degradation and improve mucosal permeation.
Polymeric and lipid-based nanoparticles enable controlled drug release and
enhance brain targeting70.
Mucoadhesive polymers such as chitosan increase formulation residence time on
nasal mucosa by binding to mucin, thereby reducing clearance and enhancing
absorption71. Penetration
enhancers and enzyme inhibitors are incorporated to improve epithelial
permeability and prevent proteolytic degradation of labile drugs72. Advanced controlled-release systems and
stimuli-responsive formulations allow for dose optimization and patient-friendly
administration73.
4.5.
Barriers to effective nasal-to-brain drug delivery
Mucociliary
clearance rapidly removes formulations from the nasal cavity, limiting
residence time and absorption opportunities74.
Enzymatic activity within the nasal environment degrades peptides and proteins,
posing stability challenges75.
Interindividual anatomical variability, pathological changes in nasal mucosa
and limited dosing volume constrain delivery efficiency76. Chronic administration risks include
mucosal irritation and toxicity, necessitating safety evaluation77.
4.6.
Pharmacokinetics and biodistribution of intranasal drugs
Intranasal
administration exhibits rapid absorption kinetics, favouring swift CNS drug
uptake and onset of therapeutic effect78.
Imaging studies reveal distinct distribution patterns in brain regions
following nasal delivery, highlighting preferential access via olfactory bulb
and related structures79.
Systemic exposure is typically minimized, mitigating peripheral side effects
while maintaining effective CNS concentrations80.
5.
Neuroprotective Agents: Scope and Evidence
Effective
neuroprotection in diabetes-associated cerebral ischemia involves targeting
multiple pathological processes such as oxidative stress, inflammation,
excitotoxicity and apoptosis, which exacerbate neuronal injury following
ischemic insult81. Several
classes of neuroprotective agents have been investigated for their potential
efficacy, focusing on synthetic drugs, peptides and naturally derived compounds
amenable to intranasal delivery systems82,83.
5.1. Synthetic
drugs and peptides
Among
synthetic neuroprotective agents, insulin has attracted substantial attention
due to its metabolic and neurotrophic effects in the brain. Intranasal insulin
enhances cerebral glucose metabolism, suppresses apoptotic pathways and reduces
infarct volume in diabetic stroke models84,85.
Insulin-like growth factor-1 (IGF-1) and other peptides such as exendin-4, a
glucagon-like peptide-1 analog, have shown similar neuroprotective effects via
intranasal administration, promoting neuronal survival and functional recovery86,87. Other pharmacological agents,
including calcium channel blockers and antioxidants, have been formulated for
nasal delivery to target ischemic cascades88.
5.2. Natural
products and phytochemicals
Natural
compounds possess inherent antioxidative and anti-inflammatory properties,
making them attractive candidates for neuroprotection. Melatonin, a potent
endogenous antioxidant, has demonstrated efficacy in reducing oxidative damage
and apoptosis when delivered intranasally in lipid nanocarriers in diabetic
cerebral ischemia models89,90.
Flavonoids and curcumin are also explored for their ability to modulate signalling
pathways involved in ischemic injury and metabolic dysregulation91,92. Formulating these compounds into
nano-sized delivery systems enhances their solubility, brain penetration and
bioavailability93.
5.3. Mechanistic
insights and pathways
Neuroprotective
agents exert their effects through diverse mechanisms, including scavenging
reactive oxygen species, inhibiting pro-inflammatory cytokines, regulating
calcium homeostasis and activating cell survival pathways such as PI3K/Akt and
Nrf2/ARE94,95. Modulation of
mitochondrial function and synaptic plasticity further supports neuronal
resilience in the face of ischemic stress96.
Intranasal delivery facilitates rapid CNS uptake, enhancing therapeutic onset
and targeting efficacy due to bypassing systemic metabolism and BBB
restrictions97.
5.4. Preclinical
and clinical evidence
Preclinical
investigations in diabetic rodent models consistently reveal that intranasal
neuroprotective agents significantly attenuate ischemic injury, improve motor
and cognitive functions and modulate biochemical markers of oxidative damage
and inflammation98,99. Notably,
studies using intranasal insulin and melatonin formulations report reduced
infarct sizes and improved neurological scores100,101.
5.5. Clinical
translation is currently emerging
With
ongoing trials exploring the safety and efficacy of intranasal insulin and
peptide therapies in stroke patients with diabetes or insulin resistance102. Preliminary results demonstrate favourable
tolerability and cognitive benefits; however, extensive trials are required to
validate therapeutic efficacy and optimize dosage regimens103.
5.6. Challenges
and future directions
While
preclinical data are promising, challenges such as variable nasal absorption,
enzymatic degradation and patient compliance remain. Advances in formulation
technology and better understanding of pharmacokinetics will facilitate
overcoming these hurdles78.
Additionally, personalized approaches considering patient-specific
physiological and pathological factors will enhance neuroprotective treatment efficacy
in clinical settings104.
6.
Clinical and Preclinical Evidence
Preclinical
studies have demonstrated significant neuroprotective effects of intranasally
delivered therapeutic agents in experimental models of diabetes-associated
cerebral ischemia, providing a strong rationale for clinical translation105. Animal models commonly involve diabetic
rodents subjected to middle cerebral artery occlusion (MCAO) to simulate
ischemic stroke, allowing assessment of pharmacological interventions106. Several studies report that intranasal
administration of neuroprotective peptides, insulin and antioxidants
substantially reduce infarct volume, oxidative stress, inflammation and
neuronal apoptosis, resulting in improved neurological function and behavioural
outcomes107,108.
For
instance, intranasal insulin therapy has been shown to improve post-stroke
neurocognitive decline and promotes synaptic plasticity in diabetic rats,
attributed to enhanced glucose metabolism and antiapoptotic effects108,109. Melatonin-loaded lipidic
nanocapsules administered intranasally have demonstrated robust antioxidant
effects and attenuation of ischemic injury in diabetic ischemic rat models110. Peptide drugs such as exendin-4 also
exhibit anti-inflammatory and neurotrophic properties when delivered via the
nasal route111.
Clinical
evidence remains limited but promising. Early phase clinical trials evaluating
intranasal insulin in ischemic stroke patients with and without diabetes
indicate good safety profiles, enhanced cognitive outcomes and functional
recovery112,113. However, large
randomized controlled trials specifically targeting the diabetic stroke
population are lacking, with ongoing studies aiming to address this gap110. Variability in dosing regimens, patient
heterogeneity and challenges in measuring CNS drug bioavailability hamper
conclusive results115.
The use
of nasal delivery devices optimized for targeting olfactory regions and
patient-friendly administration has been shown to improve drug deposition and
therapeutic efficacy in clinical settings116.
Nonetheless, mucosal irritation, enzymatic degradation and interindividual
anatomical differences affect drug absorption and distribution, emphasizing the
need for personalized approaches117.
Pharmacokinetic
studies reveal rapid CNS penetration and prolonged residence time of
neuroprotective agents delivered intranasally compared to systemic routes,
favoring therapeutic effectiveness and minimizing peripheral side effects118,119. Integration of imaging techniques
and biomarker analysis in clinical trials will enhance understanding of
treatment mechanisms and foster optimized therapy development119.
Overall,
clinical and preclinical data underscore the considerable potential of
intranasal therapies to revolutionize neuroprotective strategies in
diabetes-associated cerebral ischemia. Accelerated translational efforts with
rigorous clinical evaluation are essential to bring these promising
interventions to routine clinical practice.
7.
Challenges in Translation to Clinic
Despite
the promising preclinical and early clinical data supporting intranasal
delivery of neuroprotective agents for diabetes-associated cerebral ischemia,
several challenges hinder widespread clinical implementation and regulatory
approval107. The anatomical and
physiological variability of the nasal cavity among individuals significantly
influences drug deposition, absorption efficiency and therapeutic outcomes.
Differences in nasal mucosa thickness, mucociliary clearance rates and the
relative size of the olfactory region led to inconsistent drug delivery to the
brain across patients85.
Furthermore, pathological conditions common in diabetic patients, such as
rhinitis or nasal congestion, further impair drug absorption and pose adherence
issues109.
Formulation-related
challenges include instability of peptide and protein drugs in the
enzymatically active nasal environment. Proteases and peptidases degrade
therapeutic biomolecules, necessitating the incorporation of enzyme inhibitors
or protective nanocarriers, which may complicate formulation safety and
regulatory acceptance110. The
limited volume that can be administered intranasally restricts dosage,
demanding highly potent and concentrated formulations capable of achieving
therapeutic effects with minimal administration volumes111.
Device
design critically impacts the efficiency and precision of nasal drug delivery.
While several advanced delivery devices exist-such as breath-powered nebulizers
and propellant-based sprays-standardization is lacking and devices may vary
substantially in drug deposition patterns and user convenience112. Repeated dosing required for chronic
conditions risks mucosal irritation and local toxicity, which necessitates
long-term safety studies113.
Regulatory
frameworks pose additional obstacles. The unique delivery route and composition
of intranasal neurotherapeutics pose challenges for establishing appropriate
safety and efficacy endpoints during drug development114. Differences in guidelines across
regulatory agencies and the paucity of clear precedents for approval of complex
nanoparticle-based intranasal formulations slow clinical translation.
Manufacturing consistency, scale-up feasibility and quality control of
nanocarriers and complex formulations also remain to be optimized115.
Interindividual
variability and disease-associated changes in nasal physiology call for
personalized delivery approaches to ensure effective dosing and reduce variability
in clinical response116.
Moreover, analysing pharmacokinetics and biodistribution specifically in the
CNS following intranasal administration is technically challenging, requiring
advanced imaging and biomarker techniques117.
Despite
these challenges, advances in nanotechnology, formulation science and device
engineering continue to progress the field towards overcoming these barriers.
Enhanced understanding of nasal anatomy, mucosal immunology and
patient-specific factors, combined with rigorous preclinical safety and
efficacy assessments, will facilitate eventual successful clinical translation118. Concerted collaborative efforts
spanning pharmaceutical sciences, clinical medicine and regulatory bodies are
indispensable to realize the full therapeutic potential of intranasal
neuroprotective agents for diabetic cerebral ischemia and stroke.
8.
Future Perspectives
Intranasal
drug delivery continues to evolve rapidly, offering transformative potential to
enhance neuroprotection in diabetes-associated cerebral ischemia by
circumventing the blood-brain barrier and enabling non-invasive, direct CNS
access111. Emerging
nanotechnologies aim to develop multifunctional Nano carriers capable of
co-delivering therapeutic agents along with targeting ligands, enzyme
inhibitors or imaging moieties, which will improve brain penetration, sustained
release and real-time treatment monitoring112,113.
Advanced lipid-based and polymeric nanocarriers show promise in optimizing
payload stability, mucosal adhesion and selective neuron targeting, thereby
maximizing therapeutic efficacy while minimizing systemic side effects16.
Artificial
intelligence (AI) and machine learning are poised to revolutionize formulation
development and personalized medicine in this field by predicting optimal
carrier characteristics, dose regimes and individualized therapeutic outcomes
based on patient-specific nasal anatomy and metabolic profiles114. Integration of AI-guided therapeutic
design with biomarker-driven clinical monitoring could enable early
intervention tailoring and dynamic therapy adjustments, thus improving clinical
success rates.
The
combination of intranasal delivery with other modalities, such as systemic
treatments or physically guided approaches (e.g., focused ultrasound), may
offer synergistic benefits by addressing the multifaceted pathological changes in
diabetic cerebral ischemia113.
Personalized approaches that account for variability in nasal physiology,
disease severity and genetic predispositions will be increasingly important to
optimize therapeutic index and patient compliance115.
Clinically,
expansion of well-controlled phase II and III trials focusing on diabetic
stroke populations will critically assess safety, dosing and efficacy
parameters of novel intranasal neuroprotective agents111. Additionally, regulatory harmonization
and development of standardized protocols for complex nanocarrier-based
intranasal therapeutics are necessary to accelerate clinical translation and
market approval.
In
conclusion, continued interdisciplinary collaboration among pharmaceutical
scientists, clinicians, engineers and computational biologists is essential to
unlock the full potential of intranasal neuroprotective therapies. This
integrated approach promises a paradigm shift in the management of
diabetes-associated cerebral ischemia, significantly improving patient outcomes
and quality of life.
9.
Conclusion
Intranasal
drug delivery offers a transformative, non-invasive strategy to overcome the
challenges posed by the blood-brain barrier, enabling direct and targeted
delivery of neuroprotective agents to the central nervous system in diabetes-associated
cerebral ischemia107,111. The
vast advances in nanocarrier technology, mucoadhesive formulations and device
designs have significantly improved drug stability, brain targeting and patient
compliance, providing powerful tools for enhancing therapeutic outcomes29,32,44. Compelling preclinical and
emerging clinical data demonstrate the potential for intranasally administered
insulin, melatonin, peptides and other neuroprotectants to modulate oxidative
stress, inflammation, apoptosis and neuronal survival mechanisms critical for ischemic
brain recovery81,86,108.
Despite
encouraging successes, significant translational barriers remain, including the
variability of nasal anatomy and physiology, mucociliary clearance, enzymatic
degradation, regulatory complexities and limitations in clinical trial design26,103,110. Addressing these challenges
requires multidisciplinary efforts spanning pharmaceutical innovation, clinical
research, computational modelling and regulatory harmonization109,112. Novel approaches incorporating
artificial intelligence-guided formulation design, personalized medicine
tailored to patient-specific nasal characteristics and combination therapies
provide promising avenues to enhance clinical efficacy and safety110,111.
In
summary, intranasal delivery of neuroprotective agents marks a promising
frontier in the management of diabetes-associated cerebral ischemia and stroke.
With coordinated efforts to optimize drug design, delivery and clinical
validation, this route could revolutionize therapeutic paradigms-improving
recovery and quality of life for millions worldwide affected by diabetic
stroke. Continued investment in research, clinical trials and regulatory
frameworks is essential to fully realize the clinical potential of this
innovative delivery strategy.
10. Acknowledgments
The
authors thankful to Faculty of Pharmacy, Noble University Junagadh for
providing the facilities to carry out the review work.
11. Conflicts
of Interest
No
conflict of interest was declared by the authors.
12. References
7. Kunte
H, Busch MA, Trostdorf K, et al. Hyperglycemia is associated with infarct
growth in ischemic stroke. Stroke, 2012;43(11): 2995-3000.
18. Ahmad
N, Ahmad R, Alam MA, et al. Nanotechnology-based nose-to-brain drug delivery
for management of neurological disorders. Drug Discov Today, 2017;22(6):
966-972.
24. Illum L. Is
nose-to-brain transport of drugs in man a reality? J Pharm Pharmacol,
2004;56(1): 3-17.
29. Lochhead JJ, Thorne RG. Intranasal drug delivery to the brain.
Pharmaceutics, 2012;4(1): 37-59.
35. Hussain AA.
Intranasal drug delivery. Adv Drug Deliv Rev, 1998;29(1-2): 39-49.
38. Dahl
AR, Hadley WM. Nasal cavity as a route for systemic drug delivery. J Pharm Sci,
1991;80(10): 905-911.
41. Paliwal
R, Paliwal SR, Agrawal GP, et al. Chitosan nanoparticle-based nasal drug
delivery systems. J Control Release, 2015;201: 45-56.
43. Illum L.
Chitosan and its use as a pharmaceutical excipient. Pharm Res, 1998;15(9):
1326-1331.
49. Kanazawa
T. Brain-targeted drug delivery system based on intranasal administration of
nanoparticles. J Pharm Investig, 2015;45(6): 553-560.
71. Illum L.
Chitosan and its use as a pharmaceutical excipient. Pharm Res, 2003;15(9):
1326-1331.
75. Hussain AA.
Intranasal drug delivery. Adv Drug Deliv Rev, 1998;29(1-2): 39-49.
88. Ohtaki
H, Satoh A, Nakamachi T, et al. Regulation of oxidative stress by melatonin in
ischemic brain injury. J Pineal Res, 2008;45(4): 447-454.
90. Beker
MC, Caglayan B, Caglayan AB, et al. Intranasal melatonin attenuates brain
injury after experimental stroke. J Pineal Res, 2015;58(3): 324-334.
92. Spencer JPE.
Flavonoids: Modulators of brain function? Br J Nutr, 2008;99(1): 60-77.
95. Zhang
L, Zhang ZG, Zhang RL, et al. Delayed administration of a small-molecule
activator of Akt reduces ischemic brain damage. Stroke, 2001;32(8): 1905-1910.
97. Lochhead
JJ, Thorne RG. Intranasal drug delivery to the brain: Mechanisms and clinical
potential. Nat Rev Drug Discov, 2015;14(9): 605-622.
98. ElAli
A, Hermann DM. Diabetes and stroke: Pathophysiology and clinical implications.
Nat Rev Neurol, 2012;8(10): 573-579.
99. Li
W, Prakash R, Chawla D, et al. Early inflammatory responses in the diabetic
mouse brain after experimental stroke. Stroke, 2013;44(3): 759-764.
109. American
Diabetes Association. Standards of care in diabetes. Diabetes Care, 2023;46(1):
1-291.
112. Lochhead JJ, Thorne RG. Intranasal drug delivery to the brain.
Pharmaceutics, 2012;4(1): 37-59.
118. Lochhead
JJ, Davis TP. Clinical translation of intranasal drug delivery. Pharm Res,
2019;36(5): 1-15.