Silent Signals in Urine: A Cross Sectional Study on Urinalysis Abnormalities and Antibiotic Resistance in Women
Abstract
Introduction
Urinary tract infections (UTIs) remain a major cause of morbidity among women globally, particularly in low and middle‑income countries. Urinalysis provides rapid detection of abnormalities suggestive of infection while culture and susceptibility testing guide effective therapy. This study evaluated urinalysis abnormalities, bacterial isolates and antibiotic susceptibility patterns among women presenting with urinary symptoms.
Methods
A cross‑sectional study of 100 female participants presenting with urinary symptoms was conducted. Midstream urine samples were subjected to physical, chemical and microscopic examination, followed by culture and antimicrobial susceptibility testing using the Kirby-Bauer disc diffusion method in accordance with CLSI 2023 guidelines.
Results
Proteinuria (45%), pyuria (38%) and haematuria (18%) were the most common abnormalities. Microscopy revealed epithelial cells (79%) and pus cells (38%). A total of 22 bacterial isolates were recovered, with Klebsiella pneumoniae (37%) and Escherichia coli (25%) predominating. Aminoglycosides showed the highest sensitivity (81.3%), while β‑lactams exhibited high resistance (56.3%). Chi-square analysis demonstrated a significant association between antibiotic class and susceptibility outcome (χ² = 30.6, p = 0.001).
Conclusion
Urinalysis abnormalities are important indicators of urinary tract pathology. Culture-based diagnosis remains essential for guiding appropriate therapy. High resistance to β-lactams highlights the need for antimicrobial stewardship in primary healthcare settings.
Keywords: Urinalysis, Urinary tract infection, Antibiotic resistance, Women, Antimicrobial stewardship
1. Introduction
Urinary
tract infections (UTIs) are among the most common bacterial infections
affecting women worldwide1,2.
Symptoms of these infections often present as subtle abnormalities that serve
as signals of underlying disease and contribute significantly to morbidity
among patients, particularly from low and middle‑income countries where
diagnostic facilities are limited and empirical treatment is common3,4.
Women are particularly vulnerable to UTIs due to positioning of their anatomy including shorter urethra and proximity to the perineal region, which facilitate bacterial colonization5-8. Poor management or treatment of recurrent infections can lead to complications including chronic kidney disease and infertility8.
Urinalysis offers a rapid and cost‑effective means of detecting signs of urine infection such as proteinuria, pyuria, hematuria and other abnormalities, that may otherwise go unnoticed9,10. When combined with culture and antibiotic susceptibility testing, urinalysis provides a comprehensive picture of infection burden that could guide evidence‑based therapy11-13. Previous studies across Nigeria and other regions have consistently identified Escherichia coli and Klebsiella pneumoniae as predominant uropathogens, with rising resistance to commonly prescribed antibiotics14,15.
This study was designed to evaluate findings from both urinalysis and antibiotic susceptibility patterns among women presenting with urinary symptoms. By identifying the signals in urine and mapping the trends in resistance patterns, the research aims to strengthen diagnostic accuracy, inform empirical treatment choices and highlight the urgent need for antimicrobial stewardship in women’s health
2. Methods
2.1. Study design and population
This
cross-sectional study recruited 100 female participants aged 6 years and above presenting
with symptoms suggestive of urinary tract infection from Township Primary
Health Care (PHC), Jos, Nigeria. Due to the commercial hub of Jos, Township PHC
is one of the busiest primary healthcare facilities in the metropolis,
recording one of the highest patient attendance rates per year among PHC centres
in the area. It serves as a major healthcare access point for women from both urban
and semi-urban communities within Jos and its surrounding areas.
2.2. Inclusion and exclusion criteria
Women
aged 18 years and above who presented with symptoms suggestive of urinary tract
infection and provided informed consent to participate in the study were
included. Women who had used antibiotics within the two weeks preceding sample
collection, as well as those with a known history of chronic renal disease,
were excluded from participation.
2.3. Sample collection and analysis
Midstream
urine samples were collected from participants using sterile containers. The
samples were transported immediately to the laboratory for analysis. Urinalysis
including physical, chemical and microscopic examinations were performed on the
samples. The physical examination involved assessment of urine colour and
turbidity. Chemical analysis was carried out using standard urine dipstick test
strips to determine parameters such as pH, protein, glucose, ketones, nitrite,
leukocyte esterase, blood and urobilinogen according to the manufacturer’s
instructions. Microscopic examination of the urine sediment was conducted after
centrifugation to detect the presence of pus cells (leukocytes), red blood
cells, epithelial cells and microorganisms.
For bacteriological analysis, urine samples were incubated for 24 hours in media broth and then subculture on MacConkey agar and Blood agar at 37 °C for 18-24 hours. Following incubation, bacterial growth was examined and isolates were identified based on colony morphology, Gram staining characteristics and standard biochemical tests including catalase, coagulase, indole, citrate utilization, urease and oxidase tests, where applicable.
Antimicrobial susceptibility testing of the isolates was performed using the Kirby-Bauer disc diffusion method on Mueller-Hinton agar. Antibiotic discs representing commonly used classes such as fluoroquinolones, β-lactams, aminoglycosides and macrolide, were applied to the inoculated plates. The plates were incubated at 37 °C for 18-24 hours, after which the zones of inhibition were measured and interpreted as sensitive, intermediate or resistant according to the guidelines of the Clinical and Laboratory Standards Institute (CLSI).
2.4. Data analysis
Data
were analysed using SPSS version 27. Descriptive statistics were expressed as
frequencies and percentages. Associations were tested using Chi-square, with
p< 0.05 considered significant. Confidence interval (95% CI) was calculated
where applicable.
3. Results
Table 1: Age Distribution of
Female Participants Included in the Study (n = 100).
|
Age Group (years) |
Frequency (n) |
Percentage (%) |
|
6–18 |
7 |
7 |
|
19–25 |
11 |
11 |
|
26–40 |
57 |
57 |
|
>40 |
25 |
25 |
The
age distribution revealed that participants within the age of 26-40 years (57%)
were the majority of the population, closely followed by participants aged
above 40 years (25.0%). Participants aged, 19-25 years accounted for 11.0% of
the population while participants aged 6-18 years (7%) formed the least
population (Table 1).
Table 2: Distribution of
Urinalysis parameters among study participants (n=100)
|
Characteristic |
Parameter |
Positive (%) |
Negative (%) |
|
Chemical Properties |
Protein (Proteinuria) |
45 |
55 |
|
Blood (Hematuria) |
18 |
82 | |
|
Others (Nitrites,
Ketones, Glucose) |
16 |
84 | |
|
pH (7.0–7.5) |
100 |
0 | |
|
Microscopy |
Pus Cells (Leukocytes) |
38 |
62 |
|
Epithelial Cells |
79 |
21 | |
|
Others (Casts, Yeast
Cells, RBCs) |
21 |
69 |
Urinalysis
revealed notable abnormalities across both chemical and microscopic parameters.
Among the chemical properties analysed, proteinuria was the most prevalent
finding, observed in 45.0% of participants, followed by hematuria (18.0%) and
other abnormalities including nitrites, ketones and glucose (16.0%). Urine pH
within the range of 7.0–7.5 was observed in all samples (100%) (Table 2).
Microscopic examination showed a high presence
of epithelial cells in 79.0% of samples, while pus cells were detected in 38.0%
of participants, indicating inflammatory processes consistent with infection.
Other microscopic findings, including casts, yeast cells and red blood cells,
were present in 21.0% of samples (Figure 1).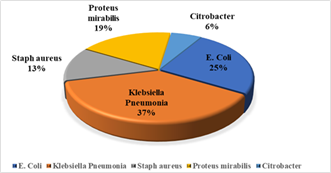
Figure 1: Distribution of uropathogens isolated from urine samples (n = 22).
A total of 22 bacterial isolates were identified, with Gram-negative organisms accounting for the majority of cases. Klebsiella pneumoniae was the most frequently isolated pathogen (37%), followed by Escherichia coli (25%) and Proteus mirabilis (19%). Gram-positive Staphylococcus aureus accounted for 13% of isolates, while Citrobacter species were least represented (6%).
Table 3: Antibiotic Susceptibility Patterns by Antibiotic
Class Among Bacterial Isolates (n = 22)
|
Antibiotic Class |
Sensitive (%) |
Resistant (%) |
Intermediate (%) |
X2 (df) |
P-value |
|
Fluoroquinolones |
68.8 |
17.5 |
13.7 |
|
|
|
β-lactams |
35.4 |
56.3 |
8.3 |
|
|
|
Aminoglycosides |
81.3 |
0 |
18.7 |
30.6 (6) |
0.001 |
|
Macrolides |
75 |
0 |
25 |
|
|
Antibiotic
susceptibility patterns varied significantly across the different antibiotic
classes (χ² = 30.6, p = 0.001). Aminoglycosides demonstrated the highest
sensitivity (81.3%) with no observed resistance, followed by macrolides
(75.0%), which also showed no resistance. Fluoroquinolones exhibited moderate
sensitivity (68.8%) with some degree of resistance (17.5%) and intermediate
response (13.7%). In contrast, β-lactam antibiotics showed the lowest
sensitivity (35.4%) and the highest resistance rate (56.3%), indicating reduced
effectiveness against the bacterial isolates in this study.
4. Discussion
The urinalysis findings in this study
highlight the signs of infection that often precede obvious clinical symptoms.
Proteinuria (45%), pyuria (38%) and hematuria (18%) were the most frequent
abnormalities, underscoring the burden of urinary tract morbidity among women.
These signals, though subtle, provide critical clues to underlying pathology.
The predominance of alkaline urine further supports conditions favorable to
bacterial growth, particularly urea‑splitting organisms such as Proteus mirabilis. Recognizing these
silent signals is essential for timely diagnosis and intervention.
Proteinuria and hematuria, consistent with urinary tract inflammation, align with previous reports from Nigerian and sub‑Saharan African populations16-18. Pyuria, detected in over one‑third of participants, strongly suggests infection and correlates with bacterial culture results. The high prevalence of epithelial cells (79%) likely reflects contamination during sample collection, emphasizing the need for improved patient education. These findings demonstrate that urinalysis, when interpreted carefully, remains a valuable screening tool for women’s health19.
Culture confirmed the presence of uropathogens, with Klebsiella pneumoniae (37%) and Escherichia coli (25%) as the leading isolates. This distribution mirrors global and regional trends, where Gram‑negative organisms dominate UTI etiology20-22. The identification of Staphylococcus aureus, Proteus mirabilis and Citrobacter species, though less frequent, highlights the diversity of pathogens capable of exploiting urinary tract vulnerabilities23,24.
The susceptibility profile revealed concerning resistance trends. β‑lactams exhibited the highest resistance (56.3%), reflecting widespread misuse and over‑prescription in community settings25,26. In contrast, aminoglycosides (Gentamicin, 81.3% sensitivity) and macrolides (Azithromycin, 75% sensitivity) retained strong efficacy, positioning them as viable empirical options. Fluoroquinolones demonstrated variable activity, with some agents showing complete sensitivity while others exhibited moderate resistance. These findings reinforce global concerns about rising resistance and the urgent need for stewardship27,28.
The integration of urinalysis and culture provides a dual lens: urinalysis reveals signals of infection, while culture confirms pathogen identity and resistance. Together, they form a powerful diagnostic framework for guiding therapy 29,30. For women, particularly in resource‑limited settings, this approach can reduce misdiagnosis, improve treatment outcomes and slow the spread of resistance. Strengthening laboratory capacity, ensuring proper urine collection and embedding antimicrobial stewardship into routine care are critical steps toward safeguarding women’s health31,32.
5. Conclusion
This
study highlights the silent signals in urine; proteinuria, pyuria and hematuria,
as common markers of infection among women. Klebsiella pneumoniae and Escherichia
coli were the leading pathogens, reflecting global UTI trends.
Antibiotic susceptibility testing revealed strong activity of aminoglycosides
and macrolides, but alarming resistance to β‑lactams. Recognizing these abnormalities
through routine urinalysis, combined with culture‑based therapy, is vital for
guiding treatment and curbing resistance. Strengthening laboratory capacity,
improving patient education on urine collection and implementing antimicrobial
stewardship programs are critical steps toward safeguarding women’s health and
preserving antibiotic effectiveness.
6. Acknowledgements
The
authors sincerely acknowledge the women who participated in this study, whose
willingness to provide samples made it possible to uncover the silent markers
of urinary health. We extend our gratitude to the laboratory team for their
technical expertise in detecting these signals through urinalysis and culture
and to the clinical staff for their support during data collection.
6.1.
Supplementary page
Table 4: Antibiotic Susceptibility Profile of Bacterial
Isolates to Individual Antibiotics (n = 22).
|
Antibiotic
Class |
Antibiotic |
Sensitive
(%) |
Resistant
(%) |
Intermediate
(%) |
|
Fluoroquinolone |
Levofloxacin |
13
(81.3) |
0 (0) |
3
(18.8) |
|
Sparfloxacin |
2
(12.5) |
9
(56.3) |
5
(31.3) | |
|
Ciprofloxacin |
8
(50.0) |
5
(31.3) |
3
(18.8) | |
|
Pefloxacin |
16
(100) |
0 (0) |
0 (0) | |
|
Ofloxacin |
16
(100) |
0 (0) |
0 (0) | |
|
β-lactam |
Cefotaxime |
14
(87.5) |
2
(12.5) |
0 (0) |
|
Augmentin |
2
(12.5) |
13
(81.3) |
1
(6.3) | |
|
Amoxicillin |
1
(6.30) |
12
(75.0) |
3
(18.8) | |
|
Aminoglycosides |
Gentamicin |
13
(81.3) |
0 (0) |
3
(18.8) |
|
Macrolides |
Azithromycin |
12
(75.0) |
0 (0) |
4
(25.0) |
The
susceptibility profile of bacterial isolates to individual antibiotics revealed
considerable variation across antimicrobial agents. Among the fluoroquinolones,
Pefloxacin and Ofloxacin demonstrated complete sensitivity (100%) with no
observed resistance, while Levofloxacin also showed high sensitivity (81.3%).
In contrast, Sparfloxacin exhibited poor performance, with the highest
resistance rate (56.3%) among this class, while Ciprofloxacin showed moderate
sensitivity (50.0%) with notable resistance (31.3%).
Within the
β-lactam group, Cefotaxime displayed high sensitivity (87.5%) and relatively
low resistance (12.5%), whereas Augmentin and Amoxicillin showed markedly
reduced effectiveness, with resistance rates of 81.3% and 75.0%, respectively.
Aminoglycoside
Gentamicin demonstrated strong activity, with high sensitivity (81.3%) and no
resistance observed. Similarly, the macrolide Azithromycin showed good
efficacy, with 75.0% sensitivity and no resistant isolates, although 25.0% of
isolates exhibited intermediate response. Overall, these findings indicate
substantial variability in antibiotic effectiveness, with notable resistance to
commonly used β-lactam antibiotics.
7. References
1. Kapesa
C, Mumbula EM, Kwenda HC. Prevalence of gram-negative bacterial causes of
urinary tract infection and their antimicrobial susceptibility profile at the
university teaching hospitals in Lusaka, Zambia. Scientific African, 2025;27: 02558.
2. Mancuso G,
Midiri A, Gerace E, et al. Urinary Tract Infections: The Current Scenario and
Future Prospects. Pathogens, 2023;12(4): 623.
3. Mukka
K, Debjit B, Kumar KP. Urinary tract infection: Causes, symptoms, diagnosis and
it's management. Journal of Chemical
and Pharmaceutical
Sciences, 2013;6: 22-28.
4. Kumwenda P, Semu
P. Characterization of urinary tract infections among females at a tertiary
hospital in Malawi: A retrospective study. BMC Infectious
Diseases, 2025;25: 466.
5. Czajkowski
K, Broś-Konopielko M, Teliga-Czajkowska J. Urinary tract infection in
women. Przeglad menopauzalny =
Menopause review, 2021;20(1):
4047.
6. Baimakhanova B,
Sadanov A, Trenozhnikova L, et al. Understanding the Burden and Management of
Urinary Tract Infections inWomen. Diseases, 2025;13(2):59.
7. Anton CI, Ștefan
I, Zamfir M, et al. Etiology and Risk Factors of Recurrent Urinary Tract
Infections in Women in a Multidisciplinary Hospital in
Romania. Microorganisms, 2025;13(3): 626.
8. Bhaduri M,
Sarris I, Bramham K. Female Infertility in Chronic Kidney Disease. Diagnostics, 2023;13(20): 3216.
9. Queremel
Milani DA, Jialal I. Urinalysis. Treasure Island (FL). StatPearls, 2026.
10. Moreland
RB, Brubaker L, Tinawi L, et al. Rapid and accurate testing for urinary tract
infection: new clothes for the emperor.
Clinical microbiology reviews, 2025;38(1): e0012924.
11. Szmulik M,
Trześniewska-Ofiara Z, Mendrycka M, et al. A novel approach to screening and
managing the urinary tract infections suspected sample in the
general human
population. Front. Cell. Infect.
Microbiol, 2022;12: 915288.
12. Froom P, Shimoni
Z. Laboratory Tests, Bacterial Resistance and Treatment Options in Adult
Patients Hospitalized with a Suspected Urinary Tract Infection.
Diagnostics, 2024;14(11): 1078.
13. Al Lawati H,
Blair B, Larnard J. Urinary tract infections: Core curriculum 2024. American Journal of Kidney Diseases, 2023;83(1):
90-100.
14. Mike-Ogburia
MI, Monsi TP, Nwokah EG. Prevalence and determinants of multidrug-resistant
uropathogenic Klebsiella species and associated
antimicrobial resistance genes
in Port Harcourt, Nigeria. BMC
infectious diseases, 2025;25(1): 1036.
15. Olowo-Okere A,
Ibrahim YKE, Nabti LZ, et al. High prevalence of multidrug-resistant
Gram-negative bacterial infections in Northwest
Nigeria. Germs, 2020;10(4): 310-321.
16. Bergstein
JM. Hematuria, proteinuria and urinary tract infections. Paediatric Clinics of North America, 1982;29(1):
55-66.
17. Kayange NM,
Smart LR, Tallman JE, et al. Kidney disease among children in sub-Saharan
Africa: systematic review. Pediatric
research, 2015;77(2):
272281.
18. Braimoh
RW, Akinkugbe AO, Ale OK, et al. Prevalence and pattern of urinary
abnormalities among apparently healthy adult Nigerians. Journal of Clinical Sciences, 2016;13(3): 153-157.
19. Bates
BN. Interpretation of urinalysis and urine culture for UTI treatment. US Pharmacist, 2013;38(11): 65-68
20. Mouanga-Ndzime
Y, Bisseye C, Longo-Pendy NM, et al. Trends in Escherichia coli and Klebsiella pneumoniae Urinary Tract Infections and
Antibiotic Resistance over a 5-Year Period in Southeastern Gabon. Antibiotics, 2024;14(1): 14.
21. Hyun
M, Lee JY, Kim HA, et al. Comparison of Escherichia coli and Klebsiella pneumoniae Acute Pyelonephritis in Korean
Patients. Infection & chemotherapy, 2019;51(2):
130-141.
22. AL Shizawi N, AL
Jabri Z, Khan F, et al. Mapping Antimicrobial Resistance in Escherichia coli and Klebsiella pneumoniae from
Complicated Urinary Tract Infections in Oman: Phenotypic and Genotypic
Insights. Diagnostics, 2025;15(9):
1062.
23. Flores-Mireles
AL, Walker JN, Caparon M, et al. Urinary tract infections: epidemiology,
mechanisms of infection and treatment options. Nature reviews. Microbiology, 2015;13(5): 269-284.
24. Sujith S,
Solomon AP and Rayappan JBB. Comprehensive insights into UTIs: from
pathophysiology to precision diagnosis and management. Front Cell Infect Microbiol, 2024;14: 1402941.
25. Esther
J, Onyebuchi OB, Eugenia OE, et al. Antimicrobial resistance in Nigeria’s
healthcare system: A comprehensive narrative review and policy implications. Discover Public Health, 2025;22: 460.
26. Alabi ED, Rabiu
AG, Adesoji AT. A review of antimicrobial resistance challenges in Nigeria: The
need for a One Health approach. One
Health, 2025;20:101053.
27. Patra
M, Gupta AK, Kumar D, et al. Antimicrobial Resistance: A Rising Global Threat
to Public Health. Infection and
drug resistance, 2025;18:
5419-5437.
28. Salam MA,
Al-Amin MY, Salam MT, et al. Antimicrobial Resistance: A Growing Serious Threat
for Global Public Health. Healthcare,
2023;11(13): 1946.
29. Baimakhanova B,
Sadanov A, Berezin V, et al. Emerging technologies for the diagnosis of urinary
tract infections: Advances in molecular detection and resistance profiling. Diagnostics, 2025;15(19): 2469.
30. Claeys
KC, Weston LE, Pineles L, et al. Implementing diagnostic stewardship to improve
diagnosis of urinary tract infections across three medical centers: A
qualitative assessment. Infection
control and hospital epidemiology, 2023;44(12): 1932-1941.
31. Amadi SC, Daniel
FM, Ikiroma S, et al. Antimicrobial stewardship in resource-limited settings.
In G. Mustafa (Ed.), Antimicrobial stewardship: New insights. IntechOpen,
2024.
32. Foxlee ND,
Townell N, Heney C, et al. Strategies Used for Implementing and Promoting
Adherence to Antibiotic Guidelines in Low- and Lower-Middle Income Countries: A
Systematic Review. Tropical Medicine
and Infectious Disease, 2021;6(3): 166.